Abstract
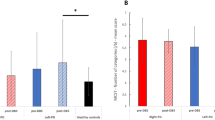
Substantial asymmetries of motor dysfunction are evident in patients with Parkinson’s disease (PD), the mechanisms of which remain largely unexplored. This study investigated how deep brain stimulation (DBS) targeting the globus pallidus interna (GPi) and subthalamic nucleus (STN) modulates characteristics of hemispheric lateralization in PD patients, with particular emphasis on motor asymmetries and hemispheric integration (via homotopic functional connectivity) and segregation (via hemispheric asymmetry in connectivity). Resting-state functional magnetic resonance imaging (fMRI) and Unified Parkinson’s Disease Rating Scale (UPDRS) III scores were analyzed from 55 PD patients who underwent either bilateral GPi- or STN-DBS. Both targets produced significant improvements in motor function. Notably, stimulation effects on motor asymmetry depend on patients’ baseline asymmetry direction (DBS OFF): STN-DBS consistently reduced asymmetry in the leftward-asymmetry patients, whereas GPi-DBS has stronger effects in rightward patients. In both cases, stimulation led to a more symmetric pattern. Beyond motor outcomes, motor gains were associated with changes in homotopic connectivity in the lateral occipital region, overlapping the extrastriate body area, suggesting a compensatory role of visual networks. These findings highlight the contribution of the visual networks to motor improvement and reveal target-dependent effects of DBS on both motor asymmetry and non-motor cognitive domains.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are provided in the Supplementary Materials. Additional raw data are available from the corresponding author upon reasonable request.
References
Bloem BR, Okun MS, Klein C. Parkinson’s disease. The Lancet. 2021;397:2284–303.
Voruz P, Guérin D, Péron JA. Impact of motor symptom asymmetry on non-motor outcomes in Parkinson’s disease: a systematic review. Npj Park Dis. 2025;11:188.
Djaldetti R, Ziv I, Melamed E. The mystery of motor asymmetry in Parkinson’s disease. Lancet Neurol. 2006;5:796–802.
Ocklenburg S, Mundorf A, Gerrits R, Karlsson EM, Papadatou-Pastou M, Vingerhoets G. Clinical implications of brain asymmetries. Nat Rev Neurol. 2024;20:383–94.
Lee CS, Schulzer M, Mak E, Hammerstad JP, Calne S, Calne DB. Patterns of asymmetry do not change over the course of idiopathic Parkinsonism: implications for pathogenesis. Neurology. 1995;45:435–9.
Miller-Patterson C, Buesa R, McLaughlin N, Jones R, Akbar U, Friedmann JH. Motor asymmetry over time in Parkinson’s disease. J Neurol Sci. 2018;393:14–7.
Riederer P, Jellinger KA, Kolber P, Hipp G, Sian-Hülsmann J, Krüger R. Lateralisation in Parkinson disease. Cell Tissue Res. 2018;373:297–312.
Ricciardi L, Ricciardi D, Lena F, Plotnik M, Petracca M, Barricella S, et al. Working on asymmetry in Parkinson’s disease: randomized, controlled pilot study. Neurol Sci. 2015;36:1337–43.
Marinus J, Van Hilten JJ. The significance of motor (A)symmetry in Parkinson’s disease. Mov Disord. 2015;30:379–85.
Steinbach MJ, Campbell RW, DeVore BB, Harrison DW. Laterality in Parkinson’s disease: A neuropsychological review. Appl Neuropsychol Adult. 2023;30:126–40.
Güntürkün O, Ströckens F, Ocklenburg S. Brain lateralization: a comparative perspective. Physiol Rev. 2020;100:1019–63.
Kong XZ, Postema MC, Guadalupe T, de Kovel C, Boedhoe PSW, Hoogman M, et al. Mapping brain asymmetry in health and disease through the ENIGMA consortium. Hum Brain Mapp. 2022;43:167–81.
Toga AW, Thompson PM. Mapping brain asymmetry. Nat Rev Neurosci. 2003;4:37–48.
Bange M, Gonzalez-Escamilla G, Marquardt T, Radetz A, Dresel C, Herz D, et al. Deficient interhemispheric connectivity underlies movement irregularities in Parkinson’s disease. J Park Dis. 2022;12:381–95.
