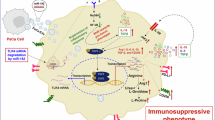
Abstract
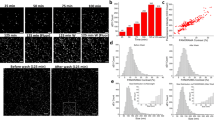
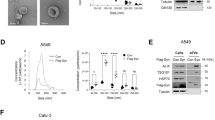
High-risk pancreatic neuroendocrine tumors (pNET) are characterized by aggressive progression and metastasis, contributing to poor clinical outcomes. This study explores molecular mechanisms underlying metastasis in high-risk pNETs and evaluates therapeutic interventions. We employed small RNA sequencing to profile miRNA signatures in tumor-derived small extracellular vesicles (sEVs) and integrated single-cell transcriptomic analysis to delineate the tumor immune microenvironment. Prognostic validation of SPP1+ macrophages was performed in clinical cohorts using multiplex immunofluorescence. Mechanistic studies utilized animal models and functional assays to elucidate the sEV-mediated crosstalk between tumor cells and macrophages. Here, we reveal that sEV-miR-183-5p from high-risk pNETs reprograms macrophages via PDCD4/PI3Kγ/AKT/mTOR signaling, inducing SPP1 overexpression and M2 polarization to promote angiogenesis and metastasis. p53 mutants were identified as upstream regulators of sEV-miR-183-5p secretion. Inhibition of SPP1 attenuated tumor progression, while elevated plasma sEV-miR-183-5p levels correlated with high-grade and advanced disease stages. Together, this study reveals a critical sEV-miRNA mediated mechanism governing tumor-macrophage communication in high-risk pNETs, offering insights into therapeutic strategies and suggesting sEV-miR-183-5p may have prognostic potential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Raw data for the scRNA-seq has been deposited in Gene Expression Omnibus (GEO) (GSE256136), which are publicly available. The raw data generated by small RNA sequencing of sEVs, ATAC-seq, and RNA sequencing are deposited in Genome Sequence Archive (GSA) database (https://ngdc.cncb.ac.cn/gsa/) and can be access by searching for bioproject PRJCA024087. The raw data generated by whole exome sequencing can be access by searching for bioproject PRJCA026652. Access to the raw data may be requested by completing the application form via GSA-Human System and is granted by the corresponding Data Access Committee. The approximate response time for accession requests is about 10 working days. Additional guidance can be found at the GSA-Human website [https://ngdc.cncb.ac.cn/gsa-human/document/GSAHuman_Request_Guide_for_Users_us.pdf]. This study did not generate original codes. All software and algorithms used in this study are publicly available.
References
Dasari A, Shen C, Halperin D, Zhao B, Zhou S, Xu Y, et al. Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol. 2017;3:1335–42.
Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182–8.
Rindi G, Mete O, Uccella S, Basturk O, La Rosa S, Brosens LAA, et al. Overview of the 2022 WHO classification of neuroendocrine neoplasms. Endocr Pathol. 2022;33:115–54.
Zaidi MY, Lopez-Aguiar AG, Switchenko JM, Lipscomb J, Andreasi V, Partelli S, et al. A novel validated recurrence risk score to guide a pragmatic surveillance strategy after resection of pancreatic neuroendocrine tumors an international study of 1006 patients. Annals Surg. 2019;270:422–33.
Genç CG, Jilesen AP, Partelli S, Falconi M, Muffatti F, van Kemenade FJ, et al. A new scoring system to predict recurrent disease in grade 1 and 2 nonfunctional pancreatic neuroendocrine tumors. Ann Surg. 2018;267:1148–54.
Soares HP, Guthrie KA, Ahmad SA, Washington MK, Ramnaraign BH, Raj NP. et al. Randomized phase II trial of postoperative adjuvant capecitabine and temozolomide versus observation in high-risk pancreatic neuroendocrine tumors: SWOG S2104. J Clin Oncol. 2022;40:TPS515–TPS5152022.
Fischer L, Bergmann F, Schimmack S, Hinz U, Prieß S, Müller-Stich BP, et al. Outcome of surgery for pancreatic neuroendocrine neoplasms. Br J Surg. 2014;101:1405–12.
Scott AT, Howe JR. Evaluation and management of neuroendocrine tumors of the pancreas. Surg Clin N Am. 2019;99:793.
Wong KP, Tsang JS, Lang BH-H. Role of surgery in pancreatic neuroendocrine tumor. Gland Surg. 2018;7:36–41.
