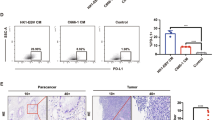
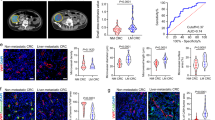
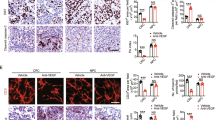
Abstract
Distant metastasis is one of the main reasons for treatment failure in nasopharyngeal carcinoma (NPC) patients. Tumour angiogenesis is a key basis for the distant metastasis of NPC. However, the molecular mechanisms underlying the mutual interaction between endothelial and NPC cells in tumour angiogenesis and NPC metastasis are still unclear. Here, we found that extracellular vesicles (EVs) mediate intercellular communication between endothelial cells and NPC cells, thereby promoting NPC cell migration, invasion, colony formation, and angiogenesis. Further experiments indicated that EV-mediated information exchange between endothelial cells and NPC cells upregulated the expression of the vascular endothelial growth factor receptor FLT1 in both types of cells. Mechanistically, FLT1-enriched EVs promoted NPC metastasis through the PI3K/AKT pathway and increased tumour angiogenesis, tumour growth, and distant lung and liver metastasis of NPC in xenografted mice. This effect was achieved through the delivery and upregulation of FLT1 in both endothelial and NPC cells. Thus, our findings reveal that FLT1-enriched EVs induce a positive feedback loop between NPC cells and endothelial cells to promote tumour angiogenesis and tumour metastasis. These results increase our understanding of the intricate interplay between tumour angiogenesis and distant metastasis and have major implications for the diagnosis and management of NPC patients with increased levels of FLT1-enriched EVs.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Sang Y, Cheng C, Zeng YX, Kang TB. Snail promotes metastasis of nasopharyngeal carcinoma partly by down-regulating TEL2. Cancer Commun. 2018;38:58.
Hu Q, Lin X, Ding L, Zeng Y, Pang D, Ouyang N, et al. ARHGAP42 promotes cell migration and invasion involving PI3K/Akt signaling pathway in nasopharyngeal carcinoma. Cancer Med. 2018;7:3862–74.
Liu X, Tang LL, Du XJ, Li WF, Chen L, Zhou GQ, et al. Changes in disease failure risk of nasopharyngeal carcinoma over time: analysis of 749 patients with long-term follow-up. J Cancer. 2017;8:455–9.
Yao JJ, Yu XL, Zhang F, Zhang WJ, Zhou GQ, Tang LL, et al. Radiotherapy with neoadjuvant chemotherapy versus concurrent chemoradiotherapy for ascending-type nasopharyngeal carcinoma: a retrospective comparison of toxicity and prognosis. Chin J Cancer. 2017;36:26.
Arifin DR, Witwer KW, Bulte JWM. Non-Invasive imaging of extracellular vesicles: Quo vaditis in vivo?. J Extracell Vesicles. 2022;11:12241.
Yang YC, Jiang Q, Yang KP, Wang LZ, Sethi G, Ma ZW. Extracellular vesicle-mediated ferroptosis, pyroptosis, and necroptosis: potential clinical applications in cancer therapy. Cell Death Discov. 2024;10:23.
Zomer A, Maynard C, Verweij FJ, Kamermans A, Schäfer R, Beerling E, et al. In vivo imaging reveals extracellular vesicle-mediated phenocopying of metastatic behavior. Cell. 2015;161:1046–57.
Samuel M, Fonseka P, Sanwlani R, Gangoda L, Chee SH, Keerthikumar S, et al. Oral administration of bovine milk-derived extracellular vesicles induces senescence in the primary tumor but accelerates cancer metastasis. Nat Commun. 2021;12:3950.
Zhao C, Chen JY, Peng WM, Yuan B, Bi Q, Xu YJ. Exosomes from adipose-derived stem cells promote chondrogenesis and suppress inflammation by upregulating miR-145 and miR-221. Mol Med Rep. 2020;21:1881–9.
Rak J. Cancer organ-seeking vesicles. Nature. 2015;527:312–4.
Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527:329–35.
