Abstract
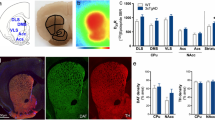
Both post-mortem and in vivo data argue for dopamine dysfunction in patients with Alzheimer’s Disease (AD). However, the timing and regional progression of dopaminergic systems alterations in AD are still debated. The aim of the study was to investigate in vivo the pattern of dopaminergic changes and connectivity using DAT-SPECT imaging in patients across the AD spectrum. Fifty-nine AD patients (n = 21 AD-MCI; n = 38 AD-DEM) and a control group (CG) of n = 45 age- and sex-matched individuals entered the study and underwent 123I-FP-CIT dopaminergic imaging. The occipital binding was used as reference region to obtain single-subject binding in different brain regions. Between-group differences in 123I-FP-CIT binding in both mesolimbic and nigrostriatal dopaminergic pathways were assessed using an ANCOVA test, adjusting for the effect of center of imaging acquisition, age, and sex. Regions resulting from the voxel-wise direct comparison between AD-MCI and AD-DEM were considered as a seed of interest for a voxel-wise interregional correlation analysis. Both AD-MCI and AD-DEM patients showed dopaminergic depletion within the basal ganglia, whereas cortico-limbic regions (namely hippocampus, amygdala, anterior and middle cingulate, frontal cortex and thalamus) resulted impaired only in the dementia phase. The brain voxel-wise interregional correlation analysis showed a progressive pattern of disruption of caudate/thalamus dopaminergic connectivity to hippocampus and amygdala from AD-MCI to AD-DEM stages. This study indicates basal ganglia dopaminergic alterations and connectivity disruption in the nigrostriatal and mesolimbic systems already in early stage AD, extending to several cortico-limbic regions in dementia phases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets used and the scrips used to analyze data during the current study are available from the corresponding author on reasonable request.
References
Palop JJ, Mucke L. Amyloid-β–induced neuronal dysfunction in Alzheimer’s disease: from synapses toward neural networks. Nat Neurosci. 2010;13:812–8.
Rudelli RD, Ambler MW, Wisniewski HM. Morphology and distribution of Alzheimer neuritic (senile) and amyloid plaques in striatum and diencephalon. Acta Neuropathol. 1984;64:273–81.
Attems J, Thal DR, Jellinger KA. The relationship between subcortical tau pathology and Alzheimer’s disease. Biochem Soc Trans. 2012;40:711–5.
Trillo L, Das D, Hsieh W, Medina B, Moghadam S, Lin B, et al. Ascending monoaminergic systems alterations in Alzheimer’s disease. Translating basic science into clinical care. Neurosci Biobehav Rev. 2013;37:1363–79.
Lopez OL, Wisnieski SR, Becker JT, Boiler F, DeKosky ST. Extrapyramidal signs in patients with probable Alzheimer disease. Arch Neurol. 1997;54:969–75.
Rinne JO, Säkö E, Paljärvi L, Mölsä PK, Rinne UK. Brain dopamine D-2 receptors in senile dementia. J Neural Transm. 1986;65:51–62.
Joyce JN, Smutzer G, Whitty CJ, Myers A, Bannon MJ. Differential modification of dopamine transporter and tyrosine hydroxylase mRNAs in midbrain of subjects with Parkinson’s, Alzheimer’s with parkinsonism, and Alzheimer’s disease. Mov Disord. 1997;12:885–97.
Gibb WR, Mountjoy CQ, Mann DM, Lees AJ. The substantia nigra and ventral tegmental area in Alzheimer’s disease and Down’s syndrome. J Neurol Neurosurg Psychiatry. 1989;52:193–200.
Kemppainen N, Laine M, Laakso MP, Kaasinen V, Någren K, Vahlberg T, et al. Hippocampal dopamine D2 receptors correlate with memory functions in Alzheimer’s disease. Eur J Neurosci. 2003;18:149–54.
Cortés R, Probst A, Palacios J. Decreased densities of dopamine D1 receptors in the putamen and hippocampus in senile dementia of the Alzheimer type. Brain Res. 1988;475:164–7.
Pievani M, Bocchetta M, Boccardi M, Cavedo E, Bonetti M, Thompson PM, et al. Striatal morphology in early-onset and late-onset Alzheimer’s disease: a preliminary study. Neurobiol Aging. 2013;34:1728–39.
Sala A, Caminiti SP, Presotto L, Pilotto A, Liguori C, Chiaravalloti A, et al. In vivo human molecular neuroimaging of dopaminergic vulnerability along the Alzheimer’s disease phases. Alzheimers Res Ther. 2021;13:1–12.
Pizzolato G, Chierichetti F, Fabbri M, Cagnin A, Dam M, Ferlin G, et al. Reduced striatal dopamine receptors in Alzheimer’s disease: Single photon emission tomography study with the D sub 2 tracer [sup 123 I]-IBZM. Neurology. 1996;47:1065–8.
Koch G, Di Lorenzo F, Bonnì S, Giacobbe V, Bozzali M, Caltagirone C, et al. Dopaminergic modulation of cortical plasticity in Alzheimer’s disease patients. Neuropsychopharmacology. 2014;39:2654–61.