Pavel M, Baudin E, Couvelard A, Krenning E, Öberg K, Steinmüller T, et al. ENETS Consensus Guidelines for the management of patients with liver and other distant metastases from neuroendocrine neoplasms of foregut, midgut, hindgut, and unknown primary. Neuroendocrinology. 2012;95:157–76.
Wang WQ, Zhang WH, Gao HL, Huang D, Xu HX, Li S, et al. A novel risk factor panel predicts early recurrence in resected pancreatic neuroendocrine tumors. J Gastroenterol. 2021;56:395–405.
Gao H, Liu L, Wang W, Xu H, Jin K, Wu C, et al. Novel recurrence risk stratification of resected pancreatic neuroendocrine tumor. Cancer Lett. 2018;412:188–93.
Pulvirenti A, Javed AA, Michelakos T, Sekigami Y, Zheng J, Kalvin HL, et al. Recurring pancreatic neuroendocrine tumor: timing and pattern of recurrence and current treatment. Ann Surg. 2023;278:e1063–e1067.
Rinke A, Muller HH, Schade-Brittinger C, Klose KJ, Barth P, Wied M, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009;27:4656–63.
Caplin ME, Pavel M, Cwikla JB, Phan AT, Raderer M, Sedlackova E, et al. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014;371:224–33.
Faivre S, Niccoli P, Castellano D, Valle JW, Hammel P, Raoul JL, et al. Sunitinib in pancreatic neuroendocrine tumors: updated progression-free survival and final overall survival from a phase III randomized study. Ann Oncol. 2017;28:339–43.
Xu J, Shen L, Bai C, Wang W, Li J, Yu X, et al. Surufatinib in advanced pancreatic neuroendocrine tumours (SANET-p): a randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 2020;21:1489–99.
Yao JC, Shah MH, Ito T, Bohas CL, Wolin EM, Van Cutsem E, et al. Everolimus for advanced pancreatic neuroendocrine tumors. N Engl J Med. 2011;364:514–23.
Kunz PL, Graham NT, Catalano PJ, Nimeiri HS, Fisher GA, Longacre TA, et al. Randomized study of temozolomide or temozolomide and capecitabine in patients with advanced pancreatic neuroendocrine tumors (ECOG-ACRIN E2211). J Clin Oncol. 2023;41:1359–69.
Yao JC, Pavel M, Lombard-Bohas C, Van Cutsem E, Voi M, Brandt U, et al. Everolimus for the treatment of advanced pancreatic neuroendocrine tumors: overall survival and circulating biomarkers from the randomized, phase III RADIANT-3 study. J Clin Oncol. 2016;34:3906–13.
Rinke A, Wittenberg M, Schade-Brittinger C, Aminossadati B, Ronicke E, Gress TM, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors (PROMID): results of long-term survival. Neuroendocrinology. 2017;104:26–32.
Isaac R, Reis FCG, Ying W, Olefsky JM. Exosomes as mediators of intercellular crosstalk in metabolism. Cell Metab. 2021;33:1744–62.
Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367:eaau6977
Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19:1423–37.
Zhang WH, Wang WQ, Gao HL, Yu XJ, Liu L. The tumor immune microenvironment in gastroenteropancreatic neuroendocrine neoplasms. Biochim Biophys Acta Rev Cancer. 2019;1872:188311.
Ma RY, Black A, Qian BZ. Macrophage diversity in cancer revisited in the era of single-cell omics. Trends Immunol. 2022;43:546–63.
Chen P, Zhao D, Li J, Liang X, Li J, Chang A, et al. Symbiotic macrophage-glioma cell interactions reveal synthetic lethality in PTEN-Null glioma. Cancer Cell. 2019;35:868.
Ahmed M, Sottnik JL, Dancik GM, Sahu D, Hansel DE, Theodorescu D, et al. An Osteopontin/CD44 axis in RhoGDI2-mediated metastasis suppression. Cancer Cell. 2016;30:432–43.
Liu Y, Zhang Q, Xing B, Luo N, Gao R, Yu K, et al. Immune phenotypic linkage between colorectal cancer and liver metastasis. Cancer Cell. 2022;40:424.
Nallasamy P, Nimmakayala RK, Karmakar S, Leon F, Seshacharyulu P, Lakshmanan I, et al. Pancreatic tumor microenvironment factor promotes cancer stemness via SPP1-CD44 axis. Gastroenterology. 2021;161:1998–2013.e1997.
Zhang WH, Wang WQ, Gao HL, Xu SS, Li S, Li TJ, et al. Tumor-infiltrating neutrophils predict poor survival of non-functional pancreatic neuroendocrine tumor. J Clin Endocrinol Metab. 2020;105:dgaa196.