Shan YH, Zhou PJ, Zhou Q, Yang LF. Extracellular vesicles in the progression and therapeutic resistance of nasopharyngeal carcinoma. Cancers. 2022;14:2289.
Mu W, Rana S, Zöller M. Host matrix modulation by tumor exosomes promotes motility and invasiveness. Neoplasia. 2013;15:875–87.
Chen QY, Gao BB, Tong DD, Huang C. Crosstalk between extracellular vesicles and tumor-associated macrophage in the tumor microenvironment. Cancer Lett. 2023;552:215979.
Gao J, Zhang XD, Jiang L, Li Y, Zheng QQ. Tumor endothelial cell-derived extracellular vesicles contribute to tumor microenvironment remodeling. Cell Commun Signal. 2022;20:97.
Shi S, Zhang Q, Xia Y, You B, Shan Y, Bao L, et al. Mesenchymal stem cell-derived exosomes facilitate nasopharyngeal carcinoma progression. Am J Cancer Res. 2016;6:459–72.
Zeng Y, Yao X, Liu X, He X, Li L, Liu X, et al. Anti-angiogenesis triggers exosomes release from endothelial cells to promote tumor vasculogenesis. J Extracell Vesicles. 2019;8:1629865.
Zhang KW, Liu D, Zhao JM, Shi S, He X, Da P et al. Nuclear exosome HMGB3 secreted by nasopharyngeal carcinoma cells promotes tumour metastasis by inducing angiogenesis. Cell Death Dis. 2021;12:554.
Suzuki K, Chosa N, Sawada S, Takizawa N, Yaegashi T, Ishisaki A. Enhancement of anti-inflammatory and osteogenic abilities of mesenchymal stem cells via cell-to-cell adhesion to periodontal ligament-derived fibroblasts. Stem Cells Int. 2017;2017:3296498.
Tsumita T, Maishi N, Annan DA, Towfik MA, Matsuda A, Onodera Y, et al. The oxidized-LDL/LOX-1 axis in tumor endothelial cells enhances metastasis by recruiting neutrophils and cancer cells. Int J Cancer. 2022;151:944–56.
Li F, Zhao X, Sun R, Ou J, Huang J, Yang N, et al. EGFR-rich extracellular vesicles derived from highly metastatic nasopharyngeal carcinoma cells accelerate tumour metastasis through PI3K/AKT pathway-suppressed ROS. J Extracell Vesicles. 2020;10:12003.
Théry C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7:1535750.
Witwer KW, Goberdhan DC, O'driscoll L, Théry C, Welsh JA, Blenkiron C, et al. Updating MISEV: Evolving the minimal requirements for studies of extracellular vesicles. J Extracell Vesicles. 2021;10:12182.
Menon R, Dixon CL, Sheller-Miller S, Fortunato SJ, Saade GR, Palma C, et al. Quantitative proteomics by SWATH-MS of maternal plasma exosomes determine pathways associated with term and preterm birth. Endocrinology. 2019;160:639–50.
Zhang DX, Dang XTT, Vu LT, Lim CMH, Yeo EYM, Lam BWS, et al. αvβ1 integrin is enriched in extracellular vesicles of metastatic breast cancer cells: a mechanism mediated by galectin-3. J Extracell Vesicles. 2022;11:e12234.
Li F, Xu T, Chen P, Sun R, Li C, Zhao X, et al. Platelet-derived extracellular vesicles inhibit ferroptosis and promote distant metastasis of nasopharyngeal carcinoma by upregulating ITGB3. Int J Biol Sci. 2022;18:5858–72.
Golfmann K, Meder L, Koker M, Volz C, Borchmann S, Tharun L, et al. Synergistic anti-angiogenic treatment effects by dual FGFR1 and VEGFR1 inhibition in FGFR1-amplified breast cancer. Oncogene. 2018;37:5682–93.
Li F, Xiong Y, Yang M, Chen P, Zhang J, Wang Q, et al. c-Mpl-del, a c-Mpl alternative splicing isoform, promotes AMKL progression and chemoresistance. Cell Death Dis. 2022;13:869.
