Abstract
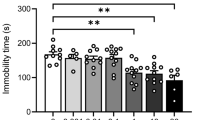
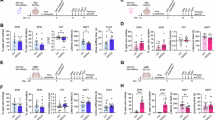
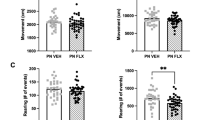
Chronic stress induces neurobiological adaptations that manifest as altered behavioral patterns in both humans and animal models. To enhance the translational value of preclinical research, we systematically evaluated chronic restraint stress (CRS) protocols in mice through longitudinal tracking of behavioral outcomes across multiple validated assays. By comparing various CRS parameters, we identified specific protocols that elicited persistent behavioral adaptations across distinct measurements in male mice. Short-duration, high-intensity CRS (6 h/day for 3 days) induced persistent phenotypes of avoidance-related and repetitive behaviors in multiple assays of approach-avoidance conflict, whereas prolonged CRS exposure (2 h/day for 10-14 days) progressively disrupted reward-seeking and behavioral coping phenotypes. When prolonging CRS exposure, we observed a behavioral transition from the initial phenotypes of avoidance/repetitive behavior to the later deficits of reward seeking/behavioral coping, accompanied by a progressive dissociation between these behavioral domains. The 10-day CRS protocol represents a critical threshold for inducing reward-seeking deficit, as well as a comorbid model of avoidance-related response and reward-processing impairment. Rapid antidepressant ketamine reversed impairments of reward seeking and behavioral coping, and typical antidepressant/anxiolytic paroxetine alleviated both repetitive/avoidance-related behaviors and coping/reward-seeking deficits. These findings demonstrated the face, construct, and predictive validity of CRS as a male mouse model of stress-related neuropsychiatric disorders. Leveraging comprehensive behavioral characterization across diverse CRS protocols, our study provides standardized protocols for recapitulating clinically-relevant behavioral adaptations to chronic stress.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
Disease GBD, Injury, Risk Factor C. Burden of 375 diseases and injuries, risk-attributable burden of 88 risk factors, and healthy life expectancy in 204 countries and territories, including 660 subnational locations, 1990-2023: a systematic analysis for the Global Burden of Disease Study 2023. Lancet. 2025;406:1873–922.
Rush AJ, Trivedi MH, Wisniewski SR, Nierenberg AA, Stewart JW, Warden D, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163:1905–17.
Szuhany KL, Simon NM. Anxiety disorders: a review. JAMA. 2022;328:2431–45.
Marx W, Penninx B, Solmi M, Furukawa TA, Firth J, Carvalho AF, et al. Major depressive disorder. Nat Rev Dis Primers. 2023;9:44.
Simon GE, Moise N, Mohr DC. Management of Depression in Adults: A Review. JAMA. 2024;332:141–52.
Scangos KW, State MW, Miller AH, Baker JT, Williams LM. New and emerging approaches to treat psychiatric disorders. Nat Med. 2023;29:317–33.
Newport DJ, Carpenter LL, McDonald WM, Potash JB, Tohen M, Nemeroff CB, et al. Ketamine and other NMDA antagonists: early clinical trials and possible mechanisms in depression. Am J Psychiatry. 2015;172:950–66.
Shemesh Y, Chen A. A paradigm shift in translational psychiatry through rodent neuroethology. Mol Psychiatry. 2023;28:993–1003.
Gururajan A, Reif A, Cryan JF, Slattery DA. The future of rodent models in depression research. Nat Rev Neurosci. 2019;20:686–701.
von Mucke-Heim IA, Urbina-Trevino L, Bordes J, Ries C, Schmidt MV, Deussing JM. Introducing a depression-like syndrome for translational neuropsychiatry: a plea for taxonomical validity and improved comparability between humans and mice. Mol Psychiatry. 2023;28:329–40.
Chang H, Hoshina N, Zhang C, Ma Y, Cao H, Wang Y, et al. The protocadherin 17 gene affects cognition, personality, amygdala structure and function, synapse development and risk of major mood disorders. Mol Psychiatry. 2018;23:400–12.
Grotzinger AD, Werme J, Peyrot WJ, Frei O, de Leeuw C, Bicks LK, et al. Mapping the genetic landscape across 14 psychiatric disorders. Nature. 2026;649:406–15.
