Abstract
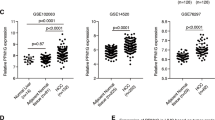
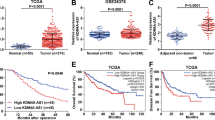
RNA polyadenylation is a key post-transcriptional modification essential for gene expression regulation. However, the role and mechanism of polyadenylation and its key molecule, polyadenylate binding protein nuclear 1 (PABPN1), in hepatocellular carcinoma (HCC) remain poorly understood. This study investigates the role of PABPN1 and its regulatory genes in HCC progression to identify potential therapeutic targets. Analysis of The Cancer Genome Atlas (TCGA) dataset and an independent HCC cohort revealed significant upregulation of PABPN1 in HCC patients, which correlates with poor prognosis. Loss-of-function studies using HCC cell lines and conditional knockout mouse models demonstrated that targeting PABPN1 inhibited HCC progression. Conversely, overexpression of PABPN1 promoted HCC development in vitro and in a hydrodynamic transfection hepatocarcinogenesis mouse model. Mechanistic investigations showed that PABPN1 modulates C5 mRNA polyadenylation and stability, with the PABPN1-C5 axis driving NF-κB activation and recruiting polymorphonuclear myeloid-derived suppressor cells (PMN-MDSCs) to promote HCC progression. Therapeutic targeting of the PABPN1-C5 axis using the C5a receptor inhibitor CCX168 significantly inhibited HCC progression in both in vitro and in vivo models. This study identifies PABPN1 as a critical regulator of HCC development and sheds light on the post-transcriptional regulation of complement components in cancer. Targeting the PABPN1-C5 axis represents a promising strategy for HCC treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The public data re-analyzed in our study was TCGA-HCC dataset. The RNA sequencing data of PABPN1 knockdown and its corresponding control HCC cells have been publicly deposited at Gene Expression Omnibus (GEO accession number: GSE289702). The data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
Anwanwan D, Singh SK, Singh S, Saikam V, Singh R. Challenges in liver cancer and possible treatment approaches. Biochim Biophys Acta Rev Cancer. 2020;1873:188314.
Li X, Ramadori P, Pfister D, Seehawer M, Zender L, Heikenwalder M. The immunological and metabolic landscape in primary and metastatic liver cancer. Nat Rev Cancer. 2021;21:541–57.
Vogel A, Meyer T, Sapisochin G, Salem R, Saborowski A. Hepatocellular carcinoma. Lancet. 2022;400:1345–62.
Barbieri I, Kouzarides T. Role of RNA modifications in cancer. Nat Rev Cancer. 2020;20:303–22.
Lin S, Kuang M. RNA modification-mediated mRNA translation regulation in liver cancer: mechanisms and clinical perspectives. Nat Rev Gastroenterol Hepatol. 2024;21:267–81.
Passmore LA, Coller J. Roles of mRNA poly(A) tails in regulation of eukaryotic gene expression. Nat Rev Mol Cell Biol. 2022;23:93–106.
Xiang Y, Ye Y, Lou Y, Yang Y, Cai C, Zhang Z, et al. Comprehensive characterization of alternative polyadenylation in human cancer. J Natl Cancer Inst. 2018;110:379–89.
Banerjee A, Vest KE, Pavlath GK, Corbett AH. Nuclear poly(A) binding protein 1 (PABPN1) and Matrin3 interact in muscle cells and regulate RNA processing. Nucleic Acids Res. 2017;45:10706–25.
Malerba A, Klein P, Bachtarzi H, Jarmin SA, Cordova G, Ferry A, et al. PABPN1 gene therapy for oculopharyngeal muscular dystrophy. Nat Commun. 2017;8:14848.
Zhao LW, Zhu YZ, Wu YW, Pi SB, Shen L, Fan HY. Nuclear poly(A) binding protein 1 (PABPN1) mediates zygotic genome activation-dependent maternal mRNA clearance during mouse early embryonic development. Nucleic Acids Res. 2022;50:458–72.
Malik A, Thanekar U, Amarachintha S, Mourya R, Nalluri S, Bondoc A, et al. “Complimenting the complement”: mechanistic insights and opportunities for therapeutics in hepatocellular carcinoma. Front Oncol. 2020;10:627701.
Xiao Z, Yeung CLS, Yam JWP, Mao X. An update on the role of complement in hepatocellular carcinoma. Front Immunol. 2022;13:1007382.
Liu J, Li W, Zhao H. CFHR3 is a potential novel biomarker for hepatocellular carcinoma. J Cell Biochem. 2020;121:2970–80.
Yeung OWH, Qi X, Pang L, Liu H, Ng KTP, Liu J, et al. Type III TGF-β receptor down-regulation promoted tumor progression via complement component C5a induction in hepatocellular carcinoma. Cancers. 2021;13:1503.
Walport MJ. Complement. First of two parts. N Engl J Med. 2001;344:1058–66.