Hu X, Zhang J, Jiang X, Zhou C, Wei L, Yin X, et al. Decreased interhemispheric functional connectivity in subtypes of Parkinson’s disease. J Neurol. 2015;262:760–7.
Luo C, Guo X, Song W, Zhao B, Cao B, Yang J, et al. Decreased resting-state interhemispheric functional connectivity in parkinson’s disease. Biomed Res Int. 2015;2015:692684.
Sun J, Gao X, Hua Q, Du R, Liu P, Liu T, et al. Brain functional specialization and cooperation in Parkinson’s disease. Brain Imaging Behav. 2022;16:565–73.
Wu J, Guo T, Zhou C, Gao T, Guan X, Xuan M, et al. Disrupted interhemispheric coordination with unaffected lateralization of global eigenvector centrality characterizes hemiparkinsonism. Brain Res. 2020;1742:146888.
Gan C, Wang L, Ji M, Ma K, Sun H, Zhang K, et al. Abnormal interhemispheric resting state functional connectivity in Parkinson’s disease patients with impulse control disorders. NPJ Park Dis. 2021;7:60.
Li J, Yuan Y, Wang M, Zhang J, Zhang L, Jiang S, et al. Decreased interhemispheric homotopic connectivity in Parkinson’s disease patients with freezing of gait: a resting state fMRI study. Parkinsonism Relat Disord. 2018;52:30–6.
Zhu Y, Song X, Xu M, Hu X, Li E, Liu J, et al. Impaired interhemispheric synchrony in Parkinson’s disease with depression. Sci Rep. 2016;6:27477.
Lubben N, Ensink E, Coetzee GA, Labrie V. The enigma and implications of brain hemispheric asymmetry in neurodegenerative diseases. Brain Commun. 2021;3:fcab211.
Blesa J, Juri C, Garcia-Cabezas MA, Adanez R, Sanchez-Gonzalez MA, Cavada C, et al. Inter-hemispheric asymmetry of nigrostriatal dopaminergic lesion: a possible compensatory mechanism in Parkinson’s disease. Front Syst Neurosci. 2011;5:92.
Lin W, Zuo CT, Wu JJ, Yang LK, Zhu J, Wang YH. Striatal asymmetry index and its correlation with the Hoehn & Yahr stage in Parkinson’s disease. Int J Neurosci. 2021;132:165–70.
Prasad S, Saini J, Yadav R, Pal PK. Motor asymmetry and neuromelanin imaging: concordance in Parkinson’s disease. Parkinsonism Relat Disord. 2018;53:28–32.
Premi E, Pilotto A, Garibotto V, Bigni B, Turrone R, Alberici A, et al. Impulse control disorder in PD: a lateralized monoaminergic frontostriatal disconnection syndrome? Parkinsonism Relat Disord. 2016;30:62–6.
Tang CC, Holtbernd F, Ma Y, Spetsieris P, Oh A, Fink GR, et al. Hemispheric network expression in parkinson’s disease: relationship to dopaminergic asymmetries. J Park Dis. 2020;10:1737–49.
Filippi M, Sarasso E, Piramide N, Stojkovic T, Stankovic I, Basaia S, et al. Progressive brain atrophy and clinical evolution in Parkinson’s disease. Neuroimage Clin. 2020;28:102374.
Claassen DO, McDonell KE, Donahue M, Rawal S, Wylie SA, Neimat JS, et al. Cortical asymmetry in Parkinson’s disease: early susceptibility of the left hemisphere. Brain Behav. 2016;6:e00573.
Hughes AJ, Ben-Shlomo Y, Daniel SE, Lees AJ. What features improve the accuracy of clinical diagnosis in Parkinson’s disease: a clinicopathologic study. Neurology. 1992;42:1142.
Roussakis AA, Zeng Z, Lao-Kaim NP, Martin-Bastida A, Piccini P, Barker RA, et al. Parkinson’s disease laterality: a 11C-PE2I PET imaging study. J Neurol. 2021;268:582–9.
Barbosa RP, Moreau C, Rolland AS, Rascol O, Brefel-Courbon C, Ory-Magne F, et al. The impact of subthalamic deep-brain stimulation in restoring motor symmetry in Parkinson’s disease patients: a prospective study. J Neurol. 2024;271:2582–95.
