Abstract
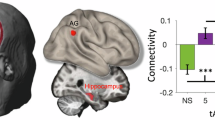
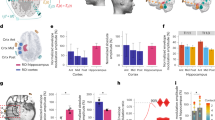
Non-invasive brain stimulation is promising for treating many neuropsychiatric and neurological conditions. It could be optimized by understanding its intracranial responses in different brain regions. We implanted multi-site intracranial electrodes and systematically assessed the acute responses in these regions to transcranial alternating current stimulation (tACS) at different frequencies. We observed robust neural oscillation changes in the hippocampus and amygdala in response to non-invasive tACS procedures, and these effects were frequency-specific and state-dependent. Notably, the hippocampus responded most strongly and stably to 10 Hz stimulation, with pronounced changes across a wide frequency range, suggesting the potential of 10 Hz oscillatory stimulation to modulate a broad range of neural activity related to cognitive functions. Future work with increased sample sizes is required to determine the clinical implications of these findings for therapeutic efficiency.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are openly available at the Open Science Foundation repository (https://doi.org/10.17605/OSF.IO/G9BCT).
References
Polanía R, Nitsche MA, Ruff CC. Studying and modifying brain function with non-invasive brain stimulation. Nat Neurosci. 2018;21:174–87.
Grover S, Nguyen JA, Reinhart RMG. Synchronizing brain rhythms to improve cognition. Annu Rev Med. 2021;72:29–43.
Krause MR, Vieira PG, Csorba BA, Pilly PK, Pack CC. Transcranial alternating current stimulation entrains single-neuron activity in the primate brain. Proc Natl Acad Sci USA. 2019;116:5747–55.
Axmacher N, Henseler MM, Jensen O, Weinreich I, Elger CE, Fell J. Cross-frequency coupling supports multi-item working memory in the human hippocampus. Proc Natl Acad Sci USA. 2010;107:3228–33.
Grover S, Wen W, Viswanathan V, Gill CT, Reinhart RMG. Long-lasting, dissociable improvements in working memory and long-term memory in older adults with repetitive neuromodulation. Nat Neurosci. 2022;25:1237–46.
Zhou D, Li A, Li X, Zhuang W, Liang Y, Zheng C-Y, et al. Effects of 40 Hz transcranial alternating current stimulation (tACS) on cognitive functions of patients with Alzheimer’s disease: a randomised, double-blind, sham-controlled clinical trial. J Neurol Neurosurg Psychiatry. 2022;93:568–70.
Benussi A, Cantoni V, Grassi M, Brechet L, Michel CM, Datta A, et al. Increasing brain gamma activity improves episodic memory and restores cholinergic dysfunction in Alzheimer’s disease. Annu Neurol. 2022;92:322–34.
Simons JS, Ritchey M, Fernyhough C. Brain mechanisms underlying the subjective experience of remembering. Annu Rev Psychol. 2022;73:159–86.
Tavakoli AV, Yun K. Transcranial alternating current stimulation (tACS) mechanisms and protocols. Front Cell Neurosci. 2017;11:214.
Underwood E. Cadaver study challenges brain stimulation methods. Science. 2016;352:397.
Opitz A, Falchier A, Linn GS, Milham MP, Schroeder CE. Limitations of ex vivo measurements for in vivo neuroscience. Proc Natl Acad Sci. 2017;114:5243–6.
Gallen CL, D’Esposito M. Brain modularity: a biomarker of intervention-related plasticity. Trends Cognit Sci. 2019;23:293–304.
Johnson L, Alekseichuk I, Krieg J, Doyle A, Yu Y, Vitek J, et al. Dose-dependent effects of transcranial alternating current stimulation on spike timing in awake nonhuman primates. Sci Adv. 2020;6:eaaz2747.
Huang WA, Stitt IM, Negahbani E, Passey DJ, Ahn S, Davey M, et al. Transcranial alternating current stimulation entrains alpha oscillations by preferential phase synchronization of fast-spiking cortical neurons to stimulation waveform. Nat Commun. 2021;12:3151.
Helfrich RF, Schneider TR, Rach S, Trautmann-Lengsfeld SA, Engel AK, Herrmann CS. Entrainment of brain oscillations by transcranial alternating current stimulation. Curr Biol. 2014;24:333–9.
Kurmann R, Gast H, Schindler K, Fröhlich F. Rational design of transcranial alternating current stimulation: identification, engagement, and validation of network oscillations as treatment targets. Clin Transl Neurosci. 2018;2:33. 2514183×18793515.
Bradley C, Nydam AS, Dux PE, Mattingley JB. State-dependent effects of neural stimulation on brain function and cognition. Nat Rev Neurosci. 2022;23:459–75.
Lafon B, Henin S, Huang Y, Friedman D, Melloni L, Thesen T, et al. Low frequency transcranial electrical stimulation does not entrain sleep rhythms measured by human intracranial recordings. Nat Commun. 2017;8:1199.
Lee TL, Lee H, Kang N. A meta-analysis showing improved cognitive performance in healthy young adults with transcranial alternating current stimulation. npj Sci Learn. 2023;8:1.
Wischnewski M, Alekseichuk I, Opitz A. Neurocognitive, physiological, and biophysical effects of transcranial alternating current stimulation. Trends Cognit Sci. 2023;27:189–205.
Chen S, Tan Z, Xia W, Gomes CA, Zhang X, Zhou W, et al. Theta oscillations synchronize human medial prefrontal cortex and amygdala during fear learning. Sci Adv. 2021;7:eabf4198.
Zheng J, Stevenson RF, Mander BA, Mnatsakanyan L, Hsu FPK, Vadera S, et al. Multiplexing of theta and alpha rhythms in the amygdala-hippocampal circuit supports pattern separation of emotional information. Neuron. 2019;102:887–98.
