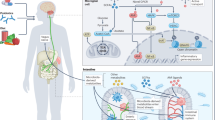
Abstract
Major depressive disorder (MDD) is a major psychiatric condition associated with neuroimmune dysregulation. Microglia, the principal innate immune cells of the central nervous system, have been implicated in MDD. Nevertheless, the specific subpopulations involved and the mechanisms that govern their responses are still elusive. Here, we found that gut microbiota intervention using bedding from control non-stressed mice reverses chronic unpredictable mild stress (CUMS)-induced alterations in behavior. Using CD45+ immune cell sorting and single-cell RNA sequencing (scRNA-seq), we built an atlas of brain-resident microglia and non-microglial immune cells in the central and peripheral compartments. CUMS primarily induced transcriptional alterations in microglia and monocytes, while the reversed changes induced by bedding exchange mainly detected in microglia (85%). Among the microglial subtypes, homeostatic microglia 2 (HM2) primarily responded to CUMS. HM2 shaped microglial states through activation trajectory: the HM2→chemokine-related microglia 3→inflame microglia 1, which could be reversed by bedding exchange. Brain Mdcs served as the mainly immune cells interact with microglia. Furthermore, we systematically characterized alterations in the myeloid-derived cells across the brain, skull, and peripheral blood mononuclear cells (PBMC). Meanwhile, CUMS disrupted immune cell differentiation by suppressing chromatin openness in global accessibility sequencing analyses, and Krüppel-like factors Klf2 emerged as a master regulator expressed in monocytes and HM2 microglial subtype. Remarkably, microbiota remodeling partially reversed this epigenetic dysregulation, restoring immune cell migration and activation processes. In summary, this study revealed that gut microbiota intervention ameliorates depressive phenotypes by dynamically reprogramming microglia via the “periphery-CNS immune” axis, providing a novel entry for antidepressant strategies targeting neuro-immune interactions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Herrman H, Patel V, Kieling C, Berk M, Buchweitz C, Cuijpers P, et al. Time for united action on depression: a lancet-world psychiatric association commission. Lancet. 2022;399:957–1022.
Marwaha S, Palmer E, Suppes T, Cons E, Young AH, Upthegrove R. Novel and emerging treatments for major depression. Lancet. 2023;401:141–53.
Cui L, Li S, Wang S, Wu X, Liu Y, Yu W, et al. Major depressive disorder: hypothesis, mechanism, prevention and treatment. Signal Transduct Target Ther. 2024;9:30.
Shen SY, Liang LF, Shi TL, Shen ZQ, Yin SY, Zhang JR, et al. Microglia-Derived interleukin-6 triggers astrocyte apoptosis in the hippocampus and mediates depression-like behavior. Adv Sci (Weinh). 2025;12:e2412556.
Zeng Y, Chourpiliadis C, Hammar N, Seitz C, Valdimarsdottir UA, Fang F, et al. Inflammatory biomarkers and risk of psychiatric disorders. JAMA Psychiatry. 2024;81:1118–29.
Lee Y, Ishikawa T, Lee H, Lee B, Ryu C, Davila Mejia I, et al. Brain-wide mapping of immune receptors uncovers a neuromodulatory role of IL-17E and the receptor IL-17RB. Cell. 2025;188:2203–17 e17.
Lee B, Kwon JT, Jeong Y, Caris H, Oh D, Feng M, et al. Inflammatory and anti-inflammatory cytokines bidirectionally modulate amygdala circuits regulating anxiety. Cell. 2025;188:2190–202 e15.
Beurel E, Toups M, Nemeroff CB. The bidirectional relationship of depression and inflammation: double trouble. Neuron. 2020;107:234–56.
Maes M, Almulla AF, You Z, Zhang Y. Neuroimmune, metabolic and oxidative stress pathways in major depressive disorder. Nat Rev Neurol. 2025;21:473–89.
Wu A, Zhang J. Neuroinflammation, memory, and depression: new approaches to hippocampal neurogenesis. J Neuroinflammation. 2023;20:283.
Wu J, Li Y, Huang Y, Liu L, Zhang H, Nagy C, et al. Integrating spatial and single-nucleus transcriptomic data elucidates microglial-specific responses in female cynomolgus macaques with depressive-like behaviors. Nat Neurosci. 2023;26:1352–64.
Huang Y, Wu J, Zhang H, Li Y, Wen L, Tan X, et al. The gut microbiome modulates the transformation of microglial subtypes. Mol Psychiatry. 2023;28:1611–21.
