Abstract
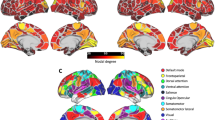
Brain functional alterations associated with overall cognitive impairments and specific cognitive domains defined by MATRICS Consensus Cognitive Battery (MCCB) and the similarities among them remain unclear in schizophrenia. Comprehensive literature review and meta-analyses of cognitive task fMRI studies were conducted to identify whole-brain differences between schizophrenia patients and healthy controls, and subgroup analyses were also conducted for each MCCB domain. Identified brain regions were mapped onto canonical brain networks. The similarity analyses between network dysfunction obtained for paired subgroup analyses with and without each MCCB domain, and between each MCCB domain and primary analysis, were calculated after controlling for sample size. Meta-regression analyses were conducted between brain alterations and demographic and clinical characteristics. The present meta-analysis encompassed 232 datasets with 5229 schizophrenia patients and 6132 healthy controls. In primary analysis, schizophrenia patients showed significant brain dysfunctions mainly within default mode and subcortical networks. Distinct brain dysfunctions for each MCCB domain were also identified. Sample size-weighted similarity analyses revealed that network alterations associated with working memory deficits, among the seven, showed the greatest convergence with brain changes identified in subgroup analysis without this domain and primary analysis (sample size-weighted Dice coefficients = 0.24 and 0.51). Significant correlations were identified between significant brain alterations and negative symptoms and years of education. Aberrant activations in default mode and subcortical networks in working memory domain showed higher similarity than those observed in the other six cognitive domains, indicating functional integration of working memory and functional specialization of remaining cognitive domains in schizophrenia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author upon reasonable request.
References
McCutcheon RA, Keefe RSE, McGuire PK. Cognitive impairment in schizophrenia: aetiology, pathophysiology, and treatment. Mol Psychiatry. 2023;28:1902–18.
August SM, Kiwanuka JN, McMahon RP, Gold JM. The MATRICS Consensus Cognitive Battery (MCCB): clinical and cognitive correlates. Schizophr Res. 2012;134:76–82.
Howes OD, Bukala BR, Beck K. Schizophrenia: from neurochemistry to circuits, symptoms and treatments. Nat Rev Neurol. 2024;20:22–35.
Catalan A, Salazar de Pablo G, Aymerich C, Damiani S, Sordi V, Radua J, et al. Neurocognitive functioning in individuals at clinical high risk for psychosis: a systematic review and meta-analysis. JAMA Psychiatry. 2021;78:859–67.
Green MF, Horan WP, Lee J. Nonsocial and social cognition in schizophrenia: current evidence and future directions. World Psychiatry. 2019;18:146–61.
Gold JM, Goldberg RW, McNary SW, Dixon LB, Lehman AF. Cognitive correlates of job tenure among patients with severe mental illness. Am J Psychiatry. 2002;159:1395–402.
Jauhar S, Johnstone M, McKenna PJ. Schizophrenia. Lancet. 2022;399:473–86.
Zhang W, Sweeney JA, Bishop JR, Gong Q, Lui S. Biological subtyping of psychiatric syndromes as a pathway for advances in drug discovery and personalized medicine. Nat Ment Health. 2023;1:88–99.
Su W, Wang J, Tang Y. A narrative literature review of white matter microstructure in individuals at clinical high risk for psychosis. Psychoradiology. 2025;5:kkae031.
Glahn DC, Ragland JD, Abramoff A, Barrett J, Laird AR, Bearden CE, et al. Beyond hypofrontality: a quantitative meta-analysis of functional neuroimaging studies of working memory in schizophrenia. Hum Brain Mapp. 2005;25:60–9.
Achim AM, Lepage M. Episodic memory-related activation in schizophrenia: meta-analysis. Br J Psychiatry. 2005;187:500–9.
Minzenberg MJ, Laird AR, Thelen S, Carter CS, Glahn DC. Meta-analysis of 41 functional neuroimaging studies of executive function in schizophrenia. Arch Gen Psychiatry. 2009;66:811–22.
Zhang W, Lei D, Keedy SK, Ivleva EI, Eum S, Yao L, et al. Brain gray matter network organization in psychotic disorders. Neuropsychopharmacology. 2020;45:666–74.
