Abstract
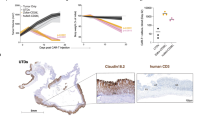
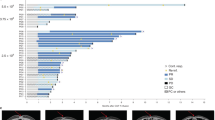
Claudin18 isoform 2 (CLDN18.2), primarily expressed in gastric tissue and upregulated in pancreatic cancer (PC), is a key target for innovative treatments like chimeric antigen receptor T (CAR-T) cell therapy. However, CAR-T’s effectiveness comes with a significant risk of on-target, off-tumor (OTOT) toxicity due to CLDN18.2’s presence in normal gastric mucosa. To address this, we developed CLDN18.2-specific synthetic T cell receptor and antigen receptor T (STAR-T) cells. Our research shows that STAR-T and CAR-T cells have comparable in vitro cytotoxicity, but STAR-T cells cause less gastric damage in vivo despite having weaker antitumor effects than CAR-T cells. Clinical tests with gastroscopes confirmed the gastric safety of STAR-T cell therapy, which effectively controlled the disease. Additionally, incorporating the IL12β p40 subunit into STAR-T cells enhanced their function in both lab and animal studies. This evidence suggests that CLDN18.2 STAR-T cell could be a safer alternative to CAR-T cell therapy for PC, meriting further clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author, L.Y.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209–49.
Klein AP. Pancreatic cancer epidemiology: understanding the role of lifestyle and inherited risk factors. Nat Rev Gastroenterol Hepatol. 2021;18:493–502.
Marin JJG, Perez-Silva L, Macias RIR, Asensio M, Peleteiro-Vigil A, Sanchez-Martin A, et al. Molecular Bases of Mechanisms Accounting for Drug Resistance in Gastric Adenocarcinoma. Cancers. 2020;12:2116.
Brown CE, Alizadeh D, Starr R, Weng L, Wagner JR, Naranjo A, et al. Regression of Glioblastoma after Chimeric Antigen Receptor T-Cell Therapy. N Engl J Med. 2016;375:2561–9.
Majzner RG, Ramakrishna S, Yeom KW, Patel S, Chinnasamy H, Schultz LM, et al. GD2-CAR T cell therapy for H3K27M-mutated diffuse midline gliomas. Nature. 2022;603:934–41.
Qi C, Liu C, Gong J, Liu D, Wang X, Zhang P, et al. Claudin18.2-specific CAR T cells in gastrointestinal cancers: phase 1 trial final results. Nat Med. 2024;30:2224–34.
Qi C, Zhang P, Liu C, Zhang J, Zhou J, Yuan J, et al. Safety and Efficacy of CT041 in Patients With Refractory Metastatic Pancreatic Cancer: A Pooled Analysis of Two Early-Phase Trials. J Clin Oncol. 2024;42:2565–77.
Del Bufalo F, De Angelis B, Caruana I, Del Baldo G, De Ioris MA, Serra A, et al. GD2-CART01 for Relapsed or Refractory High-Risk Neuroblastoma. N Engl J Med. 2023;388:1284–95.
Zhu G, Foletti D, Liu X, Ding S, Melton Witt J, Hasa-Moreno A, et al. Targeting CLDN18.2 by CD3 Bispecific and ADC Modalities for the Treatments of Gastric and Pancreatic Cancer. Sci Rep. 2019;9:8420.
Xu G, Liu W, Wang Y, Wei X, Liu F, He Y, et al. CMG901, a Claudin18.2-specific antibody-drug conjugate, for the treatment of solid tumors. Cell Rep Med. 2024;5:101710.
Zhou KI, Strickler JH, Chen H. Targeting Claudin-18.2 for cancer therapy: updates from 2024 ASCO annual meeting. J Hematol Oncol. 2024;17:73.
Qi C, Xie T, Zhou J, Wang X, Gong J, Zhang X, et al. CT041 CAR T cell therapy for Claudin18.2-positive metastatic pancreatic cancer. J Hematol Oncol. 2023;16:102.
Chen T, Wang M, Chen Y, Liu Y. Current challenges and therapeutic advances of CAR-T cell therapy for solid tumors. Cancer Cell Int. 2024;24:133.
