Abstract
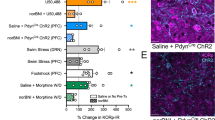
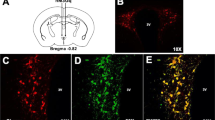
Although previous research has demonstrated a role for kappa opioid receptor-mediated signaling in escalated alcohol consumption associated with dependence and stress exposure, involvement of the dynorphin/kappa opioid receptor (DYN/KOR) system in binge-like drinking has not been fully explored. Here we used pharmacological and chemogenetic approaches to examine the influence of DYN/KOR signaling on alcohol consumption in the drinking-in-the-dark (DID) model of binge-like drinking. Systemic administration of the KOR agonist U50,488 increased binge-like drinking (Experiment 1) while, conversely, systemic administration of the KOR antagonist nor-BNI reduced drinking in the DID model (Experiment 2). These effects of systemic KOR manipulation were selective for alcohol as neither drug influenced consumption of sucrose in the DID paradigm (Experiment 3). In Experiment 4, administration of the long-acting KOR antagonist nor-BNI into the central nucleus of the amygdala (CeA) decreased alcohol intake. Next, targeted “silencing” of DYN+ neurons in the CeA was accomplished using a chemogenetic strategy. Cre-dependent viral expression in DYN+ neurons was confirmed in CeA of Pdyn-IRES-Cre mice and functionality of an inhibitory (hM4Di) DREADD was validated (Experiment 5). Activating the inhibitory DREADD by CNO injection reduced binge-like alcohol drinking, but CNO injection did not alter alcohol intake in mice that were treated with control virus (Experiment 6). Collectively, these results demonstrate that DYN/KOR signaling in the CeA contributes to excessive alcohol consumption in a binge-drinking model.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Centers for Disease Control, Prevention. Vital signs: binge drinking prevalence, frequency, and intensity among adults—United States, 2010. MMWR Morb Mortal Wkly Rep. 2012;61:14–9.
NIAAA NIoAAaA. National Institute on Alcohol Abuse and Alcoholism Council approves definition of binge drinking. NIAAA Newsl. 2004;3:3.
Jennison KM. The short-term effects and unintended long-term consequences of binge drinking in college: a 10-year follow-up study. Am J Drug Alcohol Abus. 2004;30:659–84.
McCarty CA, Ebel BE, Garrison MM, DiGiuseppe DL, Christakis DA, Rivara FP. Continuity of binge and harmful drinking from late adolescence to early adulthood. Pediatrics . 2004;114:714–9. https://doi.org/10.1542/peds.2003-0864-L
Koob GF. Neurocircuitry of alcohol addiction: synthesis from animal models. Handb Clin Neurol. 2014;125:33–54. https://doi.org/10.1016/B978-0-444-62619-6.00003-3
Koob GF, Volkow ND. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry. 2016;3:760–73. https://doi.org/10.1016/S2215-0366(16)00104-8
Crabbe JC, Harris RA, Koob GF. Preclinical studies of alcohol binge drinking. Ann N Y Acad Sci. 2011;1216:24–40. https://doi.org/10.1111/j.1749-6632.2010.05895.x
Rhodes JS, Best K, Belknap JK, Finn DA, Crabbe JC. Evaluation of a simple model of ethanol drinking to intoxication in C57BL/6J mice. Physiol Behav. 2005;84:53–63. https://doi.org/10.1016/j.physbeh.2004.10.007
Thiele TE, Crabbe JC, Boehm SL II. “Drinking in the Dark” (DID): a simple mouse model of binge-like alcohol intake. Curr Protoc Neurosci. 2014;68:9 49 1–12. https://doi.org/10.1002/0471142301.ns0949s68
Thiele TE, Navarro M. “Drinking in the dark” (DID) procedures: a model of binge-like ethanol drinking in non-dependent mice. Alcohol . 2014;48:235–41. https://doi.org/10.1016/j.alcohol.2013.08.005
