Abstract
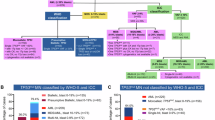
Myelodysplastic neoplasms (MDS) with TP53 multihit alterations are associated with dismal outcomes. MDS with isolated del(5q) present favorable prognosis but is defined by the absence of TP53 multihit alterations. However, whether TP53 multihit alterations exert the same adverse impact in this genetic context remains uncertain. We retrospectively analyzed the characteristics and outcome of 43 patients with MDS with isolated del(5q) harboring TP53 multihit alterations (MDS-del(5q) TP53 multihit) and compared with 68 patients with low-blast MDS with TP53 multihit and without isolated del(5q) (MDS-LB TP53 multihit). Patients with MDS-del(5q) TP53 multihit showed significantly higher platelet counts, more frequent SF3B1 mutations, were less often classified as high-risk by IPSS-R or IPSS-M, and had significantly better outcomes than patients with MDS-LB TP53 multihit: overall survival of 70.2 vs 13.9 months, and time to acute myeloid leukemia progression (AML) of 31.9 vs 7.2 months, respectively. Moreover, the superior outcomes of MDS-del(5q) TP53 multihit patients persisted significant even when compared with MDS-LB TP53 multihit cases without complex karyotype (survival of 70.2 vs 39.9 months; time to AML progression of 31.9 vs 11.4 months). These findings indicate that, in MDS-del(5q) the adverse impact of TP53 multihit alterations may be less important than in other MDS subtypes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Data have been deposited in European Genome-Phenome Archive (EGA) at https://ega-archive.org/studies/EGAS50000000649 (accession number EGAS50000000649).
References
Khoury JD, Solary E, Abla O, Arber DA, Hasserjian RP, Orazi A, et al. The 5th edition of the WHO classification of haematolymphoid tumours: myeloid and histiocytic/dendritic neoplasms. Leukemia. 2022;36:1703–19.
Arber D, Orazi A, Hasserjian RP, Borowitz MJ, Beau MM, Vardiman JW, et al. International consensus classification of myeloid neoplasms and acute leukaemias: integrating morphologic, clinical, and genomic data. Blood. 2022;140:1200–28.
Fenaux P, Giagounidis A, Selleslag D, Beyne-Rauzy O, Mufti G, Mittelman M, et al. A randomized phase 3 study of lenalidomide versus placebo in RBC transfusion-dependent patients with Low-/Intermediate-1-risk myelodysplastic syndromes with del5q. Blood. 2011;118:3765–76.
Germing U, Lauseker M, Hildebrandt B, Symeonidis A, Cermak J, Fenaux P, et al. Survival, prognostic factors and rates of leukaemic transformation in 381 untreated patients with MDS and del(5q): a multicentre study. Leukemia. 2012;26:1286–92.
Bernard E, Nannya Y, Hasserjian RP, Devlin SM, Tuechler H, Abdel-Wahab O, et al. Implications of TP53 allelic state for genome stability, clinical presentation and outcomes in MDS. Nat Med. 2020;26:1549–56.
Komrokji RS, Thol F, Al Ali NH, Padron E, Fenaux P, Platzbecker U, et al. Data-driven, harmonised classification for MDS: a consensus paper from the International Consortium. Lancet Haematol. 2023;10:e519–28.
Montoro MJ, Palomo L, Haferlach C, Malcovati L, Elena C, Cervera J, et al. Influence of TP53 mutations and their allelic status in MDS with isolated 5q deletion. Blood. 2024;144:1722–31.
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Beau MM, et al. The 2016 revision to the WHO classification of myeloid neoplasms and acute leukaemia. Blood. 2016;127:2391–405.
Greenberg PL, Tuechler H, Schanz J, Sanz G, Garcia-Manero G, Sole F, et al. Revised international prognostic scoring system for MDS. Blood. 2012;120:2454–65.
Bernard E, Tuechler H, Greenberg PL, Hasserjian RP, Arango O, Bejar R, et al. Molecular international prognostic scoring system for MDS. N Engl J Med Evid. 2022;1:1–12.
Palomo L, Ibáñez M, Abaigar M, García O, Marcos E, Velez E, et al. Spanish guidelines for the use of targeted deep sequencing in myelodysplastic syndromes and chronic myelomonocytic leukaemia. Br J Haematol. 2020;188:605–22.