Wu K, Yang S, Shi Z, Wang H, Li X, He X. 531P Enhanced antitumor efficacy and safety through a novel CAR-T strategy targeting MSLN and CLDN18.2. Annals Oncol. 2024;35:S211–S.
Wang X, Martin AD, Negri KR, McElvain ME, Oh J, Wu ML, et al. Extensive functional comparisons between chimeric antigen receptors and T cell receptors highlight fundamental similarities. Mol Immunol. 2021;138:137–49.
Liu Y, Liu G, Wang J, Zheng ZY, Jia L, Rui W, et al. Chimeric STAR receptors using TCR machinery mediate robust responses against solid tumors. Sci Transl Med. 2021;13:eabb5191.
Wang J, Zhang X, Zhou Z, Liu Y, Yu L, Jia L, et al. A novel adoptive synthetic TCR and antigen receptor (STAR) T-Cell therapy for B-Cell acute lymphoblastic leukemia. Am J Hematol. 2022;97:992–1004.
Mog BJ, Marcou N, DiNapoli SR, Pearlman AH, Nichakawade TD, Hwang MS, et al. Preclinical studies show that Co-STARs combine the advantages of chimeric antigen and T cell receptors for the treatment of tumors with low antigen densities. Sci Transl Med. 2024;16:eadg7123.
Burton J, Siller-Farfan JA, Pettmann J, Salzer B, Kutuzov M, van der Merwe PA, et al. Inefficient exploitation of accessory receptors reduces the sensitivity of chimeric antigen receptors. Proc Natl Acad Sci USA. 2023;120:e2216352120.
Gong L, Sun H, Liu L, Sun X, Fang T, Yu Z, et al. LILRB4 represents a promising target for immunotherapy by dual targeting tumor cells and myeloid-derived suppressive cells in multiple myeloma. Haematologica. 2024;109:3650–69.
Zhong G, Zhang X, Guo Z, Gao Y, Zhao B, Liu X, et al. Complete remission of advanced pancreatic cancer induced by claudin18.2-targeted CAR-T cell therapy: a case report. Front Immunol. 2024;15:1325860.
Qi C, Gong J, Li J, Liu D, Qin Y, Ge S, et al. Claudin18.2-specific CAR T cells in gastrointestinal cancers: phase 1 trial interim results. Nat Med. 2022;28:1189–98.
Ma X, Shou P, Smith C, Chen Y, Du H, Sun C, et al. Interleukin-23 engineering improves CAR T cell function in solid tumors. Nat Biotechnol. 2020;38:448–59.
Lukaszewicz-Zajac M, Mroczko B. Claudins-Promising Biomarkers for Selected Gastrointestinal (GI) Malignancies? Cancers. 2023;16:152.
Hana C, Thaw Dar NN, Galo Venegas M, Vulfovich M. Claudins in Cancer: A Current and Future Therapeutic Target. Int J Mol Sci. 2024;25:4634.
Waters R, Sewastjanow-Silva M, Yamashita K, Abdelhakeem A, Iwata KK, Moran D, et al. Retrospective Study of Claudin 18 Isoform 2 Prevalence and Prognostic Association in Gastric and Gastroesophageal Junction Adenocarcinoma. JCO Precis Oncol. 2024;8:e2300543.
Ogawa H, Abe H, Yagi K, Seto Y, Ushiku T. Claudin-18 status and its correlation with HER2 and PD-L1 expression in gastric cancer with peritoneal dissemination. Gastric Cancer. 2024;27:802–10.
Jiang H, Shi Z, Wang P, Wang C, Yang L, Du G, et al. Claudin18.2-Specific Chimeric Antigen Receptor Engineered T Cells for the Treatment of Gastric Cancer. J Natl Cancer Inst. 2019;111:409–18.
Nakayama I, Qi C, Chen Y, Nakamura Y, Shen L, Shitara K. Claudin 18.2 as a novel therapeutic target. Nat Rev Clin Oncol. 2024;21:354–69.
Shah MA, Shitara K, Ajani JA, Bang YJ, Enzinger P, Ilson D, et al. Zolbetuximab plus CAPOX in CLDN18.2-positive gastric or gastroesophageal junction adenocarcinoma: the randomized, phase 3 GLOW trial. Nat Med. 2023;29:2133–41.