Ohara TE, Hsiao EY. Microbiota-neuroepithelial signalling across the gut-brain axis. Nat Rev Microbiol. 2025;23:371–84.
Zhao Y, Ma S, Liang L, Cao S, Fan Z, He D, et al. Gut Microbiota-Metabolite-Brain Axis reconstitution reverses sevoflurane-induced social and synaptic deficits in neonatal mice. Research (Wash D C). 2024;7:0482.
Qian X, Li Q, Zhu H, Chen Y, Lin G, Zhang H, et al. Bifidobacteria with indole-3-lactic acid-producing capacity exhibit psychobiotic potential via reducing neuroinflammation. Cell Rep Med. 2024;5:101798.
White Z, Cabrera I, Mei L, Clevenger M, Ochoa-Raya A, Kapustka I, et al. Gut inflammation promotes microbiota-specific CD4 T cell-mediated neuroinflammation. Nature. 2025;643:509–18.
Yang J, Zheng P, Li Y, Wu J, Tan X, Zhou J, et al. Landscapes of bacterial and metabolic signatures and their interaction in major depressive disorders. Sci Adv. 2020;6:eaba8555.
Zheng P, Yang J, Li Y, Wu J, Liang W, Yin B, et al. Gut microbial signatures can discriminate unipolar from bipolar depression. Adv Sci (Weinh). 2020;7:1902862.
Zhao M, Ren Z, Zhao A, Tang Y, Kuang J, Li M, et al. Gut bacteria-driven homovanillic acid alleviates depression by modulating synaptic integrity. Cell Metab. 2024;36:1000–12 e6.
Liu P, Liu Z, Wang J, Wang J, Gao M, Zhang Y, et al. Immunoregulatory role of the gut microbiota in inflammatory depression. Nat Commun. 2024;15:3003.
Zhou M, Fan Y, Xu L, Yu Z, Wang S, Xu H, et al. Microbiome and tryptophan metabolomics analysis in adolescent depression: roles of the gut microbiota in the regulation of tryptophan-derived neurotransmitters and behaviors in human and mice. Microbiome. 2023;11:145.
Merchak AR, Wachamo S, Brown LC, Thakur A, Moreau B, Brown RM, et al. Lactobacillus from the altered schaedler flora maintain IFNgamma homeostasis to promote behavioral stress resilience. Brain Behav Immun. 2024;115:458–69.
Green JE, Davis JA, Berk M, Hair C, Loughman A, Castle D, et al. Efficacy and safety of fecal microbiota transplantation for the treatment of diseases other than clostridium difficile infection: a systematic review and meta-analysis. Gut Microbes. 2020;12:1–25.
Tan X, Wu J, Zhang H, Li Y, Huang Y, Zheng P, et al. Biogeography of intestinal mucus-associated microbiome: Depletion of genus Pseudomonas is associated with depressive-like behaviors in female cynomolgus macaques. J Adv Res. 2025;70:393–404.
Pereira-Iglesias M, Maldonado-Teixido J, Melero A, Piriz J, Galea E, Ransohoff RM, et al. Microglia as hunters or gatherers of brain synapses. Nat Neurosci. 2025;28:15–23.
Keane L, Clarke G, Cryan JF. A role for microglia in mediating the microbiota-gut-brain axis. Nat Rev Immunol. 2025;25:847–61.
Wu Z, Wang Y, Chen WW, Sun H, Chen X, Li X, et al. Peripheral nervous system microglia-like cells regulate neuronal soma size throughout evolution. Cell. 2025;188:2159–74 e15.
Lau SF, Wu W, Wong HY, Ouyang L, Qiao Y, Xu J, et al. The VCAM1-ApoE pathway directs microglial chemotaxis and alleviates Alzheimer’s disease pathology. Nat Aging. 2023;3:1219–36.
Gao C, Jiang J, Tan Y, Chen S. Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets. Signal Transduct Target Ther. 2023;8:359.
Chen X, Firulyova M, Manis M, Herz J, Smirnov I, Aladyeva E, et al. Microglia-mediated T cell infiltration drives neurodegeneration in tauopathy. Nature. 2023;615:668–77.
Zhang X, Wang R, Chen H, Jin C, Jin Z, Lu J, et al. Aged microglia promote peripheral T cell infiltration by reprogramming the microenvironment of neurogenic niches. Immun Ageing. 2022;19:34.
Guo H, Chou WC, Lai Y, Liang K, Tam JW, Brickey WJ, et al. Multi-omics analyses of radiation survivors identify radioprotective microbes and metabolites. Science. 2020;370:eaay9097.