Becker A, Thakur BK, Weiss JM, Kim HS, Peinado H, Lyden D. Extracellular vesicles in cancer: cell-to-cell mediators of metastasis. Cancer Cell. 2016;30:836–48.
Yang C, Dou R, Wei C, Liu K, Shi D, Zhang C, et al. Tumor-derived exosomal microRNA-106b-5p activates EMT-cancer cell and M2-subtype TAM interaction to facilitate CRC metastasis. Mol Ther. 2021;29:2088–107.
Lin F, Yin H-B, Li X-Y, Zhu G-M, He W-Y, Gou X. Bladder cancer cell-secreted exosomal miR-21 activates the PI3K/AKT pathway in macrophages to promote cancer progression. Int J Oncol. 2020;56:151–64.
Zhao S, Mi Y, Guan B, Zheng B, Wei P, Gu Y. et al. Tumor-derived exosomal miR-934 induces macrophage M2 polarization to promote liver metastasis of colorectal cancer. J Hematol Oncol. 2020;13:156–73.
Lu T, Zhang Z, Zhang J, Pan X, Zhu X, Wang X, et al. CD73 in small extracellular vesicles derived from HNSCC defines tumour-associated immunosuppression mediated by macrophages in the microenvironment. J Extracell Vesicles. 2022;11:e12218.
Crescitelli R, Lässer C, Lötvall J. Isolation and characterization of extracellular vesicle subpopulations from tissues. Nat Protoc. 2021;16:1548–80.
Lou X, Ye Z, Xu X, Jiang M, Lu R, Jing D, et al. Establishment and characterization of the third non-functional human pancreatic neuroendocrine tumor cell line. Hum Cell. 2022;35:1248–61.
Théry C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7:1535750.
Han K, Wang F-W, Cao C-H, Ling H, Chen J-W, Chen R-X, et al. CircLONP2 enhances colorectal carcinoma invasion and metastasis through modulating the maturation and exosomal dissemination of microRNA-17. Mol Cancer. 2020;19:60–77.
Yang B, Feng X, Liu H, Tong R, Wu J, Li C, et al. High-metastatic cancer cells derived exosomal miR92a-3p promotes epithelial-mesenchymal transition and metastasis of low-metastatic cancer cells by regulating PTEN/Akt pathway in hepatocellular carcinoma. Oncogene. 2020;39:6529–43.
Kaneda MM, Cappello P, Nguyen AV, Ralainirina N, Hardamon CR, Foubert P, et al. Macrophage PI3Kgamma drives pancreatic ductal adenocarcinoma progression. Cancer Discov. 2016;6:870–85.
Kaneda MM, Messer KS, Ralainirina N, Li H, Leem CJ, Gorjestani S, et al. PI3Kγ is a molecular switch that controls immune suppression. Nature. 2016;539:437–42.
Rocher C, Singla DK. SMAD-PI3K-Akt-mTOR pathway mediates BMP-7 polarization of monocytes into M2 macrophages. PLoS ONE. 2013;8:e84009–e84009.
Brina D, Ponzoni A, Troiani M, Calì B, Pasquini E, Attanasio G, et al. The Akt/mTOR and MNK/eIF4E pathways rewire the prostate cancer translatome to secrete HGF, SPP1 and BGN and recruit suppressive myeloid cells. Nat Cancer. 2023;4:1102–21.
Deng H, Yu B, Li Y. Tanshinone IIA alleviates acute ethanol-induced myocardial apoptosis mainly through inhibiting the expression of PDCD4 and activating the PI3K/Akt pathway. Phytother Res. 2021;35:4309–23.
Taboada R, Claro L, Felismino T, de Jesus VH, Barros M, Riechelmann RP. Clinicopathological and molecular profile of grade 3 gastroenteropancreatic neuroendocrine neoplasms. J Neuroendocrinol. 2022;34:e13099.
Scarpa A, Chang DK, Nones K, Corbo V, Patch AM, Bailey P, et al. Whole-genome landscape of pancreatic neuroendocrine tumours. Nature. 2017;543:65–71.
Yu X, Harris SL, Levine AJ. The regulation of exosome secretion: a novel function of the p53 protein. Cancer Res. 2006;66:4795–801.
Lespagnol A, Duflaut D, Beekman C, Blanc L, Fiucci G, Marine JC, et al. Exosome secretion, including the DNA damage-induced p53-dependent secretory pathway, is severely compromised in TSAP6/Steap3-null mice. Cell Death Differ. 2008;15:1723–33.