Koch G, Motta C, Bonnì S, Pellicciari MC, Picazio S, Casula EP, et al. Effect of Rotigotine vs Placebo on Cognitive Functions Among Patients With Mild to Moderate Alzheimer Disease: A Randomized Clinical Trial. JAMA Netw Open. 2020;3:e2010372–e2010372.
Jack CR, Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB, et al. NIA-AA research framework: toward a biological definition of Alzheimer’s disease. Alzheimer’s Dement. 2018;14:535–62.
Boccardi M, Altomare D, Ferrari C, Festari C, Guerra UP, Paghera B, et al. Assessment of the incremental diagnostic value of florbetapir F 18 imaging in patients with cognitive impairment: the incremental diagnostic value of amyloid PET with [18F]-florbetapir (INDIA-FBP) study. JAMA Neurol. 2016;73:1417–24.
Pilotto A, Schiano di Cola F, Premi E, Grasso R, Turrone R, Gipponi S, et al. Extrastriatal dopaminergic and serotonergic pathways in Parkinson’s disease and in dementia with Lewy bodies: a 123 I-FP-CIT SPECT study. Eur J Nucl Med Mol Imaging. 2019;46:1642–51.
Caminiti SP, Pilotto A, Premi E, Galli A, Ferrari E, Gipponi S, et al. Dopaminergic connectivity reconfiguration in the dementia with Lewy bodies continuum. Parkinsonism Relat Disord. 2023;108:105288.
Morbelli S, Esposito G, Arbizu J, Barthel H, Boellaard R, Bohnen NI, et al. EANM practice guideline/SNMMI procedure standard for dopaminergic imaging in Parkinsonian syndromes 1.0. Eur J Nucl Med Mol Imaging. 2020;47:1885–912.
Darcourt J, Booij J, Tatsch K, Varrone A, Vander Borght T, Kapucu ÖL, et al. EANM procedure guidelines for brain neurotransmission SPECT using 123 I-labelled dopamine transporter ligands, version 2. Eur J Nucl Med Mol Imaging. 2010;37:443–50.
Iaccarino L, Sala A, Caminiti SP, Presotto L, Perani D, Initiative ADN. In vivo MRI structural and PET metabolic connectivity study of dopamine pathways in Alzheimer’s disease. J Alzheimer’s Dis. 2020;75:1003–16.
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. 2002;15:273–89.
Shigekiyo T, Arawaka S. Laterality of specific binding ratios on DAT-SPECT for differential diagnosis of degenerative parkinsonian syndromes. Sci Rep. 2020;10:15761.
McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dementia. 2011;7:263–9.
Nobili A, Latagliata EC, Viscomi MT, Cavallucci V, Cutuli D, Giacovazzo G, et al. Dopamine neuronal loss contributes to memory and reward dysfunction in a model of Alzheimer’s disease. Nat Commun. 2017;8:14727.
Moreno-Castilla P, Rodriguez-Duran LF, Guzman-Ramos K, Barcenas-Femat A, Escobar ML, Bermudez-Rattoni F. Dopaminergic neurotransmission dysfunction induced by amyloid-β transforms cortical long-term potentiation into long-term depression and produces memory impairment. Neurobiol Aging. 2016;41:187–99.
Frings L, Hellwig S, Spehl TS, Bormann T, Buchert R, Vach W, et al. Asymmetries of amyloid-β burden and neuronal dysfunction are positively correlated in Alzheimer’s disease. Brain. 2015;138:3089–99.
Pilotto A, Bongianni M, Tirloni C, Galli A, Padovani A, Zanusso G. CSF alpha‐synuclein aggregates by seed amplification and clinical presentation of AD. Alzheimer’s Dement. 2023;19:3754–9.
Nyberg L, Karalija N, Salami A, Andersson M, Wåhlin A, Kaboovand N, et al. Dopamine D2 receptor availability is linked to hippocampal–caudate functional connectivity and episodic memory. Proc Natl Acad Sci. 2016;113:7918–23.
Buckner RL. Memory and executive function in aging and AD: multiple factors that cause decline and reserve factors that compensate. Neuron. 2004;44:195–208.
Delli Pizzi S, Franciotti R, Taylor J-P, Thomas A, Tartaro A, Onofrj M, et al. Thalamic involvement in fluctuating cognition in dementia with Lewy bodies: magnetic resonance evidences. Cereb Cortex. 2015;25:3682–9.
Kish SJ, Shannak K, Hornykiewicz O. Uneven pattern of dopamine loss in the striatum of patients with idiopathic Parkinson’s disease. N Engl J Med. 1988;318:876–80.
Masters MC, Morris JC, Roe CM. Noncognitive” symptoms of early Alzheimer disease: a longitudinal analysis. Neurology. 2015;84:617–22.
Krashia P, Spoleti E, D’Amelio M. The VTA dopaminergic system as diagnostic and therapeutical target for Alzheimer’s disease. Front Psychiatry. 2022;13:1039725.