Als TD, Kurki MI, Grove J, Voloudakis G, Therrien K, Tasanko E, et al. Depression pathophysiology, risk prediction of recurrence and comorbid psychiatric disorders using genome-wide analyses. Nat Med. 2023;29:1832–44.
He H, Cathomas F, Parise LF, David E, Rizk M, Hawkins K, et al. Major depressive disorder shares systemic immune signatures and potential therapeutic targets with inflammatory skin diseases. Mol Psychiatry. 2026.
Malezieux M, Klein AS, Gogolla N. Neural Circuits for Emotion. Annu Rev Neurosci. 2023;46:211–31.
Drevets WC, Wittenberg GM, Bullmore ET, Manji HK. Immune targets for therapeutic development in depression: towards precision medicine. Nat Rev Drug Discov. 2022;21:224–44.
Cathomas F, Lin HY, Chan KL, Li L, Parise LF, Alvarez J, et al. Circulating myeloid-derived MMP8 in stress susceptibility and depression. Nature. 2024;626:1108–15.
Yang Z, Zhou D, Li H, Cai X, Liu W, Wang L, et al. The genome-wide risk alleles for psychiatric disorders at 3p21.1 show convergent effects on mRNA expression, cognitive function, and mushroom dendritic spine. Mol Psychiatry. 2020;25:48–66.
Zhang Y, Du L, Bai Y, Han B, He C, Gong L, et al. CircDYM ameliorates depressive-like behavior by targeting miR-9 to regulate microglial activation via HSP90 ubiquitination. Mol Psychiatry. 2020;25:1175–90.
Li S, Li Y, Li X, Liu J, Huo Y, Wang J, et al. Regulatory mechanisms of major depressive disorder risk variants. Mol Psychiatry. 2020;25:1926–45.
Shi Y, Wang Q, Song R, Kong Y, Zhang Z. Non-coding RNAs in depression: Promising diagnostic and therapeutic biomarkers. EBioMedicine. 2021;71:103569.
Antunes C, Da Silva JD, Guerra-Gomes S, Alves ND, Ferreira F, Loureiro-Campos E, et al. Tet3 ablation in adult brain neurons increases anxiety-like behavior and regulates cognitive function in mice. Mol Psychiatry. 2021;26:1445–57.
Vickstrom CR, Liu X, Liu S, Hu MM, Mu L, Hu Y, et al. Role of endocannabinoid signaling in a septohabenular pathway in the regulation of anxiety- and depressive-like behavior. Mol Psychiatry. 2021;26:3178–91.
Karayol R, Medrihan L, Warner-Schmidt JL, Fait BW, Rao MN, Holzner EB, et al. Serotonin receptor 4 in the hippocampus modulates mood and anxiety. Mol Psychiatry. 2021;26:2334–49.
Umschweif G, Medrihan L, McCabe KA, Sagi Y, Greengard P. Activation of the p11/SMARCA3/Neurensin-2 pathway in parvalbumin interneurons mediates the response to chronic antidepressants. Mol Psychiatry. 2021;26:3350–62.
Xiao X, Zhang CY, Zhang Z, Hu Z, Li M, Li T. Revisiting tandem repeats in psychiatric disorders from perspectives of genetics, physiology, and brain evolution. Mol Psychiatry. 2022;27:466–75.
Wang H, Cui W, Chen W, Liu F, Dong Z, Xing G, et al. The laterodorsal tegmentum-ventral tegmental area circuit controls depression-like behaviors by activating ErbB4 in DA neurons. Mol Psychiatry. 2023;28:1027–45.
O’Leary A, Fernandez-Castillo N, Gan G, Yang Y, Yotova AY, Kranz TM, et al. Behavioural and functional evidence revealing the role of RBFOX1 variation in multiple psychiatric disorders and traits. Mol Psychiatry. 2022;27:4464–73.
Codagnone MG, Kara N, Ratsika A, Levone BR, van de Wouw M, Tan LA, et al. Inhibition of FKBP51 induces stress resilience and alters hippocampal neurogenesis. Mol Psychiatry. 2022;27:4928–38.
Nold V, Portenhauser M, Del Prete D, Blasius A, Harris I, Koros E, et al. Impact of Fkbp5 x early life adversity x sex in humanised mice on multidimensional stress responses and circadian rhythmicity. Mol Psychiatry. 2022;27:3544–55.