Rhodes JS, Ford MM, Yu CH, Brown LL, Finn DA, Garland T Jr., et al. Mouse inbred strain differences in ethanol drinking to intoxication. Genes Brain Behav. 2007;6:1–18. https://doi.org/10.1111/j.1601-183X.2006.00210.x
Crabbe JC, Metten P, Rhodes JS, Yu CH, Brown LL, Phillips TJ, et al. A line of mice selected for high blood ethanol concentrations shows drinking in the dark to intoxication. Biol Psychiatry. 2009;65:662–70. https://doi.org/10.1016/j.biopsych.2008.11.002
Sprow GM, Thiele TE. The neurobiology of binge-like ethanol drinking: evidence from rodent models. Physiol Behav. 2012;106:325–31. https://doi.org/10.1016/j.physbeh.2011.12.026
Lowery EG, Spanos M, Navarro M, Lyons AM, Hodge CW, Thiele TE. CRF-1 antagonist and CRF-2 agonist decrease binge-like ethanol drinking in C57BL/6J mice independent of the HPA axis. Neuropsychopharmacology . 2010;35:1241–52. https://doi.org/10.1038/npp.2009.209
Lowery-Gionta EG, Navarro M, Li C, Pleil KE, Rinker JA, Cox BR, et al. Corticotropin releasing factor signaling in the central amygdala is recruited during binge-like ethanol consumption in C57BL/6J mice. J Neurosci. 2012;32:3405–13. https://doi.org/10.1523/JNEUROSCI.6256-11.2012
Rinker JA, Marshall SA, Mazzone CM, Lowery-Gionta EG, Gulati V, Pleil KE, et al. Extended amygdala to ventral tegmental area corticotropin-releasing factor circuit controls binge ethanol intake. Biol Psychiatry. 2017;81:930–40. https://doi.org/10.1016/j.biopsych.2016.02.029
Pleil KE, Rinker JA, Lowery-Gionta EG, Mazzone CM, McCall NM, Kendra AM, et al. NPY signaling inhibits extended amygdala CRF neurons to suppress binge alcohol drinking. Nat Neurosci. 2015;18:545–52. https://doi.org/10.1038/nn.3972
Sparrow AM, Lowery-Gionta EG, Pleil KE, Li C, Sprow GM, Cox BR, et al. Central neuropeptide Y modulates binge-like ethanol drinking in C57BL/6J mice via Y1 and Y2 receptors. Neuropsychopharmacology . 2012;37:1409–21. https://doi.org/10.1038/npp.2011.327
Anderson RI, Becker HC, Adams BL, Jesudason CD, Rorick-Kehn LM. Orexin-1 and orexin-2 receptor antagonists reduce ethanol self-administration in high-drinking rodent models. Front Neurosci. 2014;8:33 https://doi.org/10.3389/fnins.2014.00033
Olney JJ, Navarro M, Thiele TE. Binge-like consumption of ethanol and other salient reinforcers is blocked by orexin-1 receptor inhibition and leads to a reduction of hypothalamic orexin immunoreactivity. Alcohol Clin Exp Res. 2015;39:21–9. https://doi.org/10.1111/acer.12591
King CE, Griffin WC, Luderman LN, Kates MM, McGinty JF, Becker HC. Oxytocin reduces ethanol self-administration in mice. Alcohol Clin Exp Res. 2017;41:955–64. https://doi.org/10.1111/acer.13359
Anderson RI, Becker HC. Role of the dynorphin/kappa opioid receptor system in the motivational effects of ethanol. Alcohol Clin Exp Res. 2017;41:1402–18. https://doi.org/10.1111/acer.13406
Chavkin C. Dynorphin—still an extraordinarily potent opioid peptide. Mol Pharmacol. 2013;83:729–36. https://doi.org/10.1124/mol.112.083337
Anderson RI, Moorman DE, Becker HC. Contribution of Dynorphin and Orexin Neuropeptide Systems to the Motivational Effects of Alcohol. Handb Exp Pharmacol. 2018. https://doi.org/10.1007/164_2018_100
Bruchas MR, Land BB, Chavkin C. The dynorphin/kappa opioid system as a modulator of stress-induced and pro-addictive behaviors. Brain Res. 2010;1314:44–55. https://doi.org/10.1016/j.brainres.2009.08.062
Crowley NA, Kash TL. Kappa opioid receptor signaling in the brain: circuitry and implications for treatment. Prog Neuropsychopharmacol Biol Psychiatry. 2015;62:51–60. https://doi.org/10.1016/j.pnpbp.2015.01.001