Pikor NB, Morbe U, Lutge M, Gil-Cruz C, Perez-Shibayama C, Novkovic M, et al. Remodeling of light and dark zone follicular dendritic cells governs germinal center responses. Nat Immunol. 2020;21:649–59.
Jin S, Plikus MV, Nie Q. CellChat for systematic analysis of cell-cell communication from single-cell transcriptomics. Nat Protoc. 2025;20:180–219.
Li R, Zhao H, Huang X, Zhang J, Bai R, Zhuang L, et al. Super-enhancer RNA m(6)A promotes local chromatin accessibility and oncogene transcription in pancreatic ductal adenocarcinoma. Nat Genet. 2023;55:2224–34.
Liu Z, Zhang S, James BT, Galani K, Mangan RJ, Fass SB, et al. Single-cell multiregion epigenomic rewiring in Alzheimer’s disease progression and cognitive resilience. Cell. 2025;188:4980–5002.e29.
Chawla A, Cakmakci D, Fiori LM, Zang W, Maitra M, Yang J, et al. Single-nucleus chromatin accessibility profiling identifies cell types and functional variants contributing to major depression. Nat Genet. 2025;57:1890–904.
Zhu T, Zhou X, You Y, Wang L, He Z, Chen D. cisDynet: an integrated platform for modeling gene-regulatory dynamics and networks. Imeta. 2023;2:e152.
Jiang W, Li Y, Yang J, Tan X, Tian R, Ma G, et al. Chronic stress in mice: how gut bacteria influence gene activity in key brain neurons. Translational psychiatry. 2025;15:262.
Marsh SE, Walker AJ, Kamath T, Dissing-Olesen L, Hammond TR, de Soysa TY, et al. Dissection of artifactual and confounding glial signatures by single-cell sequencing of mouse and human brain. Nature neuroscience. 2022;25:306–16.
Tripathi A, Bartosh A, Mata J, Jacks C, Madeshiya AK, Hussein U, et al. Microglial type I interferon signaling mediates chronic stress-induced synapse loss and social behavior deficits. Mol Psychiatry. 2025;30:423–34.
Zhou J, Ma S, Feng D, Tian Y, Li L, Guo H, et al. C5aR1(+) microglia exacerbate neuroinflammation and cerebral edema in acute brain injury. Neuron. 2025;114:444–462.e9.
Cathomas F, Lin HY, Chan KL, Li L, Parise LF, Alvarez J, et al. Circulating myeloid-derived MMP8 in stress susceptibility and depression. Nature. 2024;626:1108–15.
Klawonn AM, Fritz M, Castany S, Pignatelli M, Canal C, Simila F, et al. Microglial activation elicits a negative affective state through prostaglandin-mediated modulation of striatal neurons. Immunity. 2021;54:225–34 e6.
Ge K, Bai Z, Wang J, Li Z, Gao F, Liu S, et al. Engineering EVs-Mediated mRNA delivery regulates microglia function and alleviates depressive-like behaviors. Adv Mater. 2025;37:e2418872.
Martins-Ferreira R, Calafell-Segura J, Leal B, Rodriguez-Ubreva J, Martinez-Saez E, Mereu E, et al. The Human Microglia Atlas (HuMicA) unravels changes in disease-associated microglia subsets across neurodegenerative conditions. Nat Commun. 2025;16:739.
Shi FD, Yong VW. Neuroinflammation across neurological diseases. Science. 2025;388:eadx0043.
Ardura-Fabregat A, Bosch LFP, Wogram E, Mossad O, Sankowski R, Aktories P, et al. Response of spatially defined microglia states with distinct chromatin accessibility in a mouse model of Alzheimer’s disease. Nat Neurosci. 2025;28:1688–703.
Li X, Li Y, Jin Y, Zhang Y, Wu J, Xu Z, et al. Transcriptional and epigenetic decoding of the microglial aging process. Nat Aging. 2023;3:1288–311.
Sun N, Victor MB, Park YP, Xiong X, Scannail AN, Leary N, et al. Human microglial state dynamics in Alzheimer’s disease progression. Cell. 2023;186:4386–403 e29.
Weinreich MA, Takada K, Skon C, Reiner SL, Jameson SC, Hogquist KA. KLF2 transcription-factor deficiency in T cells results in unrestrained cytokine production and upregulation of bystander chemokine receptors. Immunity. 2009;31:122–30.
Laha D, Deb M, Das H. KLF2 (kruppel-like factor 2 [lung]) regulates osteoclastogenesis by modulating autophagy. Autophagy. 2019;15:2063–75.
