Abstract
Waldenström macroglobulinemia (WM) is a rare indolent B-cell lymphoma with marked clinical and molecular heterogeneity. Clinical risk models, including IPSSWM, rIPSSWM, and MSSWM, were developed prior to the widespread use of Bruton tyrosine kinase inhibitors (BTKi), and their performance in the BTKi era remains uncertain. In addition, the prognostic impact of various genomic alterations is controversial. We retrospectively analyzed 453 symptomatic WM patients, including 203 who received non-BTKi therapy and 250 who received BTKi-based therapy. All three models significantly stratified prognosis in the non-BTKi cohort, with rIPSSWM showing the highest predictive accuracy, but none effectively predicted survival in BTKi-treated patients. Notably, among patients receiving first-line BTKi-based therapy, high-risk patients by any model achieved survival outcomes comparable to those of lower-risk patients, suggesting that upfront BTKi can overcome the adverse impact of high-risk clinical features. At the molecular level, MYD88 mutation was significantly associated with favorable outcomes exclusively in patients treated with first-line BTKi-based therapy, while CXCR4 and TP53 mutations predicted significantly inferior prognosis in both BTKi-based and non-BTKi cohorts. Our findings indicate that although clinical risk models remain relevant for patients receiving non-BTKi therapy, molecular features, especially MYD88, CXCR4, and TP53 mutations, provide superior prognostic insights for patients with BTKi-based regimens.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Owen RG, Treon SP, Al-Katib A, Fonseca R, Greipp PR, McMaster ML, et al. Clinicopathological definition of Waldenstrom’s macroglobulinemia: consensus panel recommendations from the Second International Workshop on Waldenstrom’s Macroglobulinemia. Semin Oncol. 2003;30:110–5.
Kumar SK, Callander NS, Adekola K, Anderson LD Jr, Baljevic M, Baz R, et al. Waldenström macroglobulinemia/lymphoplasmacytic lymphoma, version 2.2024, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2024;22. e240001.
Gertz MA. Waldenström macroglobulinemia: 2025 update on diagnosis, risk stratification, and management. Am J Hematol. 2025;100:1061–73.
Dimopoulos MA, Kastritis E. How I treat Waldenström macroglobulinemia. Blood. 2019;134:2022–35.
Treon SP, Xu L, Yang G, Zhou Y, Liu X, Cao Y, et al. MYD88 L265P somatic mutation in Waldenström’s macroglobulinemia. New Engl J Med. 2012;367:826–33.
Hunter ZR, Xu L, Yang G, Zhou Y, Liu X, Cao Y, et al. The genomic landscape of Waldenstrom macroglobulinemia is characterized by highly recurring MYD88 and WHIM-like CXCR4 mutations, and small somatic deletions associated with B-cell lymphomagenesis. Blood. 2014;123:1637–46.
Treon SP, Cao Y, Xu L, Yang G, Liu X, Hunter ZR. Somatic mutations in MYD88 and CXCR4 are determinants of clinical presentation and overall survival in Waldenstrom macroglobulinemia. Blood. 2014;123:2791–6.
Poulain S, Roumier C, Venet-Caillault A, Figeac M, Herbaux C, Marot G, et al. Genomic landscape of CXCR4 mutations in Waldenström macroglobulinemia. Clin Cancer Res. 2016;22:1480–8.
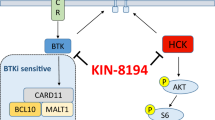
Yang G, Zhou Y, Liu X, Xu L, Cao Y, Manning RJ, et al. A mutation in MYD88 (L265P) supports the survival of lymphoplasmacytic cells by activation of Bruton tyrosine kinase in Waldenström macroglobulinemia. Blood. 2013;122:1222–32.
Yang G, Buhrlage SJ, Tan L, Liu X, Chen J, Xu L, et al. HCK is a survival determinant transactivated by mutated MYD88, and a direct target of ibrutinib. Blood. 2016;127:3237–52.
Castillo JJ, Sarosiek SR, Gustine JN, Flynn CA, Leventoff CR, White TP, et al. Response and survival predictors in a cohort of 319 patients with Waldenström macroglobulinemia treated with ibrutinib monotherapy. Blood Adv. 2022;6:1015–24.
Treon SP, Xu L, Guerrera ML, Jimenez C, Hunter ZR, Liu X, et al. Genomic landscape of Waldenström macroglobulinemia and its impact on treatment strategies. J Clin Oncol. 2020;38:1198–208.