Wee S, Koob GF. The role of the dynorphin-kappa opioid system in the reinforcing effects of drugs of abuse. Psychopharmacol (Berl). 2010;210:121–35. https://doi.org/10.1007/s00213-010-1825-8
Berger AL, Williams AM, McGinnis MM, Walker BM. Affective cue-induced escalation of alcohol self-administration and increased 22-kHz ultrasonic vocalizations during alcohol withdrawal: role of kappa-opioid receptors. Neuropsychopharmacology . 2013;38:647–54. https://doi.org/10.1038/npp.2012.229
Kissler JL, Sirohi S, Reis DJ, Jansen HT, Quock RM, Smith DG, et al. The one-two punch of alcoholism: role of central amygdala dynorphins/kappa-opioid receptors. Biol Psychiatry. 2014;75:774–82. https://doi.org/10.1016/j.biopsych.2013.03.014
Rose JH, Karkhanis AN, Chen R, Gioia D, Lopez MF, Becker HC et al. Supersensitive Kappa Opioid Receptors Promotes Ethanol Withdrawal-Related Behaviors and Reduce Dopamine Signaling in the Nucleus Accumbens. Int J Neuropsychopharmacol. 2016;19. https://doi.org/10.1093/ijnp/pyv127
Walker BM, Koob GF. Pharmacological evidence for a motivational role of kappa-opioid systems in ethanol dependence. Neuropsychopharmacology . 2008;33:643–52. https://doi.org/10.1038/sj.npp.1301438
Walker BM, Zorrilla EP, Koob GF. Systemic kappa-opioid receptor antagonism by nor-binaltorphimine reduces dependence-induced excessive alcohol self-administration in rats. Addict Biol. 2011;16:116–9. https://doi.org/10.1111/j.1369-1600.2010.00226.x
Anderson RI, Lopez MF, Becker HC. Stress-induced enhancement of ethanol intake in c57bl/6j mice with a history of chronic ethanol exposure: involvement of kappa opioid receptors. Front Cell Neurosci. 2016;10:45 https://doi.org/10.3389/fncel.2016.00045
Karkhanis AN, Rose JH, Weiner JL, Jones SR. Early-life social isolation stress increases kappa opioid receptor responsiveness and downregulates the dopamine system. Neuropsychopharmacology . 2016;41:2263–74. https://doi.org/10.1038/npp.2016.21
Racz I, Markert A, Mauer D, Stoffel-Wagner B, Zimmer A. Long-term ethanol effects on acute stress responses: modulation by dynorphin. Addict Biol. 2013;18:678–88. https://doi.org/10.1111/j.1369-1600.2012.00494.x
Sperling RE, Gomes SM, Sypek EI, Carey AN, McLaughlin JP. Endogenous kappa-opioid mediation of stress-induced potentiation of ethanol-conditioned place preference and self-administration. Psychopharmacol (Berl). 2010;210:199–209. https://doi.org/10.1007/s00213-010-1844-5
Krashes MJ, Shah BP, Madara JC, Olson DP, Strochlic DE, Garfield AS, et al. An excitatory paraventricular nucleus to AgRP neuron circuit that drives hunger. Nature . 2014;507:238–42. https://doi.org/10.1038/nature12956
Mitch CH, Quimby SJ, Diaz N, Pedregal C, de la Torre MG, Jimenez A, et al. Discovery of aminobenzyloxyarylamides as kappa opioid receptor selective antagonists: application to preclinical development of a kappa opioid receptor antagonist receptor occupancy tracer. J Med Chem. 2011;54:8000–12. https://doi.org/10.1021/jm200789r
Melief EJ, Miyatake M, Carroll FI, Beguin C, Carlezon WA Jr., Cohen BM, et al. Duration of action of a broad range of selective kappa-opioid receptor antagonists is positively correlated with c-Jun N-terminal kinase-1 activation. Mol Pharmacol. 2011;80:920–9. https://doi.org/10.1124/mol.111.074195
Armbruster BN, Li X, Pausch MH, Herlitze S, Roth BL. Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc Natl Acad Sci USA. 2007;104:5163–8. https://doi.org/10.1073/pnas.0700293104