Xie W, Wang J, Tian S, Zhao H, Cao L, Liang Z, et al. RNF126-mediated ubiquitination of FSP1 affects its subcellular localization and ferroptosis. Oncogene. 2024;43:1463–75.
Jaffe IZ, Newfell BG, Aronovitz M, Mohammad NN, McGraw AP, Perreault RE, et al. Placental growth factor mediates aldosterone-dependent vascular injury in mice. J Clin Investig. 2010;120:3891–3900.
Li F, Li J, Wang PH, Yang N, Huang J, Ou J, et al. SARS-CoV-2 spike promotes inflammation and apoptosis through autophagy by ROS-suppressed PI3K/AKT/mTOR signaling. BBA Mol Basis Dis. 2021;1867:166260.
Oggero S, de Gaetano M, Marcone S, Fitzsimons S, Pinto AL, Ikramova D, et al. Extracellular vesicles from monocyte/platelet aggregates modulate human atherosclerotic plaque reactivity. J Extracell Vesicles. 2021;10:12084.
Hsu MT, Wang YK, Tseng YJ. Exosomal proteins and lipids as potential biomarkers for lung cancer diagnosis, prognosis, and treatment. Cancers. 2022;14:732.
Georgievski A, Michel A, Thomas C, Mlamla Z, de Barros JPP, Pais de Barros JP, Lemaire-Ewing S, et al. Acute lymphoblastic leukemia-derived extracellular vesicles affect quiescence of hematopoietic stem and progenitor cells. Cell Death Dis. 2022;13:337.
Bi Y, Guo S, Xu X, Kong P, Cui H, Yan T, et al. Decreased ZNF750 promotes angiogenesis in a paracrine manner via activating DANCR/miR-4707-3p/FOXC2 axis in esophageal squamous cell carcinoma. Cell Death Dis. 2020;11:296.
Chen XW, Yu TJ, Zhang J, Li Y, Chen HL, Yang GF, et al. CYP4A in tumor-associated macrophages promotes pre-metastatic niche formation and metastasis. Oncogene. 2017;36:5045–57.
Zhou Z, Zhao C, Wang L, Cao X, Li J, Huang R, et al. A VEGFR1 antagonistic peptide inhibits tumor growth and metastasis through VEGFR1-PI3K-AKT signaling pathway inhibition. Am J Cancer Res. 2015;5:3149–61.
Sadremomtaz A, Mansouri K, Alemzadeh G, Safa M, Rastaghi AE, Asghari SM. Dual blockade of VEGFR1 and VEGFR2 by a novel peptide abrogates VEGF-driven angiogenesis, tumor growth, and metastasis through PI3K/AKT and MAPK/ERK1/2 pathway. BBA Gen Subj. 2018;1862:2688–2700.
Ahluwalia A, Matysiak-Budnik T, Yau A, Tarnawski AS. Novel mechanisms for aberrant expression of VEGF and its receptor in colon cancer cells: upregulation of importins and increased nuclear transport of P-CREB, P-STAT3 and P-MAPK/ERK1/2. Gastroenterology. 2012;142:S165–S165.
Tu L, Liu Z, He X, He Y, Yang H, Jiang Q, et al. Over-expression of eukaryotic translation initiation factor 4 gamma 1 correlates with tumor progression and poor prognosis in nasopharyngeal carcinoma. Mol Cancer. 2010;9:78.
Lin C, Li MF, Lin N, Zong JF, Pan JJ, Ye YB. RNF38 suppress growth and metastasis via ubiquitination of ACTN4 in nasopharyngeal carcinoma. BMC Cancer. 2022;22:549.
Raikwar SP, Gardner TA, Kao CHH. Antiangiogenic gene therapy targeting the endothelium-specific Flk-1 and Tie-2: Inhibition of tumor angiogenesis and growth in prostate cancer. Mol Ther. 2003;7:S132–S133.
Shen Y, Li S, Wang X, Wang M, Tian Q, Yang J, et al. Tumor vasculature remolding by thalidomide increases delivery and efficacy of cisplatin. J Exp Clin Cancer Res. 2019;38:427.