Kemp GM, Altimimi HF, Nho Y, Heir R, Klyczek A, Stellwagen D. Sustained TNF signaling is required for the synaptic and anxiety-like behavioral response to acute stress. Mol Psychiatry. 2022;27:4474–84.
Liu WZ, Huang SH, Wang Y, Wang CY, Pan HQ, Zhao K, et al. Medial prefrontal cortex input to basolateral amygdala controls acute stress-induced short-term anxiety-like behavior in mice. Neuropsychopharmacology. 2023;48:734–44.
Wu J, Li Y, Huang Y, Liu L, Zhang H, Nagy C, et al. Integrating spatial and single-nucleus transcriptomic data elucidates microglial-specific responses in female cynomolgus macaques with depressive-like behaviors. Nat Neurosci. 2023;26:1352–64.
Lin D, Li L, Chen WB, Chen J, Ren D, Zheng ZH, et al. LHPP, a risk factor for major depressive disorder, regulates stress-induced depression-like behaviors through its histidine phosphatase activity. Mol Psychiatry. 2023;28:908–18.
Lu K, Hong Y, Tao M, Shen L, Zheng Z, Fang K, et al. Depressive patient-derived GABA interneurons reveal abnormal neural activity associated with HTR2C. EMBO Mol Med. 2023;15:e16364.
Luo YF, Lu L, Song HY, Xu H, Zheng ZW, Wu ZY, et al. Divergent projections of the prelimbic cortex mediate autism- and anxiety-like behaviors. Mol Psychiatry. 2023;28:2343–54.
Li W, Chen R, Feng L, Dang X, Liu J, Chen T, et al. Genome-wide meta-analysis, functional genomics and integrative analyses implicate new risk genes and therapeutic targets for anxiety disorders. Nat Hum Behav. 2024;8:361–79.
Wang H, Wang Q, Cui L, Feng X, Dong P, Tan L, et al. A molecularly defined amygdala-independent tetra-synaptic forebrain-to-hindbrain pathway for odor-driven innate fear and anxiety. Nat Neurosci. 2024;27:514–26.
Nagarajan N, Capecchi MR. Optogenetic stimulation of mouse Hoxb8 microglia in specific regions of the brain induces anxiety, grooming, or both. Mol Psychiatry. 2024;29:1726–40.
Pu J, Liu Y, Wu H, Liu C, Chen Y, Tang W, et al. Characterizing metabolomic and proteomic changes in depression: a systematic analysis. Mol Psychiatry. 2025.
Huang Q, Lee HH, Volpe B, Zhang Q, Xue C, Liu BC, et al. Deletion of murine astrocytic vesicular nucleotide transporter increases anxiety and depressive-like behavior and attenuates motivation for reward. Mol Psychiatry. 2025;30:506–20.
Tripathi A, Bartosh A, Whitehead C, Pillai A. Activation of cell-free mtDNA-TLR9 signaling mediates chronic stress-induced social behavior deficits. Mol Psychiatry. 2023;28:3806–15.
Scarborough J, Mueller FS, Weber-Stadlbauer U, Mattei D, Opitz L, Cattaneo A, et al. A novel murine model to study the impact of maternal depression and antidepressant treatment on biobehavioral functions in the offspring. Mol Psychiatry. 2021;26:6756–72.
Nestler EJ, Russo SJ. Neurobiological basis of stress resilience. Neuron. 2024;112:1911–29.
Lim BK, Huang KW, Grueter BA, Rothwell PE, Malenka RC. Anhedonia requires MC4R-mediated synaptic adaptations in nucleus accumbens. Nature. 2012;487:183–9.
Fox ME, Lobo MK. The molecular and cellular mechanisms of depression: a focus on reward circuitry. Mol Psychiatry. 2019;24:1798–815.
Huang L, Xi Y, Peng Y, Yang Y, Huang X, Fu Y, et al. A visual circuit related to habenula underlies the antidepressive effects of light therapy. Neuron. 2019;102:128–42.e128
Shen CJ, Zheng D, Li KX, Yang JM, Pan HQ, Yu XD, et al. Cannabinoid CB(1) receptors in the amygdalar cholecystokinin glutamatergic afferents to nucleus accumbens modulate depressive-like behavior. Nat Med. 2019;25:337–49.