Cooks T, Pateras IS, Jenkins LM, Patel KM, Robles AI, Morris J. et al. Mutant p53 cancers reprogram macrophages to tumor supporting macrophages via exosomal miR-1246. Nat Commun.2018;9:771–85.
Feng RY, Yin Y, Wei YG, Li Y, Li L, Zhu R, et al. Mutant p53 activates hnRNPA2B1-AGAP1-mediated exosome formation to promote esophageal squamous cell carcinoma progression. Cancer Lett. 2023;562:216154.
Xia J, Zhang T, Sun Y, Huang Z, Shi D, Qin D, et al. Suppression of neuronal CDK9/p53/VDAC signaling provides bioenergetic support and improves post-stroke neuropsychiatric outcomes. Cell Mol Life Sci. 2024;81:384.
Kulkarni B, Kirave P, Gondaliya P, Jash K, Jain A, Tekade RK, et al. Exosomal miRNA in chemoresistance, immune evasion, metastasis and progression of cancer. Drug Discov Today. 2019;24:2058–67.
Li X, Zhao S, Bian X, Zhang L, Lu L, Pei S, et al. Signatures of EMT, immunosuppression, and inflammation in primary and recurrent human cutaneous squamous cell carcinoma at single-cell resolution. Theranostics. 2022;12:7532–49.
Zhang L, Li Z, Skrzypczynska KM, Fang Q, Zhang W, O’Brien SA, et al. Single-cell analyses inform mechanisms of myeloid-targeted therapies in colon cancer. Cell. 2020;181:442–459.e429.
Kale S, Raja R, Thorat D, Soundararajan G, Patil TV, Kundu GC. Osteopontin signaling upregulates cyclooxygenase-2 expression in tumor-associated macrophages leading to enhanced angiogenesis and melanoma growth via alpha 9 beta 1 integrin. Oncogene. 2014;33:2295–306.
Zhen Y, Fang W, Zhao M, Luo R, Liu Y, Fu Q, et al. miR-374a-CCND1-pPI3K/AKT-c-JUN feedback loop modulated by PDCD4 suppresses cell growth, metastasis, and sensitizes nasopharyngeal carcinoma to cisplatin. Oncogene. 2017;36:275–85.
Zhen Y, Li D, Li W, Yao W, Wu A, Huang J, et al. Reduced PDCD4 expression promotes cell growth through PI3K/Akt signaling in non-small cell lung cancer. Oncol Res. 2016;23:61–68.
Feng MG, Liu CF, Chen L, Feng WB, Liu M, Hai H, et al. MiR-21 attenuates apoptosis-triggered by amyloid-β via modulating PDCD4/ PI3K/AKT/GSK-3β pathway in SH-SY5Y cells. Biomed Pharmacother. 2018;101:1003–7.
Funding
This research was funded by the National Natural Science Foundation of China (82473108, 82472791, 82473108, 82141129, 82141104, and 82303943). Shanghai Pujiang Program (22PJ1401800). Sailing Project of Science and Technology Commission of Shanghai Municipality (23YF1406900), China Postdoctoral Science Foundation (2023M730670). National Natural Science Foundation of China (U21A20374), Shanghai Municipal Science and Technology Major Project (21JC1401500), Shanghai Municipal Health Commission’s Collaborative Guidance Project for Traditional Chinese and Western Medicine (ZXXT-202201), Scientific Innovation Project of Shanghai Education Committee (2019-01-07-00-07-E00057), Clinical Research Plan of Shanghai Hospital Development Center (SHDC2020CR1006A), and Xuhui District Artificial Intelligence Medical Hospital Cooperation Project (2021-011).
Author information
Authors and Affiliations
Contributions
Conceptualization, WZ, JX, XX, XY, and SJ; methodology, WZ, JX, DH, YQ, XL, ZY, FW, YW, DJ, GF, QZ, and CX; analysis, WZ, JX, DH, YQ, XL, and HG; writing—original draft preparation, all authors; writing—review and editing, all authors; supervision, YQ, JC, XX, XY, and SJ; project administration, WZ, DH, HG, ZY, FW, YW, DJ, GF, QZ, CX, JC, XY, and SJ; funding acquisition, JX, YQ, ZY, JC, XX, XY, and SJ. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, W., Xu, J., Huang, D. et al. Small extracellular vesicle-miR-183-5p mediated crosstalk between tumor cells and macrophages in high-risk pancreatic neuroendocrine tumors. Oncogene 44, 2907–2923 (2025). https://doi.org/10.1038/s41388-025-03462-5
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41388-025-03462-5