Lin Z, Zhang C, Sun B, Li D. Subthalamic stimulation changes motor laterality in Parkinson’s disease. Clin Park Relat Disord. 2020;3:100081.
Park HR, Ha S, Lee DS, Im HJ, Paek SH. Determination of Parkinson disease laterality after deep brain stimulation using 123I FP-CIT SPECT. Clin Nucl Med. 2020;45:e178.
Johnsen EL, Mogensen PH, Sunde NA, Ostergaard K. Improved asymmetry of gait in Parkinson’s disease with DBS: gait and postural instability in Parkinson’s disease treated with bilateral deep brain stimulation in the subthalamic nucleus. Mov Disord. 2009;24:590–7.
Ravi DK, Baumann CR, Bernasconi E, Gwerder M, Ignasiak NK, Uhl M, et al. Does subthalamic deep brain stimulation impact asymmetry and dyscoordination of gait in Parkinson’s disease? Neurorehabil Neural Repair. 2021;35:1020–9.
Jakobs M, Fomenko A, Lozano AM, Kiening KL. Cellular, molecular, and clinical mechanisms of action of deep brain stimulation—a systematic review on established indications and outlook on future developments. EMBO Mol Med. 2019;11:e9575.
Lizarraga K, Luca C, De Salles A, Gorgulho A, Lang A, Fasano A. Asymmetric neuromodulation of motor circuits in Parkinson’s disease: The role of subthalamic deep brain stimulation. Surg Neurol Int. 2017;8:261.
Liu Y, Li F, Luo H, He Q, Chen L, Cheng Y, et al. Improvement of deep brain stimulation in dyskinesia in Parkinson’s disease: a meta-analysis. Front Neurol. 2019;10:151.
Anderson VC, Burchiel KJ, Hogarth P, Favre J, Hammerstad JP. Pallidal vs subthalamic nucleus deep brain stimulation in parkinson disease. Arch Neurol. 2005;62:554.
Sharma VD, Patel M, Miocinovic S. Surgical treatment of Parkinson’s disease: devices and lesion approaches. Neurotherapeutics. 2020;17:1525–38.
Georgiev D, Akram H, Jahanshahi M. Deep brain stimulation for psychiatric disorders: role of imaging in identifying/confirming DBS targets, predicting, and optimizing outcome and unravelling mechanisms of action. Psychoradiology. 2021;1:118–51.
Tinaz S. Functional connectome in Parkinson’s disease and Parkinsonism. Curr Neurol Neurosci Rep. 2021;21:24.
Luo B, Dong W, Chang L, Qiu C, Lu Y, Liu D, et al. Altered interhemispheric functional connectivity associated with early verbal fluency decline after deep brain stimulation in Parkinson’s disease. Front Aging Neurosci. 2022;14:799545.
Wang J, Shang R, He L, Zhou R, Chen Z, Ma Y, et al. Prediction of deep brain stimulation outcome in Parkinson’s disease with connectome based on hemispheric asymmetry. Front Neurosci. 2021;15:620750.
Chu C, He N, Zeljic K, Zhang Z, Wang J, Li J, et al. Subthalamic and pallidal stimulation in Parkinson’s disease induce distinct brain topological reconstruction. Neuroimage. 2022;255:119196.
Zhang C, Lai Y, Li J, He N, Liu Y, Li Y, et al. Subthalamic and pallidal stimulations in patients with Parkinson’s disease: common and dissociable connections. Ann Neurol. 2021;90:670–82.
Li Z, Lai Y, Li J, He N, Li D, Yan F, et al. BOLD frequency–dependent alterations in resting-state functional connectivity by pallidal deep brain stimulation in patients with Parkinson’s disease. 2023[cited 2024 Sept 23]; Available from: https://thejns.org/view/journals/j-neurosurg/139/5/article-p1354.xml
Joliot M, Jobard G, Naveau M, Delcroix N, Petit L, Zago L, et al. AICHA: An atlas of intrinsic connectivity of homotopic areas. J Neurosci Methods. 2015;254:46–59.