Bedse G, Hill MN, Patel S. 2-Arachidonoylglycerol modulation of anxiety and stress adaptation: from grass roots to novel therapeutics. Biol Psychiatry. 2020;88:520–30.
Liu WZ, Zhang WH, Zheng ZH, Zou JX, Liu XX, Huang SH, et al. Identification of a prefrontal cortex-to-amygdala pathway for chronic stress-induced anxiety. Nat Commun. 2020;11:2221.
Pignatelli M, Tejeda HA, Barker DJ, Bontempi L, Wu J, Lopez A, et al. Cooperative synaptic and intrinsic plasticity in a disynaptic limbic circuit drive stress-induced anhedonia and passive coping in mice. Mol Psychiatry. 2021;26:1860–79.
Xiao Q, Zhou X, Wei P, Xie L, Han Y, Wang J, et al. A new GABAergic somatostatin projection from the BNST onto accumbal parvalbumin neurons controls anxiety. Mol Psychiatry. 2021;26:4719–41.
Prevot T, Sibille E. Altered GABA-mediated information processing and cognitive dysfunctions in depression and other brain disorders. Mol Psychiatry. 2021;26:151–67.
Shi DD, Zhang YD, Ren YY, Peng SY, Yuan TF, Wang Z. Predictable maternal separation confers adult stress resilience via the medial prefrontal cortex oxytocin signaling pathway in rats. Mol Psychiatry. 2021;26:7296–307.
Zhu X, Grace AA. Sex- and exposure age-dependent effects of adolescent stress on ventral tegmental area dopamine system and its afferent regulators. Mol Psychiatry. 2023;28:611–24.
Zheng Z, Guo C, Li M, Yang L, Liu P, Zhang X, et al. Hypothalamus-habenula potentiation encodes chronic stress experience and drives depression onset. Neuron. 2022;110:1400–15.e1406
Yu Z, Han Y, Hu D, Chen N, Zhang Z, Chen W, et al. Neurocan regulates vulnerability to stress and the anti-depressant effect of ketamine in adolescent rats. Mol Psychiatry. 2022;27:2522–32.
Parekh PK, Johnson SB, Liston C. Synaptic mechanisms regulating mood state transitions in depression. Annu Rev Neurosci. 2022;45:581–601.
Ren LY, Cicvaric A, Zhang H, Meyer MA, Guedea AL, Gao P, et al. Stress-induced changes of the cholinergic circuitry promote retrieval-based generalization of aversive memories. Mol Psychiatry. 2022;27:3795–805.
Wang ZJ, Shwani T, Liu J, Zhong P, Yang F, Schatz K, et al. Molecular and cellular mechanisms for differential effects of chronic social isolation stress in males and females. Mol Psychiatry. 2022;27:3056–68.
Lu J, Zhang Z, Yin X, Tang Y, Ji R, Chen H, et al. An entorhinal-visual cortical circuit regulates depression-like behaviors. Mol Psychiatry. 2022;27:3807–20.
Wang D, Pan X, Zhou Y, Wu Z, Ren K, Liu H, et al. Lateral septum-lateral hypothalamus circuit dysfunction in comorbid pain and anxiety. Mol Psychiatry. 2023;28:1090–1100.
Li HY, Zhu MZ, Yuan XR, Guo ZX, Pan YD, Li YQ, et al. A thalamic-primary auditory cortex circuit mediates resilience to stress. Cell. 2023;186:1352–68.e1318
Hu P, Wang Y, Qi XH, Shan QH, Huang ZH, Chen P, et al. SIRT1 in the BNST modulates chronic stress-induced anxiety of male mice via FKBP5 and corticotropin-releasing factor signaling. Mol Psychiatry. 2023;28:5101–17.
Zhang YM, Zong HC, Qi YB, Chang LL, Gao YN, Zhou T, et al. Anxiolytic effect of antidiabetic metformin is mediated by AMPK activation in mPFC inhibitory neurons. Mol Psychiatry. 2023;28:3955–65.
Zheng Z, Liu Y, Mu R, Guo X, Feng Y, Guo C, et al. A small population of stress-responsive neurons in the hypothalamus-habenula circuit mediates development of depression-like behavior in mice. Neuron. 2024;112:3924–39.e3925
Jiang T, Feng M, Hutsell A, Luscher B. Sex-specific GABAergic microcircuits that switch vulnerability into resilience to stress and reverse the effects of chronic stress exposure. Mol Psychiatry 2025;30:2297–308.