McTeague LM, Huemer J, Carreon DM, Jiang Y, Eickhoff SB, Etkin A. Identification of common neural circuit disruptions in cognitive control across psychiatric disorders. Am J Psychiatry. 2017;174:676–85.
Silver H, Feldman P, Bilker W, Gur RC. Working memory deficit as a core neuropsychological dysfunction in schizophrenia. Am J Psychiatry. 2003;160:1809–16.
Bijsterbosch J, Harrison SJ, Jbabdi S, Woolrich M, Beckmann C, Smith S, et al. Challenges and future directions for representations of functional brain organization. Nat Neurosci. 2020;23:1484–95.
Nuechterlein KH, Green MF, Kern RS, Baade LE, Barch DM, Cohen JD, et al. The MATRICS consensus cognitive battery, part 1: test selection, reliability, and validity. Am J Psychiatry. 2008;165:203–13.
Kern RS, Nuechterlein KH, Green MF, Baade LE, Fenton WS, Gold JM, et al. The MATRICS consensus cognitive battery, part 2: co-norming and standardization. Am J Psychiatry. 2008;165:214–20.
Voineskos AN, Hawco C, Neufeld NH, Turner JA, Ameis SH, Anticevic A, et al. Functional magnetic resonance imaging in schizophrenia: current evidence, methodological advances, limitations and future directions. World Psychiatry. 2024;23:26–51.
Smallwood J, Bernhardt BC, Leech R, Bzdok D, Jefferies E, Margulies DS. The default mode network in cognition: a topographical perspective. Nat Rev Neurosci. 2021;22:503–13.
Farahani FV, Karwowski W, Lighthall NR. Application of graph theory for identifying connectivity patterns in human brain networks: a systematic review. Front Neurosci. 2019;13:585.
Picó-Pérez M, Vieira R, Fernández-Rodríguez M, De Barros MAP, Radua J, Morgado P. Multimodal meta-analysis of structural gray matter, neurocognitive and social cognitive fMRI findings in schizophrenia patients. Psychol Med. 2022;52:614–24.
Khalil M, Hollander P, Raucher-Chéné D, Lepage M, Lavigne KM. Structural brain correlates of cognitive function in schizophrenia: a meta-analysis. Neurosci Biobehav Rev. 2022;132:37–49.
Sterne JA, Gavaghan D, Egger M. Publication and related bias in meta-analysis: power of statistical tests and prevalence in the literature. J Clin Epidemiol. 2000;53:1119–29.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA Cochrane Handbook for Systematic Reviews of Interventions version 6.5 (updated August 2024). Cochrane. 2024. www.cochrane.org/handbook.
Tahmasian M, Sepehry AA, Samea F, Khodadadifar T, Soltaninejad Z, Javaheripour N, et al. Practical recommendations to conduct a neuroimaging meta-analysis for neuropsychiatric disorders. Hum Brain Mapp. 2019;40:5142–54.
Radua J, Mataix-Cols D, Phillips ML, El-Hage W, Kronhaus DM, Cardoner N, et al. A new meta-analytic method for neuroimaging studies that combines reported peak coordinates and statistical parametric maps. Eur Psychiatry. 2012;27:605–11.
Radua J, van den Heuvel OA, Surguladze S, Mataix-Cols D. Meta-analytical comparison of voxel-based morphometry studies in obsessive-compulsive disorder vs other anxiety disorders. Arch Gen Psychiatry. 2010;67:701–11.
Yeo BT, Krienen FM, Sepulcre J, Sabuncu MR, Lashkari D, Hollinshead M, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. 2011;106:1125–65.
Zou KH, Warfield SK, Bharatha A, Tempany CM, Kaus MR, Haker SJ, et al. Statistical validation of image segmentation quality based on a spatial overlap index. Acad Radiol. 2004;11:178–89.
Alexander-Bloch AF, Shou H, Liu S, Satterthwaite TD, Glahn DC, Shinohara RT, et al. On testing for spatial correspondence between maps of human brain structure and function. Neuroimage. 2018;178:540–51.
Zhao Y, Zhang Q, Shah C, Li Q, Sweeney JA, Li F, et al. Cortical thickness abnormalities at different stages of the illness course in schizophrenia: a systematic review and meta-analysis. JAMA Psychiatry. 2022;79:560–70.