Pacheco Estefan D, Sánchez-Fibla M, Duff A, Principe A, Rocamora R, Zhang H, et al. Coordinated representational reinstatement in the human hippocampus and lateral temporal cortex during episodic memory retrieval. Nat Commun. 2019;10:2255.
Han S, Li XX, Wei S, Zhao D, Ding J, Xu Y, et al. Orbitofrontal cortex-hippocampus potentiation mediates relief for depression: a randomized double-blind trial and TMS-EEG study. Cell Rep Med. 2023;4:101060.
Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, et al. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron. 2002;33:341–55.
Brunyé TT, Beaudoin ME, Feltman KA, Heaton KJ, McKinley RA, Vartanian O et al. Neuroenhancement in military personnel: conceptual and methodological promises and challenges. In: NATO Symposium on Applying Neuroscience to Performance: From Rehabilitation to Human Cognitive 2021, Rome, Italy, 11-12 October 2021. 2022
Higgins N, Forlini C, Butorac I, Gardner J, Carter A. Anticipating the future of neurotechnological enhancement. In: The Routledge Handbook of the Ethics of Human Enhancement. New York, NY, USA: Routledge. 2024. p. 237–50.
Illes J, Hossain S. Neuroethics: anticipating the future. New York, NY, USA: Oxford University Press; 2017.
Thielscher A, Antunes A, Saturnino GB. Field modeling for transcranial magnetic stimulation: a useful tool to understand the physiological effects of TMS? In: 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). Milan, Italy. IEEE; 2015. p. 222–5.
Puonti O, Van Leemput K, Saturnino GB, Siebner HR, Madsen KH, Thielscher A. Accurate and robust whole-head segmentation from magnetic resonance images for individualized head modeling. NeuroImage. 2020;219:117044.
Shan Y, Wang H, Yang Y, Wang J, Zhao W, Huang Y, et al. Evidence of a large current of transcranial alternating current stimulation directly to deep brain regions. Mol Psychiatry. 2023;28:5402–10.
Louviot S, Tyvaert L, Maillard LG, Colnat-Coulbois S, Dmochowski J, Koessler L. Transcranial electrical stimulation generates electric fields in deep human brain structures. Brain Stimulation. 2022;15:1–12.
Weinrich CA, Brittain JS, Nowak M, Salimi-Khorshidi R, Brown P, Stagg CJ. Modulation of long-range connectivity patterns via frequency-specific stimulation of human cortex. Curr Biol. 2017;27:3061–3068.e3.
Clancy KJ, Andrzejewski JA, You Y, Rosenberg JT, Ding M, Li W. Transcranial stimulation of alpha oscillations up-regulates the default mode network. Proc Natl Acad Sci USA. 2022;119:e2110868119.
Scangos KW, Makhoul GS, Sugrue LP, Chang EF, Krystal AD. State-dependent responses to intracranial brain stimulation in a patient with depression. Nat Med. 2021;27:229–31.
Acknowledgements
The authors would like to acknowledge Professor Jian Jiang of Lingang Laboratory for supporting technical to help test the robust of tACS equipment, Hui Zheng for his great help in visualization and all volunteers for participating in this study.
Funding
This work was supported by the National Natural Science Foundation of China (T2394535, T2394533, 82325019, 32241015, 82401432), the Shanghai Yangfan Program (21YF1439700), the Science and Technology Commission of Shanghai Municipality (23XD1423000, 23ZR1480800, 24ZR1461000), Shanghai Municipal Commission of Health (2022JC016), Shanghai Municipal Education Commission - Gaofeng Clinical Medicine Grant Support (20181715), and Excellent Young Scientists Fund in Shenzhen (RCYX20231211090405003).
Author information
Authors and Affiliations
Contributions
Conceptualization: Ti-Fei Yuan, Jiwen Xu, Quanying Liu. Funding acquisition: Ti-Fei Yuan, Quanying Liu, Pengfei Wei, Huichun Luo. Data collection: Huichun Luo, Xiaolai Ye, Hui-Ting Cai, Qiangqiang Liu, Ying Xu, Ziyu Mao, Yanqing Cai, Jing Hong. Methodology: Huichun Luo, Xiaolai Ye, Hui-Ting Cai, Mo Wang. Writing - original draft: Huichun Luo, Xiaolai Ye, Hui-Ting Cai, Mo Wang, Yue Wang, Chencheng Zhang. Writing - review & editing: Huichun Luo, Hui-Ting Cai, Pengfei Wei, Yong Lu, Quanying Liu, Jiwen Xu, Ti-Fei Yuan.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luo, H., Ye, X., Cai, HT. et al. Frequency-specific and state-dependent neural responses to brain stimulation. Mol Psychiatry 30, 2880–2890 (2025). https://doi.org/10.1038/s41380-025-02892-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41380-025-02892-7
This article is cited by
-
Randomized, double-blind, sham-controlled pilot trial of theta-band transcranial alternating current stimulation during cognitive training in mild Alzheimer’s disease
Translational Psychiatry (2026)
-
Neuromodulation and cognition in late-life depression
Neuropsychopharmacology (2026)
-
COMMENT Targeting hippocampal networks: frequency- and state-dependent effects of transcranial alternating current stimulation
Molecular Psychiatry (2025)
-
Repetitive transcranial magnetic stimulation targeting the dorsolateral prefrontal cortex promotes recovery of consciousness in patients with disorders of consciousness: a meta-analysis of randomized controlled trials
Journal of Neurology (2025)
-
New Perspectives on Non-Invasive Cerebellar Stimulation for Social and Affective Functions in Children and Adolescents
The Cerebellum (2025)