Huang T, Guo X, Huang X, Yi C, Cui Y, Dong Y. Input-output specific orchestration of aversive valence in lateral habenula during stress dynamics. J Zhejiang Univ Sci B. 2024;25:1055–65.
Sanacora G, Yan Z, Popoli M. The stressed synapse 2.0: pathophysiological mechanisms in stress-related neuropsychiatric disorders. Nat Rev Neurosci. 2022;23:86–103.
Fries GR, Saldana VA, Finnstein J, Rein T. Molecular pathways of major depressive disorder converge on the synapse. Mol Psychiatry. 2023;28:284–97.
Musazzi L, Mingardi J, Ieraci A, Barbon A, Popoli M. Stress, microRNAs, and stress-related psychiatric disorders: an overview. Mol Psychiatry. 2023;28:4977–94.
Camargo A, Nilsson A, Shariatgorji R, Appleton E, Branzell N, Doyon D, et al. Enduring modulation of dorsal raphe nuclei regulates (R,S)-ketamine-mediated resilient stress-coping behavior. Mol Psychiatry 2025;30:2504–16.
Song N, Liu Z, Gao Y, Lu S, Yang S, Yuan C. NAc-DBS corrects depression-like behaviors in CUMS mouse model via disinhibition of DA neurons in the VTA. Mol Psychiatry. 2024;29:1550–66.
Manas-Ojeda A, Hidalgo-Cortes J, Garcia-Mompo C, Zahran MA, Gil-Miravet I, Olucha-Bordonau FE, et al. Activation of somatostatin neurons in the medial amygdala reverses long-term aggression and social deficits associated to early-life stress in male mice. Mol Psychiatry. 2024.
Cheng Z, Zhao F, Piao J, Yang W, Cui R, Li B. Rasd2 regulates depression-like behaviors via DRD2 neurons in the prelimbic cortex afferent to nucleus accumbens core circuit. Mol Psychiatry. 2025;30:435–49.
Wu F, Gu C, Xu R, Ma J, Gao L, Zhang Y, et al. A visual cortical-lateral posterior thalamic nucleus circuit regulates depressive-like behaviors in male mice. Nat Commun. 2025;16:1395.
Rodrigues B, Leitao RA, Santos M, Trofimov A, Silva M, Inacio AS, et al. MiR-186-5p inhibition restores synaptic transmission and neuronal network activity in a model of chronic stress. Mol Psychiatry. 2025;30:1034–46.
Codeluppi SA, Xu M, Bansal Y, Lepack AE, Duric V, Chow M, et al. Prefrontal cortex astroglia modulate anhedonia-like behavior. Mol Psychiatry. 2023;28:4632–41.
Zhang X, Asim M, Fang W, Md Monir H, Wang H, Kim K, et al. Cholecystokinin B receptor antagonists for the treatment of depression via blocking long-term potentiation in the basolateral amygdala. Mol Psychiatry. 2023;28:3459–74.
Gonzalez-Arias C, Sanchez-Ruiz A, Esparza J, Sanchez-Puelles C, Arancibia L, Ramirez-Franco J, et al. Dysfunctional serotonergic neuron-astrocyte signaling in depressive-like states. Mol Psychiatry. 2023;28:3856–73.
Liu MY, Wei LL, Zhu XH, Ding HC, Liu XH, Li H, et al. Prenatal stress modulates HPA axis homeostasis of offspring through dentate TERT independently of glucocorticoids receptor. Mol Psychiatry. 2023;28:1383–95.
Fang X, Chen Y, Wang J, Zhang Z, Bai Y, Denney K, et al. Increased intrinsic and synaptic excitability of hypothalamic POMC neurons underlies chronic stress-induced behavioral deficits. Mol Psychiatry. 2023;28:1365–82.
Zhen F, Yu L, Wang L, Wang S, Lu W, Wang X, et al. Salvianolic acids alleviate chronic mild stress-induced depressive-like behaviors in rats. J Integr Neurosci. 2023;22:60.
Kim KS, Han PL. Optimization of chronic stress paradigms using anxiety- and depression-like behavioral parameters. J Neurosci Res. 2006;83:497–507.