Morel P, Duhamel A, Gobbi P, Dimopoulos MA, Dhodapkar MV, McCoy J, et al. International prognostic scoring system for Waldenstrom macroglobulinemia. Blood. 2009;113:4163–70.
Kastritis E, Morel P, Duhamel A, Gavriatopoulou M, Kyrtsonis MC, Durot E, et al. A revised international prognostic score system for Waldenström’s macroglobulinemia. Leukemia. 2019;33:2654–61.
Zanwar S, Le-Rademacher J, Durot E, D’Sa S, Abeykoon JP, Mondello P, et al. Simplified risk stratification model for patients with Waldenström macroglobulinemia. J Clin Oncol. 2024;42:2527–36.
Treon SP, Gustine J, Xu L, Manning RJ, Tsakmaklis N, Demos M, et al. MYD88 wild-type Waldenstrom macroglobulinaemia: differential diagnosis, risk of histological transformation, and overall survival. Br J Haematol. 2018;180:374–80.
Abeykoon JP, Paludo J, King RL, Ansell SM, Gertz MA, LaPlant BR, et al. MYD88 mutation status does not impact overall survival in Waldenström macroglobulinemia. Am J Hematol. 2018;93:187–94.
Wang Y, Gali VL, Xu-Monette ZY, Sano D, Thomas SK, Weber DM, et al. Molecular and genetic biomarkers implemented from next-generation sequencing provide treatment insights in clinical practice for Waldenström macroglobulinemia. Neoplasia. 2021;23:361–74.
Krzisch D, Guedes N, Boccon-Gibod C, Baron M, Bravetti C, Davi F, et al. Cytogenetic and molecular abnormalities in Waldenström’s macroglobulinemia patients: correlations and prognostic impact. Am J Hematol. 2021;96:1569–79.
Yan Y, Yu Y, Xiong W, Wang J, Yao Y, Jia Y, et al. Determination of MYD88 and CXCR4 mutations for clinical detection and their significance in Waldenström macroglobulinemia. Clin Cancer Res. 2024;30:5483–93.
Chan WL, Chong VCL, Wee IJY, Poon LM, Chan EHL, Lee J, et al. Efficacy and safety of front-line treatment regimens for Waldenstrom macroglobulinaemia: a systematic review and meta-analysis. Blood cancer J. 2023;13:140.
Liu Z, Jiang S, Gu J, Liu H, Song G, Cao X. Bortezomib-based chemotherapy for patients with Waldenström macroglobulinemia: a single-center experience. Ann Hematol. 2023;102:167–74.
Treon SP, Ioakimidis L, Soumerai JD, Patterson CJ, Sheehy P, Nelson M, et al. Primary therapy of Waldenström macroglobulinemia with bortezomib, dexamethasone, and rituximab: WMCTG clinical trial 05-180. J Clin Oncol. 2009;27:3830–5.
Buske C, Dimopoulos MA, Grunenberg A, Kastritis E, Tomowiak C, Mahé B, et al. Bortezomib-dexamethasone, rituximab, and cyclophosphamide as first-line treatment for Waldenström’s macroglobulinemia: a prospectively randomized trial of the European Consortium for Waldenström’s macroglobulinemia. J Clin Oncol. 2023;41:2607–16.
Xiong W, Lyu R, Yu Y, Wang T, Yan Y, Wang Y, et al. Rituximab plus cyclophosphamide and dexamethasone versus bortezomib plus cyclophosphamide and dexamethasone in newly diagnosed symptomatic Waldenström macroglobulinemia: a randomized controlled trial. Haematologica. 2024;109:1614–8.
Treon SP, Xu L, Hunter Z. MYD88 mutations and response to ibrutinib in Waldenström’s macroglobulinemia. New Engl J Med. 2015;373:584–6.
Dimopoulos M, Sanz RG, Lee HP, Trneny M, Varettoni M, Opat S, et al. Zanubrutinib for the treatment of MYD88 wild-type Waldenström macroglobulinemia: a substudy of the phase 3 ASPEN trial. Blood Adv. 2020;4:6009–18.
Castillo JJ, Xu L, Gustine JN, Keezer A, Meid K, Dubeau TE, et al. CXCR4 mutation subtypes impact response and survival outcomes in patients with Waldenström macroglobulinaemia treated with ibrutinib. Br J Haematol. 2019;187:356–63.
