Abstract
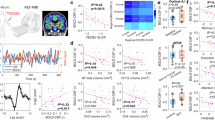
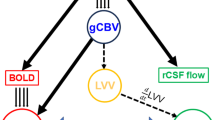
Psychosis involves neuroinflammation and oxidative stress, both affecting the glymphatic system, the lymphatic-like, fluid-transport system in the brain. However, it is unclear whether early psychosis is related to impairments in glymphatic functions. In resting-state fMRI, it has been recently established in a number of neurodegenerative diseases that the coupling relationship between cortical blood-oxygen-level-dependent (BOLD) signal and ventricular cerebrospinal fluid (CSF) flow is associated with brain waste clearance, a key glymphatic function that has not been examined in psychosis or any other psychiatric populations. In a large dataset (total n = 137, age = 23.86 ± 4.16), we demonstrated that glymphatic clearance marked by BOLD-CSF coupling was weaker and more delayed in patients with early psychosis compared to healthy controls. BOLD-CSF coupling also varied between the non-affective and affective psychosis groups with group differences most prominent in high-order but not low-order cortical regions. Finally, reduced global BOLD-CSF coupling was associated with cognitive decline and more severe psychotic symptoms. We provided novel evidence highlighting dysregulated coupling between cortical activity and macroscopic CSF flow as a biomarker for early psychosis. Similar to recent observations in neurodegenerative disorders, the association between reduced BOLD-CSF coupling and psychotic symptoms suggested that waste clearance is disrupted in psychosis which shed light on the pathophysiology of this disease from a glymphatic point of view.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Data used in the preparation of this article were obtained from the Human Connectome Project for Early Psychosis (HCP-EP) database (https://nda.nih.gov/general-query.html?q=query=featured-datasets:Connectomes%20Related%20to%20Human%20Disease).
Code availability
All analyses used open-source software with URL links already included in Methods. Code used in the analyses described in this paper will be made available upon acceptance of the manuscript.
References
Arciniegas DB. Psychosis. Continuum. 2015;21:715.
Lewandowski KE, Bouix S, Ongur D, Shenton ME. Neuroprogression across the early course of psychosis. J Psychiatry Brain Sci. 2020;5:e200002.
Raij TT, Valkonen-Korhonen M, Holi M, Therman S, Lehtonen J, Hari R. Reality of auditory verbal hallucinations. Brain. 2009;132:2994–3001.
Amador X, David A. Insight and psychosis. Nord J Psychiatry. 1999;53:467–8.
Jackson HJ, McGorry PD. The recognition and management of early psychosis: a preventive approach. Cambrige, UK: Cambridge University Press; 2009.
Kane JM, Robinson DG, Schooler NR, Mueser KT, Penn DL, Rosenheck RA, et al. Comprehensive versus usual community care for first-episode psychosis: 2-year outcomes from the NIMH RAISE early treatment program. Am J Psychiatry. 2016;173:362–72.
Buckner RL. The brain’s default network: origins and implications for the study of psychosis. Dialogues Clin Neurosci. 2013;15:351–8.
Gerretsen P, Menon M, Mamo DC, Fervaha G, Remington G, Pollock BG, et al. Impaired insight into illness and cognitive insight in schizophrenia spectrum disorders: resting state functional connectivity. Schizophr Res. 2014;160:43–50.
Holmes A, Levi PT, Chen Y-C, Chopra S, Aquino KM, Pang JC, et al. Disruptions of hierarchical cortical organization in early psychosis and schizophrenia. Biol Psychiatry Cogn Neurosci Neuroimaging. 2023;8:1240–50.
Wang AK, Miller BJ. Meta-analysis of cerebrospinal fluid cytokine and tryptophan catabolite alterations in psychiatric patients: comparisons between schizophrenia, bipolar disorder, and depression. Schizophr Bull. 2018;44:75–83.
Coughlin J, Wang Y, Ambinder E, Ward R, Minn I, Vranesic M, et al. In vivo markers of inflammatory response in recent-onset schizophrenia: a combined study using [11C] DPA-713 PET and analysis of CSF and plasma. Transl Psychiatry. 2016;6:e777–e777.
Schwieler L, Larsson MK, Skogh E, Kegel ME, Orhan F, Abdelmoaty S, et al. Increased levels of IL-6 in the cerebrospinal fluid of patients with chronic schizophrenia—significance for activation of the kynurenine pathway. J Psychiatry Neurosci. 2015;40:126–33.