Schank JR, Goldstein AL, Rowe KE, King CE, Marusich JA, Wiley JL, et al. The kappa opioid receptor antagonist JDTic attenuates alcohol seeking and withdrawal anxiety. Addict Biol. 2012;17:634–47. https://doi.org/10.1111/j.1369-1600.2012.00455.x
Domi E, Barbier E, Augier E, Augier G, Gehlert D, Barchiesi R, et al. Preclinical evaluation of the kappa-opioid receptor antagonist CERC-501 as a candidate therapeutic for alcohol use disorders. Neuropsychopharmacology. 2018;43:1805–12. https://doi.org/10.1038/s41386-018-0015-y
Mansour A, Fox CA, Meng F, Akil H, Watson SJ. Kappa 1 receptor mRNA distribution in the rat CNS: comparison to kappa receptor binding and prodynorphin mRNA. Mol Cell Neurosci. 1994;5:124–44. https://doi.org/10.1006/mcne.1994.1015
Marchant NJ, Densmore VS, Osborne PB. Coexpression of prodynorphin and corticotrophin-releasing hormone in the rat central amygdala: evidence of two distinct endogenous opioid systems in the lateral division. J Comp Neurol. 2007;504:702–15. https://doi.org/10.1002/cne.21464
Gilpin NW, Herman MA, Roberto M. The central amygdala as an integrative hub for anxiety and alcohol use disorders. Biol Psychiatry. 2015;77:859–69. https://doi.org/10.1016/j.biopsych.2014.09.008
Lam MP, Marinelli PW, Bai L, Gianoulakis C. Effects of acute ethanol on opioid peptide release in the central amygdala: an in vivo microdialysis study. Psychopharmacol (Berl). 2008;201:261–71. https://doi.org/10.1007/s00213-008-1267-8
Kissler JL, Walker BM. Dissociating motivational from physiological withdrawal in alcohol dependence: role of central amygdala kappa-opioid receptors. Neuropsychopharmacology . 2016;41:560–7. https://doi.org/10.1038/npp.2015.183
Gomez JL, Bonaventura J, Lesniak W, Mathews WB, Sysa-Shah P, Rodriguez LA, et al. Chemogenetics revealed: DREADD occupancy and activation via converted clozapine. Science . 2017;357:503–7. https://doi.org/10.1126/science.aan2475
Manvich DF, Webster KA, Foster SL, Farrell MS, Ritchie JC, Porter JH, et al. The DREADD agonist clozapine N-oxide (CNO) is reverse-metabolized to clozapine and produces clozapine-like interoceptive stimulus effects in rats and mice. Sci Rep. 2018;8:3840 https://doi.org/10.1038/s41598-018-22116-z
Kang-Park M, Kieffer BL, Roberts AJ, Siggins GR, Moore SD. kappa-Opioid receptors in the central amygdala regulate ethanol actions at presynaptic GABAergic sites. J Pharmacol Exp Ther. 2013;346:130–7. https://doi.org/10.1124/jpet.112.202903
Kang-Park M, Kieffer BL, Roberts AJ, Siggins GR, Moore SD. Interaction of CRF and kappa opioid systems on GABAergic neurotransmission in the mouse central amygdala. J Pharmacol Exp Ther. 2015;355:206–11. https://doi.org/10.1124/jpet.115.225870
Li C, Pleil KE, Stamatakis AM, Busan S, Vong L, Lowell BB, et al. Presynaptic inhibition of gamma-aminobutyric acid release in the bed nucleus of the stria terminalis by kappa opioid receptor signaling. Biol Psychiatry. 2012;71:725–32. https://doi.org/10.1016/j.biopsych.2011.11.015
Stamatakis AM, Sparta DR, Jennings JH, McElligott ZA, Decot H, Stuber GD. Amygdala and bed nucleus of the stria terminalis circuitry: implications for addiction-related behaviors. Neuropharmacology . 2014;76:320–8. https://doi.org/10.1016/j.neuropharm.2013.05.046
Roberto M, Cruz MT, Gilpin NW, Sabino V, Schweitzer P, Bajo M, et al. Corticotropin releasing factor-induced amygdala gamma-aminobutyric acid release plays a key role in alcohol dependence. Biol Psychiatry. 2010;67:831–9. https://doi.org/10.1016/j.biopsych.2009.11.007
Beczkowska IW, Bowen WD, Bodnar RJ. Central opioid receptor subtype antagonists differentially alter sucrose and deprivation-induced water intake in rats. Brain Res. 1992;589:291–301.