Gustine JN, Tsakmaklis N, Demos MG, Kofides A, Chen JG, Liu X, et al. TP53 mutations are associated with mutated MYD88 and CXCR4, and confer an adverse outcome in Waldenström macroglobulinaemia. Br J Haematol. 2019;184:242–5.
Tam CS, Opat S, D’Sa S, Jurczak W, Lee HP, Cull G, et al. Biomarker analysis of the ASPEN study comparing zanubrutinib with ibrutinib for patients with Waldenström macroglobulinemia. Blood Adv. 2024;8:1639–50.
Nguyen-Khac F, Lambert J, Chapiro E, Grelier A, Mould S, Barin C, et al. Chromosomal aberrations and their prognostic value in a series of 174 untreated patients with Waldenström’s macroglobulinemia. Haematologica. 2013;98:649–54.
Schmitz R, Wright GW, Huang DW, Johnson CA, Phelan JD, Wang JQ, et al. Genetics and pathogenesis of diffuse large B-cell lymphoma. N Engl J Med. 2018;378:1396–407.
Gonzalez-Aguilar A, Idbaih A, Boisselier B, Habbita N, Rossetto M, Laurenge A, et al. Recurrent mutations of MYD88 and TBL1XR1 in primary central nervous system lymphomas. Clin Cancer Res. 2012;18:5203–11.
Panea RI, Love CL, Shingleton JR, Reddy A, Bailey JA, Moormann AM, et al. The whole-genome landscape of Burkitt lymphoma subtypes. Blood. 2019;134:1598–607.
Venturutti L, Teater M, Zhai A, Chadburn A, Babiker L, Kim D, et al. TBL1XR1 mutations drive extranodal lymphoma by inducing a pro-tumorigenic memory fate. Cell. 2020;182:297–316.e27.
Hosoi H, Tabata S, Kosako H, Hori Y, Okamura T, Yamashita Y, et al. IGLL5, controlled by a super-enhancer, affects cell survival and MYC expression in mature B-cell lymphoma. Leuk Res Rep. 2024;21:100451.
Varettoni M, Zibellini S, Defrancesco I, Ferretti VV, Rizzo E, Malcovati L, et al. Pattern of somatic mutations in patients with Waldenström macroglobulinemia or IgM monoclonal gammopathy of undetermined significance. Haematologica. 2017;102:2077–85.
Hunter ZR, Yang G, Xu L, Liu X, Castillo JJ, Treon SP. Genomics, signaling, and treatment of waldenström macroglobulinemia. J Clin Oncol. 2017;35:994–1001.
Ngo VN, Young RM, Schmitz R, Jhavar S, Xiao W, Lim KH, et al. Oncogenically active MYD88 mutations in human lymphoma. Nature. 2011;470:115–9.
Shuai W, Lin P, Strati P, Patel KP, Routbort MJ, Hu S, et al. Clinicopathological characterization of chronic lymphocytic leukemia with MYD88 mutations: L265P and non-L265P mutations are associated with different features. Blood Cancer J. 2020;10:86.
Kim JA, Hwang B, Park SN, Huh S, Im K, Choi S, et al. Genomic profile of chronic lymphocytic leukemia in Korea identified by targeted sequencing. PloS one. 2016;11:e0167641.
Fu H, Wang T, Yang Y, Qiu C, Wang H, Qiu Y, et al. Next-generation sequencing of circulating tumor DNA in cerebrospinal fluid for detecting gene mutations in central nervous system lymphoma patients. Ther Adv Hematol. 2025;16:20406207251321721.
Acknowledgements
This work was supported by grants from the National Nature Science Foundation of China (82200215, 82570248, 82370197, 82170194), the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (2025-I2M-C&T-B-074), Beijing Xisike Clinical Oncology Research Foundation (Y-2024AZ(BTK)ZD--0074) and the Shenzhen Science and Technology Innovation Commission (JCYJ20240813140803005).
Author information
Authors and Affiliations
Consortia
Contributions
SHY conceptualized the study design. YTY and WHC collected patients’ data and wrote the manuscript. YTY and WHC conducted data analysis and statistics. YY, YXL, WJX, XYZ, TYW, WL, WWS, ZY, MH, GA, and DHZ acquired the data and managed the patients. LGQ and SHY revised the manuscript critically and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yan, Y., Chen, W., Yu, Y. et al. Prognostic significance of clinical risk models and genomic alterations in Waldenström macroglobulinemia before or after the BTK inhibitor era. Leukemia (2026). https://doi.org/10.1038/s41375-026-02919-0
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41375-026-02919-0