Meggendorfer M, Haferlach C, Kern W, Nadarajah N, Alpermann T, Falini B, et al. Molecular analysis of MDS with isolated 5q deletion reveals a specific mutation spectrum: a study on 123 patients. Haematologica. 2017;102:1502–10.
Chan O, Ali NA, Sallman D, Padron E, Komrokji R, Lancet J, et al. Therapeutic outcomes and impact of gene mutations including TP53 and SF3B1 in MDS with del(5q). Clin Lymphoma Myeloma Leuk. 2022;22:e467–76.
Jädersten M, Saft L, Smith A, Kulasekararaj A, Pomplun S, Göhring G, et al. TP53 mutations in low-risk myelodysplastic syndromes with del(5q) predict disease progression. J Clin Oncol. 2011;29:1971–9.
Mallo M, del Rey M, Ibáñez M, Calasanz MJ, Arenillas L, Larrayoz MJ, et al. Response to lenalidomide in myelodysplastic syndromes with del(5q): influence of cytogenetics and mutations. Br J Haematol. 2013;162:74–86.
Díez-Campelo M, López-Cadenas F, Xicoy B, Lumbreras E, González T, del Rey González M, et al. Low-dose lenalidomide versus placebo in non-transfusion-dependent patients with low-risk del(5q) myelodysplastic syndromes (SintraREV): a randomised, double-blind, phase 3 trial. Lancet Haematol. 2024;11:e659–70.
Author information
Authors and Affiliations
Contributions
MJM and DV designed the study, analyzed the data and wrote the manuscript. VN performed statistical analysis. TG, TB, FL, BX, NDV and AJ collected clinical data and contributed to patient management. PA, LP, CH, MMg, FSol, FS, ES, BB, and TH performed molecular and genomic analyses. OC, NHA, TK, AMZ, RSK, CG, JM, AAK, MS, RB, YW, DHW, AK, UP, DH, UG, and PF contributed to data acquisition, interpretation, and critical revision of the manuscript. YK, AC, FB, VS, AP, ED, MM, CZ, MDC, MGDP, and GGM contributed to data interpretation and manuscript revision. DV supervised the study and critically revised the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
MJM: Received honoraria for consulting and scientific support from Jazz Pharmaceuticals; AA-K: Received honoraria from Celgene-BMS/Novartis/Astex/ALX Oncology/Aprea/Medimmune-AstraZeneca/H3B-Hemavant: research support. Novartis: scientific steering committee membership; AMZ participated in advisory boards, consulted, participated in clinical trial committees, and/or received honoraria from AbbVie, Akesobio, Agios, Amgen, Astellas, BioCryst, Beigene, Boehringer-Ingelheim, Celgene/BMS, Chiesi/Cornerstone biopharma, Daiichi Sankyo, Dr Reddy, Epizyme, Faron, Fibrogen, GSK, Glycomimetics, Genentech, Gilead, Geron, Janssen, Jasper, Karyopharm, Kyowa Kirin, Keros, Kura, Novartis, Notable, Orum, Otsuka, Pfizer, Regeneron, Rigel, Seattle Genetics, Shattuck labs, Schrodinger, Syros, Syndax, Servier, Takeda, Treadwell, Taiho, Vincerx, and Zentalis; DV: participated in advisory boards, consulted, participated in clinical trial committees, and/or received honoraria from AbbVie, Agios, Amgen, Asofarma, Astellas, Celgene/BMS, Grifols, Janssen, Jazz, MSD, Novartis, Pfizer, Sanofy, Servier, Sobi, Syros and Takeda.
Ethics approval and consent to participate
The study was conducted in accordance with the principles of the Declaration of Helsinki. The study protocol was approved by the Clinical Research Ethics Committee of Vall d’Hebron University Hospital (PR(AG)497/2018). All patients provided written informed consent. Additional approvals were obtained from the local ethics committees of participating centers when required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Montoro, M.J., Acha, P., Haferlach, C. et al. How should myelodysplastic neoplasms with isolated deletion 5q and TP53 multihit alterations be classified?. Leukemia (2026). https://doi.org/10.1038/s41375-026-02939-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41375-026-02939-w