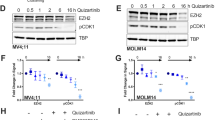
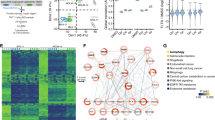
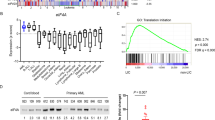
Abstract
Cellular senescence serves as a critical tumor-suppressive mechanism across various cancer types, yet its role in FLT3-ITD-positive acute myeloid leukemia (AML) remains poorly understood. Through the analysis of multiple sequencing datasets, we identified that FLT3-ITD-positive patients with low p16INK4a expression have significantly worse prognoses. Consistent with these clinical findings, knockout of p16INK4a in mice was shown to accelerate FLT3-ITD AML onset. Mechanistic investigations further revealed that the FLT3-ITD mutation suppresses p16INK4a expression via the STAT5A-E2F3-EZH2 signaling axis. This downregulation of p16INK4a allows cells to evade senescence, thereby promoting increased malignancy and establishing a positive feedback loop that exacerbates disease progression. This mechanism provides a molecular explanation for the poorer long-term survival observed in this patient subset. Furthermore, the FLT3-ITD-STAT5A/E2F3/EZH2-p16INK4a axis identified in this study represents a promising therapeutic target for addressing refractory FLT3-ITD AML with low p16INK4a expression.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data sets generated by this study are available through the Genome Sequence Archive, accession number: HRA010951. The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Sun Y-M, Wang W-T, Zeng Z-C, Chen T-Q, Han C, Pan Q, et al. circMYBL2, a circRNA from MYBL2, regulates FLT3 translation by recruiting PTBP1 to promote FLT3-ITD AML progression. Blood. 2019;134:1533–46.
Kottaridis PD, Gale RE, Frew ME, Harrison G, Langabeer SE, Belton AA, et al. The presence of a FLT3 internal tandem duplication in patients with acute myeloid leukemia (AML) adds important prognostic information to cytogenetic risk group and response to the first cycle of chemotherapy: analysis of 854 patients from the United Kingdom Medical Research Council AML 10 and 12 trials. Blood. 2001;98:1752–9.
Oñate G, Pratcorona M, Garrido A, Artigas-Baleri A, Bataller A, Tormo M, et al. Survival improvement of patients with FLT3 mutated acute myeloid leukemia: results from a prospective 9 years cohort. Blood Cancer J. 2023;13:69.
Majothi S, Adams D, Loke J, Stevens SP, Wheatley K, Wilson JS. FLT3 inhibitors in acute myeloid leukaemia: assessment of clinical effectiveness, adverse events and future research-a systematic review and meta-analysis. Syst Rev. 2020;9:285.
Perl AE, Martinelli G, Cortes JE, Neubauer A, Berman E, Paolini S, et al. Gilteritinib or chemotherapy for relapsed or refractory FLT3-mutated AML. N Engl J Med. 2019;381:1728–40.
Cortes JE, Khaled S, Martinelli G, Perl AE, Ganguly S, Russell N, et al. Quizartinib versus salvage chemotherapy in relapsed or refractory FLT3-ITD acute myeloid leukaemia (QuANTUM-R): a multicentre, randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2019;20:984–97.
Stone RM, Mandrekar SJ, Sanford BL, Laumann K, Geyer S, Bloomfield CD, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377:454–64.
Smith CC, Levis MJ, Perl AE, Hill JE, Rosales M, Bahceci E. Molecular profile of FLT3-mutated relapsed/refractory patients with AML in the phase 3 ADMIRAL study of gilteritinib. Blood Adv. 2022;6:2144–55.
Chang E, Ganguly S, Rajkhowa T, Gocke CD, Levis M, Konig H. The combination of FLT3 and DNA methyltransferase inhibition is synergistically cytotoxic to FLT3/ITD acute myeloid leukemia cells. Leukemia. 2016;30:1025–32.
Ravandi F, Alattar ML, Grunwald MR, Rudek MA, Rajkhowa T, Richie MA, et al. Phase 2 study of azacytidine plus sorafenib in patients with acute myeloid leukemia and FLT-3 internal tandem duplication mutation. Blood. 2013;121:4655–62.
Al-Jamal HAN, Mat Jusoh SA, Hassan R, Johan MF. Enhancing SHP-1 expression with 5-azacytidine may inhibit STAT3 activation and confer sensitivity in lestaurtinib (CEP-701)-resistant FLT3-ITD positive acute myeloid leukemia. BMC Cancer. 2015;15:869.
Rayess H, Wang MB, Srivatsan ES. Cellular senescence and tumor suppressor gene p16. Int J Cancer. 2012;130:1715–25.
Campisi J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell. 2005;120:513–22.
Sharpless NE, Sherr CJ. Forging a signature of in vivo senescence. Nat Rev Cancer. 2015;15:397–408.
Quesnel B, Preudhomme C, Philippe N, Vanrumbeke M, Dervite I, Lai JL, et al. p16 gene homozygous deletions in acute lymphoblastic leukemia. Blood. 1995;85:657–63.
Drexler HG. Review of alterations of the cyclin-dependent kinase inhibitor INK4 family genes p15, p16, p18 and p19 in human leukemia-lymphoma cells. Leukemia. 1998;12:845–59.
Liggett WH, Sidransky D. Role of the p16 tumor suppressor gene in cancer. J Clin Oncol J Am Soc Clin Oncol. 1998;16:1197–206.
