Abstract
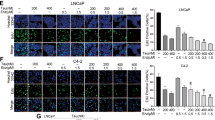
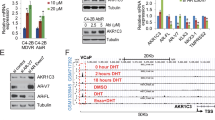
The introduction of next-generation androgen receptor signaling inhibitors (ARSIs) like enzalutamide (ENZ), has improved the clinical management of castration-resistant prostate cancer (CRPC). However, acquired resistance to these therapies often develops rapidly, and the underlying resistance mechanisms remain largely unclear. Here, we identified the aryl hydrocarbon receptor (AHR) as a crucial operator of ENZ-resistant CRPC. AHR is upregulated in three ENZ-resistant human CRPC cell lines (C4-2BENZR, CWR-R1ENZR, and VCaPENZR) as well as in high-grade prostate tumors from patients receiving ENZ treatment. Stable knockdown of AHR substantially reduced the growth of ENZ-resistant CRPC cells and xenografts. Mechanistically, AHR engages in distinct transcriptional programs in a cellular context-dependent manner. AHR directly regulates the transcription and expression of androgen receptor (AR)/glucocorticoid receptor (GR) co-target genes in CWR-R1ENZR cells, suggesting an AR-dependent mechanism of ENZ resistance. AHR promotes neuroendocrine differentiation while suppressing the expression of AR/GR targets in C4-2BENZR cells, indicating an AR-indifferent mechanism of ENZ resistance. The diverse mechanisms triggered by ENZ were also manifested in clinical samples. Collectively, these findings characterize AHR’s contribution to ENZ resistance in CRPC and illuminate the potential of targeting AHR for treating ARSI-resistant advanced prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All sequencing results are available in the NCBI GEO database under the following accession numbers: GSE289313 for AHR ChIP-seq of control and AHR-knockdown CWR-R1ENZR cells, GSE289910 for RNA-seq of control and AHR-knockdown CWR-R1ENZR and C4-2BENZR cells, and GSE290016 for RNA-seq of ENZR and ENZS C4-2B, CWR-R1, and VCaP cells.
References
Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA: A Cancer J Clinicians. 2024;74:12–49.
James ND, Tannock I, N’Dow J, Feng F, Gillessen S, Ali SA, et al. The Lancet Commission on prostate cancer: planning for the surge in cases. Lancet. 2024;403:1683–722.
Scher HI, Beer TM, Higano CS, Anand A, Taplin ME, Efstathiou E, et al. Antitumour activity of MDV3100 in castration-resistant prostate cancer: a phase 1-2 study. Lancet. 2010;375:1437–46.
Scher HI, Fizazi K, Saad F, Taplin ME, Sternberg CN, Miller K, et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187–97.
Sternberg CN, Fizazi K, Saad F, Shore ND, De Giorgi U, Penson DF, et al. Enzalutamide and Survival in Nonmetastatic, Castration-Resistant Prostate Cancer. N Engl J Med. 2020;382:2197–206.
Viswanathan SR, Ha G, Hoff AM, Wala JA, Carrot-Zhang J, Whelan CW, et al. Structural Alterations Driving Castration-Resistant Prostate Cancer Revealed by Linked-Read Genome Sequencing. Cell. 2018;174:433–47.e419.
Antonarakis ES, Lu C, Wang H, Luber B, Nakazawa M, Roeser JC, et al. AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N Engl J Med. 2014;371:1028–38.
Li Y, Chan SC, Brand LJ, Hwang TH, Silverstein KA, Dehm SM. Androgen receptor splice variants mediate enzalutamide resistance in castration-resistant prostate cancer cell lines. Cancer Res. 2013;73:483–9.
Joseph JD, Lu N, Qian J, Sensintaffar J, Shao G, Brigham D, et al. A clinically relevant androgen receptor mutation confers resistance to second-generation antiandrogens enzalutamide and ARN-509. Cancer Discov. 2013;3:1020–9.
Huhtaniemi R, Sipila P, Junnila A, Oksala R, Knuuttila M, Mehmood A, et al. High intratumoral dihydrotestosterone is associated with antiandrogen resistance in VCaP prostate cancer xenografts in castrated mice. iScience. 2022;25:104287.