Udo N, Hashimoto N, Toyonaga T, Isoyama T, Oyanagi Y, Narita H, et al. Apathy in Alzheimer’s disease correlates with the dopamine transporter level in the caudate nuclei. Dement Geriatr Cogn Dis Extra. 2020;10:86–93.
Mitchell RA, Herrmann N, Lanctôt KL. The role of dopamine in symptoms and treatment of apathy in Alzheimer’s disease. CNS Neurosci Ther. 2011;17:411–27.
Grahn JA, Parkinson JA, Owen AM. The cognitive functions of the caudate nucleus. Prog Neurobiol. 2008;86:141–55.
Clément F, Gauthier S, Belleville S. Executive functions in mild cognitive impairment: emergence and breakdown of neural plasticity. Cortex. 2013;49:1268–79.
Manca R, Valera-Bermejo JM, Venneri A, Initiative ADN. Accelerated atrophy in dopaminergic targets and medial temporo-parietal regions precedes the onset of delusions in patients with Alzheimer’s disease. Eur Arch Psychiatry Clin Neurosci. 2023;273:229–41.
Venneri A, De Marco M. Reduced monoaminergic nuclei MRI signal detectable in pre-symptomatic older adults with future memory decline. Sci Rep. 2020;10:18707.
Serra L, D’Amelio M, Esposito S, Di Domenico C, Koch G, Marra C, et al. Ventral tegmental area disconnection contributes two years early to correctly classify patients converted to Alzheimer’s disease: implications for treatment. J Alzheimer’s Dis. 2021;82:985–1000.
Broussard JI, Yang K, Levine AT, Tsetsenis T, Jenson D, Cao F, et al. Dopamine regulates aversive contextual learning and associated in vivo synaptic plasticity in the hippocampus. Cell Rep. 2016;14:1930–9.
Rossato JI, Bevilaqua LRM, Izquierdo I, Medina JH, Cammarota M. Dopamine controls persistence of long-term memory storage. Science. 2009;325:1017–20.
Cordella A, Krashia P, Nobili A, Pignataro A, La Barbera L, Viscomi MT, et al. Dopamine loss alters the hippocampus-nucleus accumbens synaptic transmission in the Tg2576 mouse model of Alzheimer’s disease. Neurobiol Dis. 2018;116:142–54.
La Barbera L, Vedele F, Nobili A, Krashia P, Spoleti E, Latagliata EC, et al. Nilotinib restores memory function by preventing dopaminergic neuron degeneration in a mouse model of Alzheimer’s disease. Prog Neurobiol. 2021;202:102031.
Gloria Y, Ceyzériat K, Tsartsalis S, Millet P, Tournier BB. Dopaminergic dysfunction in the 3xTg-AD mice model of Alzheimer’s disease. Sci Rep. 2021;11:19412.
Vorobyov V, Bakharev B, Medvinskaya N, Nesterova I, Samokhin A, Deev A, et al. Loss of midbrain dopamine neurons and altered apomorphine EEG effects in the 5xFAD mouse model of Alzheimer’s disease. J Alzheimer’s Dis. 2019;70:241–56.
Jung WH, Jang JH, Park JW, Kim E, Goo E-H, Im O-S, et al. Unravelling the intrinsic functional organization of the human striatum: a parcellation and connectivity study based on resting-state FMRI. PLoS One. 2014;9:e106768.
López OL, DeKosky ST. Clinical symptoms in Alzheimer’s disease. Handb Clin Neurol. Elsevier; 2008. p. 207–216.
Acknowledgements
We thank the participants in this study for their time and willingness to contribute this research. The study was partially supported by the Italian Ministry of Health with the grant RF-2018-12366209 and by #NEXTGENERATIONEU (NGEU) funded by the Ministry of University and Research (MIUR), National Recovery and Resilience Plan (NRRP), project MNESYS (PE0000006) – a multiscale integrated approach to the study of the nervous system in health and disease (DN. 1553 11.10.2022) to Silvia Paola Caminiti, Nicola Biagio Mercuri and Claudio Liguori; by the American Alzheimer’s Association (AARG-21-851219) and by Fondazione Roma (Rome, Italy) to Marcello D’Amelio.
Author information
Authors and Affiliations
Contributions
A.Pi. and A.G. performed the analysis and interpretation of data for the work and drafted the manuscript; A.S., S.P.C., C.L., A.C., V.G., GB.F., B.P., L.S., F.B., and O.S. performed the acquisition of data and revised the manuscript; E.P., M.D’A. and N.M. revised the manuscript; D.P. and A.Pa were responsible for the design and conceptualization of the study, performed data interpretation, and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pilotto, A., Galli, A., Sala, A. et al. Dopaminergic deficits along the spectrum of Alzheimer’s disease. Mol Psychiatry 30, 3069–3076 (2025). https://doi.org/10.1038/s41380-025-02913-5
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41380-025-02913-5
This article is cited by
-
Optogenetic stimulation of midbrain dopaminergic neurons rescues hippocampal synaptic plasticity deficits in a mouse model of Alzheimer’s disease
Translational Psychiatry (2025)
-
Midbrain degeneration triggers astrocyte reactivity and tau pathology in experimental Alzheimer’s Disease
Molecular Neurodegeneration (2025)