Johnson MK, McMahon RP, Robinson BM, Harvey AN, Hahn B, Leonard CJ, et al. The relationship between working memory capacity and broad measures of cognitive ability in healthy adults and people with schizophrenia. Neuropsychology. 2013;27:220–9.
Yamashita M, Yoshihara Y, Hashimoto R, Yahata N, Ichikawa N, Sakai Y, et al. A prediction model of working memory across health and psychiatric disease using whole-brain functional connectivity. Elife. 2018;7:e38844.
Gold JM, Luck SJ. Working memory in people with schizophrenia. Curr Top Behav Neurosci. 2023;63:137–52.
Gold JM, Barch DM, Feuerstahler LM, Carter CS, MacDonald AW, Ragland JD, et al. Working memory impairment across psychotic disorders. Schizophr Bull. 2019;45:804–12.
Damgaard V, Schandorff JM, Macoveanu J, Sankar A, Zarp J, Fisher PM, et al. Network-wide aberrancies in neuronal activity during working memory in a large cohort of patients with mood disorders: associations with cognitive impairment and functional disability. Mol Psychiatry. 2025;30:4836–44.
Anticevic A, Cole MW, Murray JD, Corlett PR, Wang XJ, Krystal JH. The role of default network deactivation in cognition and disease. Trends Cogn Sci. 2012;16:584–92.
Floresco SB, Braaksma DN, Phillips AG. Thalamic-cortical-striatal circuitry subserves working memory during delayed responding on a radial arm maze. J Neurosci. 1999;19:11061–71.
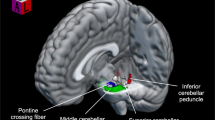
Cao H, Argyelan M, Yan J, Velioglu HA, Fang F, Joanlanne A, et al. Mapping cerebellar connectivity to cognition in psychosis: convergent evidence from functional magnetic resonance imaging and transcranial magnetic stimulation. Biol Psychiatry. 2025;99:134–41.
Whitfield-Gabrieli S, Thermenos HW, Milanovic S, Tsuang MT, Faraone SV, McCarley RW, et al. Hyperactivity and hyperconnectivity of the default network in schizophrenia and in first-degree relatives of persons with schizophrenia. Proc Natl Acad Sci USA. 2009;106:1279–84.
Pavuluri MN, Passarotti AM, Fitzgerald JM, Wegbreit E, Sweeney JA. Risperidone and divalproex differentially engage the fronto-striato-temporal circuitry in pediatric mania: a pharmacological functional magnetic resonance imaging study. J Am Acad Child Adolesc Psychiatry. 2012;51:157–70.e5.
Van Snellenberg JX, Girgis RR, Horga G, van de Giessen E, Slifstein M, Ojeil N, et al. Mechanisms of working memory impairment in schizophrenia. Biol Psychiatry. 2016;80:617–26.
Barch DM, Sheffield JM. Cognitive impairments in psychotic disorders: common mechanisms and measurement. World Psychiatry. 2014;13:224–32.
Bolkan SS, Stujenske JM, Parnaudeau S, Spellman TJ, Rauffenbart C, Abbas AI, et al. Thalamic projections sustain prefrontal activity during working memory maintenance. Nat Neurosci. 2017;20:987–96.
Cao H, Chén OY, Chung Y, Forsyth JK, McEwen SC, Gee DG, et al. Cerebello-thalamo-cortical hyperconnectivity as a state-independent functional neural signature for psychosis prediction and characterization. Nat Commun. 2018;9:3836.
Cattaneo Z, Ferrari C, Ciricugno A, Heleven E, Schutter D, Manto M, et al. New horizons on non-invasive brain stimulation of the social and affective cerebellum. Cerebellum. 2022;21:482–96.
Cao H, Cannon TD. Cerebellar dysfunction and schizophrenia: from “Cognitive Dysmetria” to a potential therapeutic target. Am J Psychiatry. 2019;176:498–500.
Knowles EE, David AS, Reichenberg A. Processing speed deficits in schizophrenia: reexamining the evidence. Am J Psychiatry. 2010;167:828–35.
Sheffield JM, Barch DM. Cognition and resting-state functional connectivity in schizophrenia. Neurosci Biobehav Rev. 2016;61:108–20.