Guo J, Lesh T, Niendam T, Ragland J, Tully L, Carter C. Brain free water alterations in first-episode psychosis: a longitudinal analysis of diagnosis, course of illness, and medication effects. Psychol Med. 2021;51:1001–10.
Iliff JJ, Goldman SA, Nedergaard M. Implications of the discovery of brain lymphatic pathways. Lancet Neurol. 2015;14:977–9.
Iliff JJ, Wang M, Zeppenfeld DM, Venkataraman A, Plog BA, Liao Y, et al. Cerebral arterial pulsation drives paravascular CSF–interstitial fluid exchange in the murine brain. J Neurosci. 2013;33:18190–9.
Jessen NA, Munk ASF, Lundgaard I, Nedergaard M. The glymphatic system: a beginner’s guide. Neurochem Res. 2015;40:2583–99.
Benveniste H, Lee H, Volkow ND. The glymphatic pathway: waste removal from the CNS via cerebrospinal fluid transport. Neuroscientist. 2017;23:454–65.
Ahn JH, Cho H, Kim J-H, Kim SH, Ham J-S, Park I, et al. Meningeal lymphatic vessels at the skull base drain cerebrospinal fluid. Nature. 2019;572:62–6.
Cai Y, Yangqiqi Z, Shuo L, Ma Y, Jiang Q, Wen Q, et al. The relationship between inflammation, impaired glymphatic system, and neurodegenerative disorders: a vicious cycle. Neurobiol Dis. 2024;192:106426.
Jha MK, Kim J-H, Song GJ, Lee W-H, Lee I-K, Lee H-W, et al. Functional dissection of astrocyte-secreted proteins: implications in brain health and diseases. Prog Neurobiol. 2018;162:37–69.
Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature. 2017;541:481–7.
Bechter K, Reiber H, Herzog S, Fuchs D, Tumani H, Maxeiner HG. P03-76 - Mild encephalitis inflammation subgroups in affective and schizophrenic disorders. Eur Psychiatry. 2010;25:1198.
Li L, Zhang H, Varrin-Doyer M, Zamvil SS, Verkman A. Proinflammatory role of aquaporin-4 in autoimmune neuroinflammation. FASEB J. 2011;25:1556.
Muratake T, Fukui N, Kaneko N, Amagane H, Someya T. Linkage disequilibrium in aquaporin 4 gene and association study with schizophrenia. Psychiatry Clin Neurosci. 2005;59:595–8.
Xiao M, Hu G. Involvement of aquaporin 4 in astrocyte function and neuropsychiatric disorders. CNS Neurosci Ther. 2014;20:385–90.
Genel O, Pariante CM, Borsini A. The role of AQP4 in the pathogenesis of depression, and possible related mechanisms. Brain Behav Immun. 2021;98:366–77.
Han F, Liu X, Mailman RB, Huang X, Liu X. Resting-state global brain activity affects early β-amyloid accumulation in default mode network. Nat Commun. 2023;14:7788.
Jiang D, Liu L, Kong Y, Chen Z, Rosa‐Neto P, Chen K, et al. Regional glymphatic abnormality in behavioral variant frontotemporal dementia. Ann Neurol. 2023;94:442–56.
Fultz NE, Bonmassar G, Setsompop K, Stickgold RA, Rosen BR, Polimeni JR, et al. Coupled electrophysiological, hemodynamic, and cerebrospinal fluid oscillations in human sleep. Science. 2019;366:628–31.
Xie L, Kang H, Xu Q, Chen MJ, Liao Y, Thiyagarajan M, et al. Sleep drives metabolite clearance from the adult brain. Science. 2013;342:373–7.
Han F, Chen J, Belkin-Rosen A, Gu Y, Luo L, Buxton OM, et al. Reduced coupling between cerebrospinal fluid flow and global brain activity is linked to Alzheimer disease–related pathology. PLoS Biol. 2021;19:e3001233.
Wang Z, Song Z, Zhou C, Fang Y, Gu L, Yang W, et al. Reduced coupling of global brain function and cerebrospinal fluid dynamics in Parkinson’s disease. J Cereb Blood Flow Metab. 2023;43:1328–39.
Han F, Brown GL, Zhu Y, Belkin‐Rosen AE, Lewis MM, Du G, et al. Decoupling of global brain activity and cerebrospinal fluid flow in Parkinson’s disease cognitive decline. Mov Disord. 2021;36:2066–76.