Fox MD, Corbetta M, Snyder AZ, Vincent JL, Raichle ME. Spontaneous neuronal activity distinguishes human dorsal and ventral attention systems. Proc Natl Acad Sci. 2006;103:10046–51.
Vingerhoets G, Verhelst H, Gerrits R, Badcock N, Bishop DVM, Carey D, et al. Laterality indices consensus initiative (LICI): A Delphi expert survey report on recommendations to record, assess, and report asymmetry in human behavioural and brain research. Laterality. 2023;28:122–91.
Yarkoni T, Poldrack RA, Nichols TE, Van Essen DC, Wager TD. Large-scale automated synthesis of human functional neuroimaging data. Nat Methods. 2011;8:665–70.
Karolis VR, Corbetta M, Thiebaut de Schotten M. The architecture of functional lateralisation and its relationship to callosal connectivity in the human brain. Nat Commun. 2019;10:1417.
Greene DJ, Marek S, Gordon EM, Siegel JS, Gratton C, Laumann TO, et al. Integrative and network-specific connectivity of the basal ganglia and thalamus defined in individuals. Neuron. 2020;105:742–58.e6.
Hansen JY, Shafiei G, Markello RD, Smart K, Cox SML, Nørgaard M, et al. Mapping neurotransmitter systems to the structural and functional organization of the human neocortex. Nat Neurosci. 2022;25:1569–81.
Hansen JY, Markello RD, Vogel JW, Seidlitz J, Bzdok D, Misic B. Mapping gene transcription and neurocognition across human neocortex. Nat Hum Behav. 2021;5:1240–50.
Kohoutová L, Heo J, Cha S, Lee S, Moon T, Wager TD, et al. Toward a unified framework for interpreting machine-learning models in neuroimaging. Nat Protoc. 2020;15:1399–435.
Hettwer MD, Larivière S, Park BY, van den Heuvel OA, Schmaal L, Andreassen OA, et al. Coordinated cortical thickness alterations across six neurodevelopmental and psychiatric disorders. Nat Commun. 2022;13:6851.
Markello RD, Hansen JY, Liu ZQ, Bazinet V, Shafiei G, Suárez LE, et al. neuromaps: structural and functional interpretation of brain maps. Nat Methods. 2022;19:1472–9.
Fox MD, Zhang D, Snyder AZ, Raichle ME. The global signal and observed anticorrelated resting state brain networks. J Neurophysiol. 2009;101:3270–83.
Murphy K, Birn RM, Handwerker DA, Jones TB, Bandettini PA. The impact of global signal regression on resting state correlations: are anti-correlated networks introduced? Neuroimage. 2009;44:893–905.
Huang R, Wang K, Zhang Y, Zhao G. Symptom-specific networks and the DBS-modulated network in Parkinson’s disease: a connectivity-based review. Brain Sci. 2025;16:16.
Horn A, Li N, Dembek TA, Kappel A, Boulay C, Ewert S, et al. Lead-DBS v2: Towards a comprehensive pipeline for deep brain stimulation imaging. Neuroimage. 2019;184:293–316.
Miao J, Tantawi M, Koa V, Zhang AB, Zhang V, Sharan A, et al. Use of functional mri in deep brain stimulation in Parkinson’s diseases: a systematic review. Front Neurol. 2022;13:849918.
Parker AJ, Woodhead ZVJ, Carey DP, Groen MA, Gutierrez-Sigut E, Hodgson J, et al. Inconsistent language lateralisation – Testing the dissociable language laterality hypothesis using behaviour and lateralised cerebral blood flow. Cortex. 2022;154:105–34.
Kong XZ, Postema M, Schijven D, Castillo AC, Pepe A, Crivello F, et al. Large-scale phenomic and genomic analysis of brain asymmetrical skew. Cereb Cortex. 2021;31:4151–68.
Chormai P, Pu Y, Hu H, Fisher SE, Francks C, Kong XZ. Machine learning of large-scale multimodal brain imaging data reveals neural correlates of hand preference. Neuroimage. 2022;262:119534.
Karlsson EM, Johnstone LT, Carey DP. The depth and breadth of multiple perceptual asymmetries in right handers and non-right handers. Laterality Asymmetries Body Brain Cogn. 2019;24:707–39.