Holmlund TB, Chandler C, Foltz PW, Cohen AS, Cheng J, Bernstein JC, et al. Applying speech technologies to assess verbal memory in patients with serious mental illness. NPJ Digit Med. 2020;3:33.
Bremner JD, Vythilingam M, Vermetten E, Vaccarino V, Charney DS. Deficits in hippocampal and anterior cingulate functioning during verbal declarative memory encoding in midlife major depression. Am J Psychiatry. 2004;161:637–45.
Green MF, Horan WP, Lee J. Social cognition in schizophrenia. Nat Rev Neurosci. 2015;16:620–31.
Meisner OC, Nair A, Chang SWC. Amygdala connectivity and implications for social cognition and disorders. Handb Clin Neurol. 2022;187:381–403.
Lesh TA, Niendam TA, Minzenberg MJ, Carter CS. Cognitive control deficits in schizophrenia: mechanisms and meaning. Neuropsychopharmacology. 2011;36:316–38.
Collier AK, Wolf DH, Valdez JN, Gur RE, Gur RC. Subsequent memory effects in schizophrenia. Psychiatry Res. 2014;224:211–7.
Eisenberg DP, Berman KF. Executive function, neural circuitry, and genetic mechanisms in schizophrenia. Neuropsychopharmacology. 2010;35:258–77.
Sui J, Qi S, van Erp TGM, Bustillo J, Jiang R, Lin D, et al. Multimodal neuromarkers in schizophrenia via cognition-guided MRI fusion. Nat Commun. 2018;9:3028.
Menon V. Brain networks and cognitive impairment in psychiatric disorders. World Psychiatry. 2020;19:309–10.
Li X, Zeng J, Liu N, Yang C, Tao B, Sun H, et al. Progressive alterations of resting-state hypothalamic dysconnectivity in schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry. 2024;135:111127.
Galderisi S, Mucci A, Buchanan RW, Arango C. Negative symptoms of schizophrenia: new developments and unanswered research questions. Lancet Psychiatry. 2018;5:664–77.
Crossley NA, Alliende LM, Czepielewski LS, Aceituno D, Castañeda CP, Diaz C, et al. The enduring gap in educational attainment in schizophrenia according to the past 50 years of published research: a systematic review and meta-analysis. Lancet Psychiatry. 2022;9:565–73.
Ma Y, Zou Y, Liu X, Chen T, Kemp GJ, Gong Q, et al. Social intelligence mediates the protective role of resting-state brain activity in the social cognition network against social anxiety. Psychoradiology. 2024;4:kkae009.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (Project Nos. 82471959, 82120108014, and 82441007), Sichuan Science and Technology Program (Grant No. 2024NSFSC1794), Institutional Research Fund from Sichuan University (No. 2023SCUH0064), 1.3.5 project for Disciplines of Excellence (Project No. ZYGD23003), West China Hospital, Sichuan University, “Qimingxing” Research Fund for Young Talents from West China Hospital of Sichuan University (Grant No. HXQMX0100).
Author information
Authors and Affiliations
Contributions
Xing Li, Jiaxin Zeng, Su Lui and Wenjing Zhang designed the study. Xing Li, Jiaxin Zeng and Naici Liu collected and analyzed the data. Xing Li, Biqiu Tang and Hui Sun performed data visualization. Xing Li and Jiaxin Zeng drafted the manuscript. Changjian Qiu, James M. Gold, Su Lui and Wenjing Zhang reviewed and edited the manuscript. Su Lui and Wenjing Zhang provided resources and supervised the project. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
Drs. Wenjing Zhang and James M. Gold consult to VeraSci. The remaining authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethics approval and consent to participate
This study includes a meta-analysis based on secondary data from previously published, publicly available data. No new human or animal participants were recruited or studied directly by the authors. This study was performed in accordance with the relevant guidelines and regulations. All studies included in the meta-analysis had received ethical approval from their respective institutional review boards, and informed consent was obtained from all participants in those original studies. Therefore, no additional ethical approval or informed consent was required for this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, X., Zeng, J., Liu, N. et al. Functional brain abnormalities of cognitive impairments in schizophrenia indicating higher integration of working memory than other domains. Mol Psychiatry (2026). https://doi.org/10.1038/s41380-026-03518-2
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41380-026-03518-2