Kuwana Y, Asakura Y, Utsunomiya N, Nakanishi M, Arata Y, Itoh S, et al. Expression of chimeric receptor composed of immunoglobulin-derived V regions and T-cell receptor-derived C regions. Biochem Biophys Res Commun. 1987;149:960–8.
Gross G, Waks T, Eshhar Z. Expression of immunoglobulin-T-cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci USA. 1989;86:10024–8.
Akatsuka Y. TCR-Like CAR-T Cells Targeting MHC-Bound Minor Histocompatibility Antigens. Front Immunol. 2020;11:257.
Weigelin B, den Boer AT, Wagena E, Broen K, Dolstra H, de Boer RJ, et al. Cytotoxic T cells are able to efficiently eliminate cancer cells by additive cytotoxicity. Nat Commun. 2021;12:5217.
Mao R, Hussein MS, He Y. Chimeric antigen receptor engineered T cells and their application in the immunotherapy of solid tumours. Expert Rev Mol Med. 2022;24:e7.
Labanieh L, Mackall CL. CAR immune cells: design principles, resistance and the next generation. Nature. 2023;614:635–48.
Nie S, Song Y, Hu K, Zu W, Zhang F, Chen L, et al. CXCL10 and IL15 co-expressing chimeric antigen receptor T cells enhance anti-tumor effects in gastric cancer by increasing cytotoxic effector cell accumulation and survival. Oncoimmunology. 2024;13:2358590.
Huang Y, Wang H. From suppressor to enhancer: IL-10’s alternative role in CAR-T cell therapies against solid tumors. Cell Stem Cell. 2024;31:285–7.
Noviello D, Mager R, Roda G, Borroni RG, Fiorino G, Vetrano S. The IL23-IL17 Immune Axis in the Treatment of Ulcerative Colitis: Successes, Defeats, and Ongoing Challenges. Front Immunol. 2021;12:611256.
Lah S, Kim S, Kang I, Kim H, Hupperetz C, Jung H, et al. Engineering second-generation TCR-T cells by site-specific integration of TRAF-binding motifs into the CD247 locus. J Immunother Cancer. 2023;11:e005519.
Brazin KN, Mallis RJ, Boeszoermenyi A, Feng Y, Yoshizawa A, Reche PA, et al. The T Cell Antigen Receptor alpha Transmembrane Domain Coordinates Triggering through Regulation of Bilayer Immersion and CD3 Subunit Associations. Immunity. 2018;49:829–41.e6.
Funding
This study was supported by National Natural Science Foundation of China (82030076, 82470229), Shenzhen Clinical Research Center of Hematology (LCYSSQ20220823091401002), Sanming Project of Medicine in Shenzhen (SZSM202111004), Shenzhen Key Laboratory Foundation (ZDSYS20200811143757022), Medicine Plus Program of Shenzhen University (000003011601).
Author information
Authors and Affiliations
Contributions
WZ and YL conceived and designed the experiments. WZ, MZ and XM performed the experimental work. WZ and MZ wrote the manuscript. WZ, JC and YL revised the manuscript. GZ and LY performed the clinical research. JQ and ZH performed the clinical samples analysis. WZ, MZ, XM and JC contributed to the analysis of experimental data. YL and LY supervised the experiments. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This clinical study was proved by the Ethics Committee of Shenzhen University General Hospital with ethics code KYLLHS-20240104A and conducted according to Helsinki Declaration’s principles. Written informed consent was obtained from each participant before specimen collection. All animal experiments were performed according to protocols approved by the Institutional Animal Care and Use Committee (IACUC) of Shenzhen University (SYXK(粤)2022-0302), the ethics approval code is IACUC-202400138.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, W., Zeng, M., Ma, X. et al. CLDN18.2-targeting STAR-T cell therapy for pancreatic cancer: a strategy to minimize gastric off-tumor toxicity compared to CLDN18.2 CAR-T. Oncogene 44, 2440–2452 (2025). https://doi.org/10.1038/s41388-025-03414-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41388-025-03414-z
This article is cited by
-
Synergistic Effect of Fecal Microbiota Transplantation, γδT Cell Immunotherapy, and Pembrolizumab in Refractory Advanced Pancreatic Cancer: A Case Report
Journal of Gastrointestinal Cancer (2026)