Ramirez S, Liu X, MacDonald CJ, Moffa A, Zhou J, Redondo RL, et al. Activating positive memory engrams suppresses depression-like behaviour. Nature. 2015;522:335–9.
Shin S, Kwon O, Kang JI, Kwon S, Oh S, Choi J, et al. mGluR5 in the nucleus accumbens is critical for promoting resilience to chronic stress. Nat Neurosci. 2015;18:1017–24.
Moda-Sava RN, Murdock MH, Parekh PK, Fetcho RN, Huang BS, Huynh TN, et al. Sustained rescue of prefrontal circuit dysfunction by antidepressant-induced spine formation. Science. 2019;364:eaat8078.
Dong Y, Li Y, Xiang X, Xiao ZC, Hu J, Li Y, et al. Stress relief as a natural resilience mechanism against depression-like behaviors. Neuron. 2023;111:3789–801.e3786
Ma S, Chen M, Jiang Y, Xiang X, Wang S, Wu Z, et al. Sustained antidepressant effect of ketamine through NMDAR trapping in the LHb. Nature. 2023;622:802–9.
Calhoon GG, Tye KM. Resolving the neural circuits of anxiety. Nat Neurosci. 2015;18:1394–404.
Sztainberg Y, Zoghbi HY. Lessons learned from studying syndromic autism spectrum disorders. Nat Neurosci. 2016;19:1408–17.
Knoedler JR, Inoue S, Bayless DW, Yang T, Tantry A, Davis CH, et al. A functional cellular framework for sex and estrous cycle-dependent gene expression and behavior. Cell. 2022;185:654–71.e622
Wang Y, Song X, Chen X, Zhou Y, Ma J, Zhang F, et al. Integrating reproductive states and social cues in the control of sociosexual behaviors. Cell. 2025;188:3530–49.e3524
McEwen BS, Bowles NP, Gray JD, Hill MN, Hunter RG, Karatsoreos IN, et al. Mechanisms of stress in the brain. Nat Neurosci. 2015;18:1353–63.
McIntyre RS, Rosenblat JD, Nemeroff CB, Sanacora G, Murrough JW, Berk M, et al. Synthesizing the evidence for ketamine and esketamine in treatment-resistant depression: an international expert opinion on the available evidence and implementation. Am J Psychiatry. 2021;178:383–99.
Yang Y, Cui Y, Sang K, Dong Y, Ni Z, Ma S, et al. Ketamine blocks bursting in the lateral habenula to rapidly relieve depression. Nature. 2018;554:317–22.
Trivedi MH, Thase ME, Manning JS, Weisler RH. Utilizing the DSM-5 anxious distress specifier to develop treatment strategies for patients with major depressive disorder. J Clin Psychiatry 2017;78:1351–62.
Gordon JA, Dzirasa K, Petzschner FH. The neuroscience of mental illness: building toward the future. Cell. 2024;187:5858–70.
Acknowledgements
We thank Dr. Peng Shi for technical support. This work was supported by grants from the STI2030-Major projects (2022ZD0211700 to Y. Cui); the National Natural Science Foundation of China (32400830 to Y. Dong).
Author information
Authors and Affiliations
Contributions
Conceptualization, Funding acquisition, Supervision: Y Dong, Y Cui; Data curation: Z Lv, Q Xie, K Li, Z Fan; Formal analysis: Z Lv, Q Xie, Y Dong; Visualization: Z Lv, Q Xie; Investigation: Z Lv, Q Xie, K Li, Z Fan, Y Cui, Y Dong; Methodology: Z Lv, Q Xie, Y Dong, Y Cui; Writing – original draft: Y Dong, Y Cui, Z Lv; Writing – review and editing: Y Dong, Y Cui, Z Lv, Q Xie, K Li.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All methods performed in this study were conducted in accordance with the relevant guidelines and regulations. Ethical approval was obtained from the Animal Care and Use Committee of the animal facility at Zhejiang University (reference number ZJU20220377 and ZJU20230320).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lv, Z., Xie, Q., Li, K. et al. Standardized chronic restraint stress protocols reveal dynamic evolution of behavioral adaptations in male mice: implications for translational neuroscience. Mol Psychiatry (2026). https://doi.org/10.1038/s41380-026-03569-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41380-026-03569-5