Smyth LCD, Kipnis J. Redefining CNS immune privilege. Nat Rev Immunol. 2025;25:766–75.
Herisson F, Frodermann V, Courties G, Rohde D, Sun Y, Vandoorne K, et al. Direct vascular channels connect skull bone marrow and the brain surface enabling myeloid cell migration. Nat Neurosci. 2018;21:1209–17.
Rustenhoven J, Drieu A, Mamuladze T, de Lima KA, Dykstra T, Wall M, et al. Functional characterization of the dural sinuses as a neuroimmune interface. Cell. 2021;184:1000–16 e27.
Smyth LCD, Xu D, Okar SV, Dykstra T, Rustenhoven J, Papadopoulos Z, et al. Identification of direct connections between the dura and the brain. Nature. 2024;627:165–73.
Kigar SL, Lynall ME, DePuyt AE, Atkinson R, Sun VH, Samuels JD, et al. Chronic social defeat stress induces meningeal neutrophilia via type I interferon signaling in male mice. Nat Commun. 2025;16:8153.
Yang R, Wang X, Yang J, Zhou X, Wu Y, Li Y, et al. Perturbations in the microbiota-gut-brain axis shaped by social status loss. Commun Biol. 2025;8:401.
Garcia-Bonilla L, Shahanoor Z, Sciortino R, Nazarzoda O, Racchumi G, Iadecola C, et al. Analysis of brain and blood single-cell transcriptomics in acute and subacute phases after experimental stroke. Nat Immunol. 2024;25:357–70.
Yin W, Gallagher NR, Sawicki CM, McKim DB, Godbout JP, Sheridan JF. Repeated social defeat in female mice induces anxiety-like behavior associated with enhanced myelopoiesis and increased monocyte accumulation in the brain. Brain Behav Immun. 2019;78:131–42.
McKim DB, Weber MD, Niraula A, Sawicki CM, Liu X, Jarrett BL, et al. Microglial recruitment of IL-1beta-producing monocytes to brain endothelium causes stress-induced anxiety. Mol Psychiatry. 2018;23:1421–31.
Wohleb ES, McKim DB, Sheridan JF, Godbout JP. Monocyte trafficking to the brain with stress and inflammation: a novel axis of immune-to-brain communication that influences mood and behavior. Front Neurosci. 2014;8:447.
Bastos J, O’Brien C, Vara-Perez M, Mampay M, van Olst L, Barry-Carroll L, et al. Monocytes can efficiently replace all brain macrophages and fetal liver monocytes can generate bona fide SALL1(+) microglia. Immunity. 2025;58:1269–88 e12.
Zuo Y, Li S, Zhu X, Yang J, Hu X, Wen W, et al. Monocyte-derived cells promote transient insult-induced brain injury by enhancing CD8(+) T cell response. Cell Rep. 2025;44:116528.
Acknowledgements
This work was supported by Chongqing medical scientific research project (Joint project of Chongqing Health Commission and Science and Technology Bureau, 2026DBXM002), the National Key R&D Program of China (STI2030-Major Projects2021ZD0200600), the National Natural Science Foundation Project of China(82571748, 82471545, 82401784, 32400850, 82401523, 82501836, 82501469, 82501837), National Reserve Talent Project in the Health and Wellness Sector of Chongqing (HBRC202410, HBRC202417), China Postdoctoral Science Foundation (2024MD754023, 2025MD774171, 2025T180580), the Key Project of the Natural Science Foundation of Chongqing (Chongqing Science and Technology Development Foundation) under Grant No. 2024NSCQ-KJFZZDX0005, the New Chongqing Youth Innovation Talent Project (Life and Health) under Grant No. 2024NSCQ-qncxX0029.
Author information
Authors and Affiliations
Contributions
PZ conceived, designed, and supervised the project; XW, RY and YW performed the animal experiments; RT, YYW and WJ processed the sequencing samples; YPW, JW and YH performed 16S rRNA analysis; JZ, YL and PL participated in flow cytometry sorting experiments; XT, MY and HZ participated in ATAC data analysis; JW and XZ performed Single-Cell RNA Sequencing analysis and wrote the manuscript; MLW, JL and PZ revised the manuscript and provided guidance for experiments and analysis. All the authors validated and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, J., Zhou, X., Luo, T. et al. Microbiota-Driven microglia reprogramming reverses depressive-like behaviors through Peripheral-to-Central immune crosstalk. Mol Psychiatry (2026). https://doi.org/10.1038/s41380-026-03545-z
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41380-026-03545-z