Liu C, Lou W, Zhu Y, Yang JC, Nadiminty N, Gaikwad NW, et al. Intracrine Androgens and AKR1C3 Activation Confer Resistance to Enzalutamide in Prostate Cancer. Cancer Res (Res Support, NIH, Extramural). 2015;75:1413–22.
He Y, Wei T, Ye Z, Orme JJ, Lin D, Sheng H, et al. A noncanonical AR addiction drives enzalutamide resistance in prostate cancer. Nat Commun. 2021;12:1521.
Arora VK, Schenkein E, Murali R, Subudhi SK, Wongvipat J, Balbas MD, et al. Glucocorticoid receptor confers resistance to antiandrogens by bypassing androgen receptor blockade. Cell. 2013;155:1309–22.
Puhr M, Eigentler A, Handle F, Hackl H, Ploner C, Heidegger I, et al. Targeting the glucocorticoid receptor signature gene Mono Amine Oxidase-A enhances the efficacy of chemo- and anti-androgen therapy in advanced prostate cancer. Oncogene. 2021;40:3087–3100.
Dutta S, Khedmatgozar H, Patel GK, Latour D, Welsh J, Mustafi M, et al. A TBX2-driven signaling switch from androgen receptor to glucocorticoid receptor confers therapeutic resistance in prostate cancer. Oncogene. 2025;44:877–92.
Linder S, Hoogstraat M, Stelloo S, Eickhoff N, Schuurman K, de Barros H, et al. Drug-Induced Epigenomic Plasticity Reprograms Circadian Rhythm Regulation to Drive Prostate Cancer toward Androgen Independence. Cancer Discov. 2022;12:2074–97.
Moll JM, Hofland J, Teubel WJ, de Ridder CMA, Taylor AE, Graeser R, et al. Abiraterone switches castration-resistant prostate cancer dependency from adrenal androgens towards androgen receptor variants and glucocorticoid receptor signalling. Prostate. 2022;82:505–16.
Puhr M, Hoefer J, Eigentler A, Ploner C, Handle F, Schaefer G, et al. The Glucocorticoid Receptor Is a Key Player for Prostate Cancer Cell Survival and a Target for Improved Antiandrogen Therapy. Clin Cancer Res: Off J Am Assoc Cancer Res. 2018;24:927–38.
Beltran H, Prandi D, Mosquera JM, Benelli M, Puca L, Cyrta J, et al. Divergent clonal evolution of castration-resistant neuroendocrine prostate cancer. Nat Med. 2016;22:298–305.
Labrecque MP, Coleman IM, Brown LG, True LD, Kollath L, Lakely B, et al. Molecular profiling stratifies diverse phenotypes of treatment-refractory metastatic castration-resistant prostate cancer. J Clin Invest. 2019;129:4492–505.
Cejas P, Xie Y, Font-Tello A, Lim K, Syamala S, Qiu X, et al. Subtype heterogeneity and epigenetic convergence in neuroendocrine prostate cancer. Nat Commun. 2021;12:5775.
Lin TP, Chang YT, Lee SY, Campbell M, Wang TC, Shen SH, et al. REST reduction is essential for hypoxia-induced neuroendocrine differentiation of prostate cancer cells by activating autophagy signaling. Oncotarget. 2016;7:26137–51.
Lee JK, Phillips JW, Smith BA, Park JW, Stoyanova T, McCaffrey EF, et al. N-Myc Drives Neuroendocrine Prostate Cancer Initiated from Human Prostate Epithelial Cells. Cancer cell. 2016;29:536–47.
Akamatsu S, Wyatt AW, Lin D, Lysakowski S, Zhang F, Kim S, et al. The Placental Gene PEG10 Promotes Progression of Neuroendocrine Prostate Cancer. Cell Rep. 2015;12:922–36.
Guo H, Ci X, Ahmed M, Hua JT, Soares F, Lin D, et al. ONECUT2 is a driver of neuroendocrine prostate cancer. Nat Commun. 2019;10:278.