Murdock MH, Yang C-Y, Sun N, Pao P-C, Blanco-Duque C, Kahn MC, et al. Multisensory gamma stimulation promotes glymphatic clearance of amyloid. Nature. 2024;627:149–56.
Rasmussen MK, Mestre H, Nedergaard M. The glymphatic pathway in neurological disorders. Lancet Neurol. 2018;17:1016–24.
Yang GJ, Murray JD, Repovs G, Cole MW, Savic A, Glasser MF, et al. Altered global brain signal in schizophrenia. Proc Natl Acad Sci USA. 2014;111:7438–43.
Saarinen A, Lieslehto J, Kiviniemi V, Häkli J, Tuovinen T, Hintsanen M, et al. Symptomatic psychosis risk and physiological fluctuation in functional MRI data. Schizophr Res. 2020;216:339–46.
Demro C, Mueller BA, Kent JS, Burton PC, Olman CA, Schallmo M-P, et al. The psychosis human connectome project: an overview. Neuroimage. 2021;241:118439.
Yeo BT, Krienen FM, Sepulcre J, Sabuncu MR, Lashkari D, Hollinshead M, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. 2011;106:1125–65.
Zuo X-N, Xing X-X. Test-retest reliabilities of resting-state FMRI measurements in human brain functional connectomics: a systems neuroscience perspective. Neurosci Biobehav Rev. 2014;45:100–18.
Zuo X-N, Xu T, Milham MP. Harnessing reliability for neuroscience research. Nat Hum Behav. 2019;3:768–71.
Liu X Decoupling between brain activity and cerebrospinal fluid movement in neurological disorders. J Magn Reson Imaging. 2024;60:1743–52.
Zhang Y, Peng B, Chen S, Liang Q, Zhang Y, Lin S, et al. Reduced coupling between global signal and cerebrospinal fluid inflow in patients with depressive disorder: A resting state functional MRI study. J Affect Disord. 2024;354:136–42.
Davies G, Haddock G, Yung AR, Mulligan LD, Kyle SD. A systematic review of the nature and correlates of sleep disturbance in early psychosis. Sleep Med Rev. 2017;31:25–38.
Kogan S, Ospina LH, Mittal VA, Kimhy D. The impact of inflammation on neurocognition and risk for psychosis: a critical review. Eur Arch Psychiatry Clin Neurosci. 2020;270:793–802.
Hablitz LM, Vinitsky HS, Sun Q, Stæger FF, Sigurdsson B, Mortensen KN, et al. Increased glymphatic influx is correlated with high EEG delta power and low heart rate in mice under anesthesia. Sci Adv. 2019;5:eaav5447.
Chong PL, Garic D, Shen MD, Lundgaard I, Schwichtenberg AJ. Sleep, cerebrospinal fluid, and the glymphatic system: a systematic review. Sleep Med Rev. 2022;61:101572.
Dong H-M, Zhang X-H, Labache L, Zhang S, Ooi LQR, Yeo BT, et al. Ventral attention network connectivity is linked to cortical maturation and cognitive ability in childhood. Nat Neurosci. 2024;27:2009–20.
Smieskova R, Roiser JP, Chaddock CA, Schmidt A, Harrisberger F, Bendfeldt K, et al. Modulation of motivational salience processing during the early stages of psychosis. Schizophr Res. 2015;166:17–23.
Howes OD, Hird EJ, Adams RA, Corlett PR, McGuire P. Aberrant salience, information processing, and dopaminergic signaling in people at clinical high risk for psychosis. Biol Psychiatry. 2020;88:304–14.
Barch DM, Ceaser A. Cognition in schizophrenia: core psychological and neural mechanisms. Trends Cogn Sci. 2012;16:27–34.
Zhou Z-X, Zuo X-N. Lifespan connectome gradients for a road to mental health. J Am Acad Child Adolesc Psychiatry. 2024;63:25–8.
Dong H-M, Margulies DS, Zuo X-N, Holmes AJ. Shifting gradients of macroscale cortical organization mark the transition from childhood to adolescence. Proc Natl Acad Sci USA. 2021;118:e2024448118.
Zhou Z-X, Chen L-Z, Milham MP, Zuo X-N. Six cornerstones for translational brain charts. Sci Bull. 2023;68:795–9.
Wang D, Peng X, Pelletier-Baldelli A, Orlov N, Farabaugh A, Nasr S, et al. Altered temporal, but intact spatial, features of transient network dynamics in psychosis. Mol Psychiatry. 2021;26:2493–503.