Barreiro K, Lay AC, Leparc G, Tran V, Rosler M, Dayalan L, et al. An in vitro approach to understand contribution of kidney cells to human urinary extracellular vesicles. J Extracell Vesicles. 2023;12:12304.
Rahimi N. VEGFR-1 and VEGFR-2: two non-identical twins with a unique physiognomy. Front Biosci Landmrk. 2006;11:818–29.
Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9:669–76.
Tomanek RJ, Ishii Y, Holifield JS, Sjogren CL, Hansen HK, Mikawa T. VEGF family members regulate myocardial tubulogenesis and coronary artery formation in the embryo. Circ Res. 2006;98:947–53.
Lukason M, DuFresne E, Rubin H, Pechan P, Li Q, Kim I, et al. Inhibition of choroidal neovascularization in a nonhuman primate model by intravitreal administration of an AAV2 vector expressing a novel anti-VEGF molecule. Mol Ther. 2011;19:260–5.
Owen LA, Morrison MA, Ahn J, Woo SJ, Sato H, Robinson R, et al. FLT1 genetic variation predisposes to neovascular AMD in ethnically diverse populations and alters systemic FLT1 expression. Invest Ophthamol Vis Sci. 2014;55:3543–54.
Orecchia A, Mettouchi A, Uva P, Simon GC, Arcelli D, Avitabile S, et al. Endothelial cell adhesion to soluble vascular endothelial growth factor receptor-1 triggers a cell dynamic and angiogenic phenotype. FASEB J. 2014;28:692–704.
Verma M, Shimizu-Motohashi Y, Asakura Y, Ennen JP, Bosco J, Zhou Z, et al. Inhibition of FLT1 ameliorates muscular dystrophy phenotype by increased vasculature in a mouse model of Duchenne muscular dystrophy. PLoS Genet. 2019;15:e1008468.
He QL, Ye AH, Ye WBA, Liao XM, Qin GQ, Xu YQ et al. Cancer-secreted exosomal miR-21-5p induces angiogenesis and vascular permeability by targeting KRIT1. Cell Death Dis. 2021;12:576.
Lv J, Chen Y, Zhou G, Qi Z, Tan K, Wang H, et al. Liquid biopsy tracking during sequential chemo-radiotherapy identifies distinct prognostic phenotypes in nasopharyngeal carcinoma. Nat Commun. 2019;10:3941.
Kung CP, Raab-Traub N. Epstein-Barr virus latent membrane protein 1 induces expression of the epidermal growth factor receptor through effects on Bcl-3 and STAT3. J Virol. 2008;82:5486–93.
Acknowledgements
The schematic representation and mechanistic scheme of this study were drawn using Figdraw (https://www.figdraw.com).
Funding
This work was supported by National Natural Science Foundation of China (31771273, 82273467, and 82403550), China Postdoctoral Science Foundation (2024T171077), Guangdong Province Basic and Applied Basic Research Fund (2022A1515140100), Guangdong Province of China Grant (2021A1515011209), Research Funds for the Shenzhen of China (JCYJ20210324120812035), and the Fundamental Research Funds for the Central Universities, Sun Yat-sen University (24qnpy180).
Author information
Authors and Affiliations
Contributions
FL planned and carried out experiments, analysed data, and wrote the manuscript. LS, YH, PC, JW, MZ, CL, JC, HC, QG, JF, XH, and QW performed the research and analysed data. QZ supervised the research, planned experiments, analysed data, and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All clinical samples were obtained with informed consent from the Sun Yat-sen University Cancer Center and approved by the Hospital’s Ethical Review Committees. The animal studies were authorized by the Institutional Animal Care and Use Committee of the Sun Yat-sen University (approval number: SYSU-LS-IACUC-2023-0087). All animal experiments were strictly implemented in compliance with the ARRIVE guidelines.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, F., Song, L., He, Y. et al. FLT1-enriched extracellular vesicles induce a positive feedback loop between nasopharyngeal carcinoma cells and endothelial cells to promote angiogenesis and tumour metastasis. Oncogene 44, 2253–2267 (2025). https://doi.org/10.1038/s41388-025-03389-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41388-025-03389-x