Morley JE, Levine AS. Involvement of dynorphin and the kappa opioid receptor in feeding. Peptides . 1983;4:797–800.
Chartoff EH, Mavrikaki M. Sex differences in kappa opioid receptor function and their potential impact on addiction. Front Neurosci. 2015;9:466 https://doi.org/10.3389/fnins.2015.00466
Robles CF, McMackin MZ, Campi KL, Doig IE, Takahashi EY, Pride MC, et al. Effects of kappa opioid receptors on conditioned place aversion and social interaction in males and females. Behav Brain Res. 2014;262:84–93. https://doi.org/10.1016/j.bbr.2014.01.003
Russell SE, Rachlin AB, Smith KL, Muschamp J, Berry L, Zhao Z, et al. Sex differences in sensitivity to the depressive-like effects of the kappa opioid receptor agonist U-50488 in rats. Biol Psychiatry. 2014;76:213–22. https://doi.org/10.1016/j.biopsych.2013.07.042
Van’t Veer A, Smith KL, Cohen BM, Carlezon WA Jr., Bechtholt AJ. Kappa-opioid receptors differentially regulate low and high levels of ethanol intake in female mice. Brain Behav. 2016;6:e00523 https://doi.org/10.1002/brb3.523
Zhou Y, Crowley RS, Ben K, Prisinzano TE, Kreek MJ. Synergistic blockade of alcohol escalation drinking in mice by a combination of novel kappa opioid receptor agonist Mesyl Salvinorin B and naltrexone. Brain Res. 2017;1662:75–86. https://doi.org/10.1016/j.brainres.2017.02.027
Acknowledgements
Funding
This work was supported by NIH grants P50 AA10716 (HCB), U01 AA014095 (HCB, MFL, WCG HLH), F32 AA023700 (RIA), T32 AA007474 (RIA), U01 AA020911 (TLK), U24 AA025475 (TLK), P60 AA011605 (TLK), and F31 AA025835 (DB), and VA grant BX000813 (HCB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Anderson, R.I., Lopez, M.F., Griffin, W.C. et al. Dynorphin-kappa opioid receptor activity in the central amygdala modulates binge-like alcohol drinking in mice. Neuropsychopharmacol. 44, 1084–1092 (2019). https://doi.org/10.1038/s41386-018-0294-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41386-018-0294-3
This article is cited by
-
The Dynorphin/-Opioid Receptor System at the Interface of Hyperalgesia/Hyperkatifeia and Addiction
Current Addiction Reports (2025)
-
Kappa-opioid receptor stimulation in the nucleus accumbens shell and ethanol drinking: Differential effects by rostro-caudal location and level of drinking
Neuropsychopharmacology (2024)
-
Behavioral and slice electrophysiological assessment of DREADD ligand, deschloroclozapine (DCZ) in rats
Scientific Reports (2022)
-
Kappa opioid receptor and dynorphin signaling in the central amygdala regulates alcohol intake
Molecular Psychiatry (2021)
-
Activation of hypothalamic oxytocin neurons reduces binge-like alcohol drinking through signaling at central oxytocin receptors
Neuropsychopharmacology (2021)