Morgan SE, Young J, Patel AX, Whitaker KJ, Scarpazza C, Van Amelsvoort T, et al. Functional magnetic resonance imaging connectivity accurately distinguishes cases with psychotic disorders from healthy controls, based on cortical features associated with brain network development. Biol Psychiatry Cogn Neurosci Neuroimaging. 2021;6:1125–34.
Meda SA, Ruaño G, Windemuth A, O’Neil K, Berwise C, Dunn SM, et al. Multivariate analysis reveals genetic associations of the resting default mode network in psychotic bipolar disorder and schizophrenia. Proc Natl Acad Sci USA. 2014;111:E2066–75.
Clark SV, Mittal VA, Bernard JA, Ahmadi A, King TZ, Turner JA. Stronger default mode network connectivity is associated with poorer clinical insight in youth at ultra high-risk for psychotic disorders. Schizophr Res. 2018;193:244–50.
Sukumar N, Sabesan P, Anazodo U, Palaniyappan L. Neurovascular uncoupling in schizophrenia: a bimodal meta-analysis of brain perfusion and glucose metabolism. Front Psychiatry. 2020;11:754.
Shergill SS, White TP, Joyce DW, Bays PM, Wolpert DM, Frith CD. Functional magnetic resonance imaging of impaired sensory prediction in schizophrenia. JAMA Psychiatry. 2014;71:28–35.
Hanlon FM, Shaff NA, Dodd AB, Ling JM, Bustillo JR, Abbott CC, et al. Hemodynamic response function abnormalities in schizophrenia during a multisensory detection task. Hum Brain Mapp. 2016;37:745–55.
Kress BT, Iliff JJ, Xia M, Wang M, Wei HS, Zeppenfeld D, et al. Impairment of paravascular clearance pathways in the aging brain. Ann Neurol. 2014;76:845–61.
Hsu JL, Wei YC, Toh CH, Hsiao IT, Lin KJ, Yen TC, et al. Magnetic resonance images implicate that glymphatic alterations mediate cognitive dysfunction in Alzheimer disease. Ann Neurol. 2023;93:164–74.
Reeve S, Sheaves B, Freeman D. Sleep disorders in early psychosis: incidence, severity, and association with clinical symptoms. Schizophr Bull. 2019;45:287–95.
Wu Y-F, Sytwu H-K, Lung F-W. Polymorphisms in the human Aquaporin 4 gene are associated with schizophrenia in the southern Chinese Han population: a case–control study. Front Psychiatry. 2020;11:596.
Murphy CE, Walker AK, Weickert CS. Neuroinflammation in schizophrenia: the role of nuclear factor kappa B. Transl Psychiatry. 2021;11:528.
Kindler J, Schultze-Lutter F, Hauf M, Dierks T, Federspiel A, Walther S, et al. Increased striatal and reduced prefrontal cerebral blood flow in clinical high risk for psychosis. Schizophr Bull. 2018;44:182–92.
Gong Z-Q, Zuo X-N. Dark brain energy: toward an integrative model of spontaneous slow oscillations. Phys Life Rev. 2025;52:278–97.
Acknowledgements
This study was supported by the University of Macau (MYRG-GRG2023-00038-FHS, and MYRG-GRG2024-00259-FHS), and the Macao Science and Technology Development Fund (FDCT 0014/2024/RIB1, 0015/2023/ITP1).
Author information
Authors and Affiliations
Contributions
LH: conceptualization, data curation, methodology, software, data analysis, visualization, writing—original draft, writing—review and editing, visualization. XZ: software, data analysis. KZ: data curation, data analysis. ZZ: conceptualization, data curation, methodology, funding acquisition, project administration, writing and editing. ZY: supervision, funding support, writing and editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent informed
All participants provided their written informed consent to participate in the HCP-EP, and the HCP-EP was reviewed and approved by the Human Connectome Project. And, all methods used in this study were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hua, L., Zeng, X., Zhang, K. et al. Reduced glymphatic clearance in early psychosis. Mol Psychiatry 30, 4665–4676 (2025). https://doi.org/10.1038/s41380-025-03058-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41380-025-03058-1
This article is cited by
-
The glymphatic system in neurodegenerative diseases and brain tumors: mechanistic insights, biomarker advances, and therapeutic opportunities
Acta Neuropathologica Communications (2025)