Bland T, Wang J, Yin L, Pu T, Li J, Gao J, et al. WLS-Wnt signaling promotes neuroendocrine prostate cancer. iScience. 2021;24:101970.
Murray IA, Patterson AD, Perdew GH. Aryl hydrocarbon receptor ligands in cancer: friend and foe. Nat Rev Cancer. 2014;14:801–14.
Sadik A, Somarribas Patterson LF, Ozturk S, Mohapatra SR, Panitz V, Secker PF, et al. IL4I1 Is a Metabolic Immune Checkpoint that Activates the AHR and Promotes Tumor Progression. Cell. 2020;182:1252–70.e1234.
Tran C, Richmond O, Aaron L, Powell JB. Inhibition of constitutive aryl hydrocarbon receptor (AhR) signaling attenuates androgen independent signaling and growth in (C4-2) prostate cancer cells. Biochemical Pharmacol. 2013;85:753–62.
Richmond O, Ghotbaddini M, Allen C, Walker A, Zahir S, Powell JB. The aryl hydrocarbon receptor is constitutively active in advanced prostate cancer cells. PloS one. 2014;9:e95058.
Fritz WA, Lin TM, Cardiff RD, Peterson RE. The aryl hydrocarbon receptor inhibits prostate carcinogenesis in TRAMP mice. Carcinogenesis. 2007;28:497–505.
Morrow D, Qin C, Smith R 3rd, Safe S. Aryl hydrocarbon receptor-mediated inhibition of LNCaP prostate cancer cell growth and hormone-induced transactivation. J Steroid Biochem Mol Biol. 2004;88:27–36.
Chen Z, Cai A, Zheng H, Huang H, Sun R, Cui X, et al. Carbidopa suppresses prostate cancer via aryl hydrocarbon receptor-mediated ubiquitination and degradation of androgen receptor. Oncogenesis. 2020;9:49.
Sun F, Indran IR, Zhang ZW, Tan MH, Li Y, Lim ZL, et al. A novel prostate cancer therapeutic strategy using icaritin-activated arylhydrocarbon-receptor to co-target androgen receptor and its splice variants. Carcinogenesis. 2015;36:757–68.
Kregel S, Chen JL, Tom W, Krishnan V, Kach J, Brechka H, et al. Acquired resistance to the second-generation androgen receptor antagonist enzalutamide in castration-resistant prostate cancer. Oncotarget. 2016;7:26259–74.
Ho PP, Steinman L. The aryl hydrocarbon receptor: a regulator of Th17 and Treg cell development in disease. Cell Res. 2008;18:605–8.
Li C, Liu J, He D, Mao F, Rao X, Zhao Y, et al. GSTM2 is a key molecular determinant of resistance to SG-ARIs. Oncogene. 2022;41:4498–511.
Hruba E, Vondracek J, Libalova H, Topinka J, Bryja V, Soucek K, et al. Gene expression changes in human prostate carcinoma cells exposed to genotoxic and nongenotoxic aryl hydrocarbon receptor ligands. Toxicol Lett. 2011;206:178–88.
Abida W, Cyrta J, Heller G, Prandi D, Armenia J, Coleman I, et al. Genomic correlates of clinical outcome in advanced prostate cancer. Proc Natl Acad Sci USA. 2019;116:11428–36.
Sayar E, Patel RA, Coleman IM, Roudier MP, Zhang A, Mustafi P, et al. Reversible epigenetic alterations mediate PSMA expression heterogeneity in advanced metastatic prostate cancer. JCI Insight. 2023;8:e162907.
Ohtake F, Fujii-Kuriyama Y, Kato S. AhR acts as an E3 ubiquitin ligase to modulate steroid receptor functions. Biochemical Pharmacol. 2009;77:474–84.
Wei J, Yin L, Li J, Wang J, Pu T, Duan P, et al. Bidirectional Cross-talk between MAOA and AR Promotes Hormone-Dependent and Castration-Resistant Prostate Cancer. Cancer Res. 2021;81:4275–89.