Reis ES, Mastellos DC, Hajishengallis G, Lambris JD. New insights into the immune functions of complement. Nat Rev Immunol. 2019;19:503–16.
Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell. 2010;140:883–99.
Markiewski MM, DeAngelis RA, Benencia F, Ricklin-Lichtsteiner SK, Koutoulaki A, Gerard C, et al. Modulation of the antitumor immune response by complement. Nat Immunol. 2008;9:1225–35.
Pan ZK. Anaphylatoxins C5a and C3a induce nuclear factor kappaB activation in human peripheral blood monocytes. Biochim Biophys Acta. 1998;1443:90–98.
Carmona-Fontaine C, Theveneau E, Tzekou A, Tada M, Woods M, Page KM, et al. Complement fragment C3a controls mutual cell attraction during collective cell migration. Dev Cell. 2011;21:1026–37.
Patil DP, Bakthavachalu B, Schoenberg DR. Poly(A) polymerase-based poly(A) length assay. Methods Mol Biol. 2014;1125:13–23.
Choe J, Lin S, Zhang W, Liu Q, Wang L, Ramirez-Moya J, et al. mRNA circularization by METTL3-eIF3h enhances translation and promotes oncogenesis. Nature. 2018;561:556–60.
Bankhead P, Loughrey MB, Fernández JA, Dombrowski Y, McArt DG, Dunne PD, et al. QuPath: open source software for digital pathology image analysis. Sci Rep. 2017;7:16878.
Elieh Ali Komi D, Shafaghat F, Kovanen PT, Meri S. Mast cells and complement system: ancient interactions between components of innate immunity. Allergy. 2020;75:2818–28.
Hess C, Kemper C. Complement-mediated regulation of metabolism and basic cellular processes. Immunity. 2016;45:240–54.
Afshar-Kharghan V. The role of the complement system in cancer. J Clin Invest. 2017;127:780–9.
de Klerk E, Venema A, Anvar SY, Goeman JJ, Hu O, Trollet C, et al. Poly(A) binding protein nuclear 1 levels affect alternative polyadenylation. Nucleic Acids Res. 2012;40:9089–101.
Gordon JM, Phizicky DV, Schärfen L, Brown CL, Arias Escayola D, Kanyo J, et al. Phosphorylation of the nuclear poly(A) binding protein (PABPN1) during mitosis protects mRNA from hyperadenylation and maintains transcriptome dynamics. Nucleic Acids Res. 2024;52:9886–903.
Liu P, Kimmoun E, Legrand A, Sauvanet A, Degott C, Lardeux B, et al. Activation of NF-kappa B, AP-1 and STAT transcription factors is a frequent and early event in human hepatocellular carcinomas. J Hepatol. 2002;37:63–71.
Luedde T, Schwabe RF. NF-κB in the liver-linking injury, fibrosis and hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2011;8:108–18.
Liu Y, Nie H, Liu H, Lu F. Poly(A) inclusive RNA isoform sequencing (PAIso-seq) reveals wide-spread non-adenosine residues within RNA poly(A) tails. Nat Commun. 2019;10:5292.
Fan J, To KKW, Chen ZS, Fu L. ABC transporters affects tumor immune microenvironment to regulate cancer immunotherapy and multidrug resistance. Drug Resist Updat. 2023;66:100905.
He G, Karin M. NF-κB and STAT3 - key players in liver inflammation and cancer. Cell Res. 2011;21:159–68.
Zhou J, Nefedova Y, Lei A, Gabrilovich D. Neutrophils and PMN-MDSC: their biological role and interaction with stromal cells. Semin Immunol. 2018;35:19–28.
Jayne DRW, Merkel PA, Schall TJ, Bekker P. Avacopan for the treatment of ANCA-associated vasculitis. N Engl J Med. 2021;384:599–609.
Acknowledgements
This work was funded by National Key Research and Development Program of China (2023YFA1800804 to SL, 2022YFE0138700 to SL, 2022YFA1105300 to SL), National Natural Science Foundation of China (82325036 to SL, 82201734 and 82472753 to CZ, 82200954 to JM), Fundamental Research Funds for the Central Universities, Sun Yat-sen University (23ykzy004 to SL).
Author information
Authors and Affiliations
Contributions
SG and CZ designed the protocol of this study. QZ and JM performed in vitro assays. SG, YZ, ZW and SZ performed in vivo assays. All other data analysis was performed by SG, HQ, CZ and JC. SG, CZ and SL wrote the paper and all authors reviewed the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All human specimens were approved by the Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University (Approved No: [2020]356 and [2022]180). We have obtained patients’ informed consent for publication. All animal experiments were approved by the Institutional Care and Animal Use Committee of the First Affiliated Hospital of Sun Yat-sen University (Approval No [2023]147). All methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, S., Zhang, Q., Ma, J. et al. PABPN1-C5 axis promotes hepatocellular carcinoma progression via NF-κB activation. Oncogene 44, 3512–3524 (2025). https://doi.org/10.1038/s41388-025-03501-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41388-025-03501-1