Gündogdu MS, Liu H, Metzdorf D, Hildebrand D, Aigner M, Aktories K, et al. The haematopoietic GTPase RhoH modulates IL3 signalling through regulation of STAT activity and IL3 receptor expression. Mol Cancer. 2010;9:225.
Terada K, Kaziro Y, Satoh T. Ras is not required for the interleukin 3-induced proliferation of a mouse pro-B cell line, BaF3. J Biol Chem. 1995;270:27880–6.
Shibayama H, Anzai N, Braun SE, Fukuda S, Mantel C, Broxmeyer HE. H-Ras is involved in the inside-out signaling pathway of interleukin-3-induced integrin activation. Blood. 1999;93:1540–8.
Bolouri H, Farrar JE, Triche T, Ries RE, Lim EL, Alonzo TA, et al. The molecular landscape of pediatric acute myeloid leukemia reveals recurrent structural alterations and age-specific mutational interactions. Nat Med. 2018;24:103–12.
Lv K, Ren J-G, Han X, Gui J, Gong C, Tong W. Depalmitoylation rewires FLT3-ITD signaling and exacerbates leukemia progression. Blood. 2021;138:2244–55.
Schwab C, Ryan SL, Chilton L, Elliott A, Murray J, Richardson S, et al. EBF1-PDGFRB fusion in pediatric B-cell precursor acute lymphoblastic leukemia (BCP-ALL): genetic profile and clinical implications. Blood. 2016;127:2214–8.
Cabezas-Wallscheid N, Klimmeck D, Hansson J, Lipka DB, Reyes A, Wang Q, et al. Identification of regulatory networks in HSCs and their immediate progeny via integrated proteome, transcriptome, and DNA methylome analysis. Cell Stem Cell. 2014;15:507–22.
Rong Q-X, Wang F, Guo Z-X, Hu Y, An S-N, Luo M, et al. GM-CSF mediates immune evasion via upregulation of PD-L1 expression in extranodal natural killer/T cell lymphoma. Mol Cancer. 2021;20:80.
Suske T, Sorger H, Manhart G, Ruge F, Prutsch N, Zimmerman MW, et al. Hyperactive STAT5 hijacks T cell receptor signaling and drives immature T cell acute lymphoblastic leukemia. J Clin Invest. 2024;134:e168536.
Pan Y-M, Wang C-G, Zhu M, Xing R, Cui J-T, Li W-M, et al. STAT3 signaling drives EZH2 transcriptional activation and mediates poor prognosis in gastric cancer. Mol Cancer. 2016;15:79.
Riquelme E, Suraokar M, Behrens C, Lin HY, Girard L, Nilsson MB, et al. VEGF/VEGFR-2 upregulates EZH2 expression in lung adenocarcinoma cells and EZH2 depletion enhances the response to platinum-based and VEGFR-2-targeted therapy. Clin Cancer Res J Am Assoc Cancer Res. 2014;20:3849–61.
Maurer B, Nivarthi H, Wingelhofer B, Pham HTT, Schlederer M, Suske T, et al. High activation of STAT5A drives peripheral T-cell lymphoma and leukemia. Haematologica. 2020;105:435–47.
Cortes J. Quizartinib: a potent and selective FLT3 inhibitor for the treatment of patients with FLT3-ITD-positive AML. J Hematol Oncol J Hematol Oncol. 2024;17:111.
Di Micco R, Krizhanovsky V, Baker D, d’Adda di Fagagna F. Cellular senescence in ageing: from mechanisms to therapeutic opportunities. Nat Rev Mol Cell Biol. 2021;22:75–95.
Zimmerman EI, Turner DC, Buaboonnam J, Hu S, Orwick S, Roberts MS, et al. Crenolanib is active against models of drug-resistant FLT3-ITD-positive acute myeloid leukemia. Blood. 2013;122:3607–15.
Gozgit JM, Wong MJ, Wardwell S, Tyner JW, Loriaux MM, Mohemmad QK, et al. Potent activity of ponatinib (AP24534) in models of FLT3-driven acute myeloid leukemia and other hematologic malignancies. Mol Cancer Ther. 2011;10:1028–35.
Acknowledgements
This work was supported by a Ministry of Science and Technology of the People’s Republic of China grant (2021YFA1101603), Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences grants (CIFMS) (2023-I2M-C&T-B-106, 2021-I2M-1-040, 2023-I2M-2-007, 2022-I2M-1-022), the National Natural Science Foundation of China (82270144, 82100198), and the Haihe Laboratory of Cell Ecosystem Innovation Fund (22HHXBSS00039).
Author contibutions
These authors jointly supervised this work: Wenyu Yang, Tianyuan Hu, and Yingchi Zhang. Y.Z., W.Y. and X.Z. conceived the study; Y.Z. and T.H. designed the project outline and acquired its main funding, supervised experimental studies, helped write the manuscript; T.H. and J.Z. determined the methodology; J.Z. wrote the manuscript, carried out experiments, drew figures; L.J. carried out experiments, drew figures; Y.C. carried out experiments, drew figures; S.Z. carried out experiments; Y.D. carried out experiments; P.W. drew figures, reviewed the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declarations
Protocols covering all study procedures were approved by the institutional review board of the Institute of Hematology, Blood Diseases Hospital, Peking Union Medical College/Chinese Academy of Medical Sciences (PUMC/CAMS). All patients were provided written informed consent to participate in studies and for the research use of their specimens in agreement with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, J., Jin, L., Chen, Y. et al. Targeting p16INK4a-mediated cellular senescence as a therapeutic strategy for FLT3-ITD-driven acute myeloid leukemia. Leukemia 39, 2652–2662 (2025). https://doi.org/10.1038/s41375-025-02743-y
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41375-025-02743-y