Zhang XQ, Kondrikov D, Yuan TC, Lin FF, Hansen J, Lin MF. Receptor protein tyrosine phosphatase alpha signaling is involved in androgen depletion-induced neuroendocrine differentiation of androgen-sensitive LNCaP human prostate cancer cells. Oncogene. 2003;22:6704–16.
Bishop JL, Thaper D, Vahid S, Davies A, Ketola K, Kuruma H, et al. The Master Neural Transcription Factor BRN2 Is an Androgen Receptor-Suppressed Driver of Neuroendocrine Differentiation in Prostate Cancer. Cancer Discov. 2017;7:54–71.
Haffner MC, Zwart W, Roudier MP, True LD, Nelson WG, Epstein JI, et al. Genomic and phenotypic heterogeneity in prostate cancer. Nat Rev Urol. 2021;18:79–92.
Sladekova L, Mani S, Dvorak Z. Ligands and agonists of the aryl hydrocarbon receptor AhR: Facts and myths. Biochemical Pharmacol. 2023;213:115626.
Gerbal-Chaloin S, Pichard-Garcia L, Fabre JM, Sa-Cunha A, Poellinger L, Maurel P, et al. Role of CYP3A4 in the regulation of the aryl hydrocarbon receptor by omeprazole sulphide. Cell Signal. 2006;18:740–50.
Zhao B, Degroot DE, Hayashi A, He G, Denison MS. CH223191 is a ligand-selective antagonist of the Ah (Dioxin) receptor. Toxicol Sci. 2010;117:393–403.
Granados JC, Falah K, Koo I, Morgan EW, Perdew GH, Patterson AD, et al. AHR is a master regulator of diverse pathways in endogenous metabolism. Sci Rep. 2022;12:16625.
Keam SJ. Tapinarof Cream 1%: First Approval. Drugs. 2022;82:1221–8.
Beltran H, Hruszkewycz A, Scher HI, Hildesheim J, Isaacs J, Yu EY, et al. The Role of Lineage Plasticity in Prostate Cancer Therapy Resistance. Clin Cancer Res: Off J Am Assoc Cancer Res. 2019;25:6916–24.
Sondermann NC, Fassbender S, Hartung F, Hatala AM, Rolfes KM, Vogel CFA, et al. Functions of the aryl hydrocarbon receptor (AHR) beyond the canonical AHR/ARNT signaling pathway. Biochemical Pharmacol. 2023;208:115371.
Chan JM, Zaidi S, Love JR, Zhao JL, Setty M, Wadosky KM, et al. Lineage plasticity in prostate cancer depends on JAK/STAT inflammatory signaling. Science. 2022;377:1180–91.
Deng S, Wang C, Wang Y, Xu Y, Li X, Johnson NA, et al. Ectopic JAK-STAT activation enables the transition to a stem-like and multilineage state conferring AR-targeted therapy resistance. Nat Cancer. 2022;3:1071–87.
Xiong J, Zhang X, Zhang Y, Wu B, Fang L, Wang N, et al. Aryl hydrocarbon receptor mediates Jak2/STAT3 signaling for non-small cell lung cancer stem cell maintenance. Exp Cell Res. 2020;396:112288.
Acknowledgements
We thank Jing Wei for technical help and Gary Mawyer for editorial assistance. This work was supported by NIH/NCI grants R37CA233658, R01CA258634, and R01CA279528 (to BJW).
Author information
Authors and Affiliations
Contributions
BJW and CHC conceptualized the study. CHC performed all experiments, analyzed data, and wrote the draft of the manuscript. BJW designed experiments, edited the manuscript, and acquired funding. RB, DVG, and ACG provided ENZS and ENZR cell lines.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All methods were performed in accordance with the relevant guidelines and regulations. All animal studies received prior approval from the Institutional Animal Care and Use Committee (IACUC) at Washington State University (No. 6635) and complied with IACUC recommendations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, CH., Brown, R., Vander Griend, D.J. et al. Aryl hydrocarbon receptor is critical for both AR-dependent and AR-indifferent enzalutamide resistance in castration-resistant prostate cancer. Oncogene 45, 1312–1323 (2026). https://doi.org/10.1038/s41388-026-03723-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41388-026-03723-x