Johnstone LT, Karlsson EM, Carey DP. Left-handers are less lateralized than right-handers for both left and right hemispheric functions. Cereb Cortex. 2021;31:3780–7.
Li Xin-tian. The distribution of left and right handedness in Chinese people. Acta Psychol Sin. 1983;15:27–35.
Kong XZ, Huang Y, Hao X, Hu S, Liu J. Sex-linked association between cortical scene selectivity and navigational ability. Neuroimage. 2017;158:397–405.
Johansson ME, Toni I, Kessels RPC, Bloem BR, Helmich RC. Clinical severity in Parkinson’s disease is determined by decline in cortical compensation. Brain. 2024;147:871–86.
Myers A, Sowden PT. Your hand or mine? The extrastriate body area. Neuroimage. 2008;42:1669–77.
Urgesi C, Berlucchi G, Aglioti SM. Magnetic stimulation of extrastriate body area impairs visual processing of nonfacial body parts. Curr Biol. 2004;14:2130–4.
Astafiev SV, Stanley CM, Shulman GL, Corbetta M. Extrastriate body area in human occipital cortex responds to the performance of motor actions. Nat Neurosci. 2004;7:542–8.
Helmich RC, de Lange FP, Bloem BR, Toni I. Cerebral compensation during motor imagery in Parkinson’s disease. Neuropsychologia. 2007;45:2201–15.
van Nuenen BF, Helmich RC, Buenen N, van de Warrenburg BP, Bloem BR, Toni I. Compensatory activity in the extrastriate body area of Parkinson’s disease patients. J Neurosci. 2012;32:9546–53.
Frizon LA, Gopalakrishnan R, Hogue O, Floden D, Nagel SJ, Baker KB, et al. Cortical thickness in visuo-motor areas is related to motor outcomes after STN DBS for Parkinson’s disease. Parkinsonism Relat Disord. 2020;71:17–22.
Min HK, Hwang SC, Marsh MP, Kim I, Knight E, Striemer B, et al. Deep brain stimulation induces BOLD activation in motor and non-motor networks: an fMRI comparison study of STN and EN/GPi DBS in large animals. Neuroimage. 2012;63:1408–20.
Ramirez-Zamora A, Ostrem JL. Globus pallidus interna or subthalamic nucleus deep brain stimulation for Parkinson disease: a review. JAMA Neurol. 2018;75:367–72.
Kong XZ, Group ELW, Francks C. Reproducibility in the absence of selective reporting: an illustration from large-scale brain asymmetry research. Hum Brain Mapp. 2022;43:244–54.
Marek S, Tervo-Clemmens B, Calabro FJ, Montez DF, Kay BP, Hatoum AS, et al. Reproducible brain-wide association studies require thousands of individuals. Nature. 2022;603:654–60.
Thompson PM, Jahanshad N, Ching CRK, Salminen LE, Thomopoulos SI, Bright J, et al. ENIGMA and global neuroscience: a decade of large-scale studies of the brain in health and disease across more than 40 countries. Transl Psychiatry. 2020;10:1–28.
Acknowledgements
Xiang-Zhen Kong was supported by Brain Science and Brain-like Intelligence Technology-National Science and Technology Major Project (2025ZD0218801; 2021ZD0200409), National Natural Science Foundation of China (32171031; 32571219), Fundamental Research Funds for the Central Universities (226-2025-00144), and Information Technology Center of Zhejiang University. Chencheng Zhang was supported by National Natural Science Foundation of China (82101547; 82471495). Yi Pu was supported by National Natural Science Foundation of China (32400882).
Author information
Authors and Affiliations
Contributions
SM: study conception and design, data preparation, analysis, visualization, preparing the first draft and editing; NH: data collection, study conception and design, manuscript editing; WH, JY, CC, ZZ, YF, DL: data collection and manuscript editing; YP: manuscript editing; CZ: data collection, study conception and design, manuscript editing; XK: study conception and design, data preparation, analysis, visualization, preparing the first draft and editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mei, S., He, N., He, W. et al. Pallidal and subthalamic stimulations modulate inter-hemispheric interaction and asymmetry in Parkinson’s disease. Mol Psychiatry (2026). https://doi.org/10.1038/s41380-026-03557-9
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41380-026-03557-9