Abstract
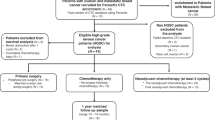
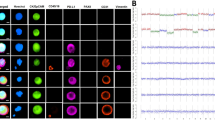
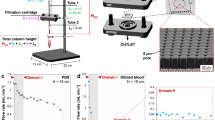
High-grade serous ovarian cancer (HGSOC) is the most lethal gynecological malignancy. Pre- and post-operative, non-invasive biomarkers for reliable treatment outcome prediction, (minimal) residual disease detection, and prognosis are not yet established in clinical practice for these patients. This prospective study quantified circulating tumor cells (CTCs) in 7.5 ml peripheral blood in 56 women with FIGO stages IIIC and IV HGSOC before and after primary cytoreductive surgery and in 9 women with benign ovarian disease. Clinical outcomes were assessed during the median follow-up of 35.4 months. CTCs were detected in 48.2% (27/56) of patients pre-operatively and in 46.4% (26/56) post-operatively, but not in benign controls. Pre-operative CTCs were associated with suboptimal cytoreductive surgery (OR = 15.6, 95% CI: 2.97–127.0, p = 0.0031), worse platinum response (p = 0.0173), lymph node metastases (p = 0.0151), and shorter progression-free (p = 0.0045) and overall survival (p = 0.0241). Post-operative CTC-augmented residual tumor was significantly associated with worse OS (p = 0.047). In multivariable analyses, pre-operative CTCs remained an independent surrogate marker for incomplete debulking, platinum resistance, and poor survival in HGSOC. Therefore, the quantification of CTCs in HGSOC pre- and post-operatively may be used to guide treatment selection, reduce surgical morbidity, improve the healthcare provider’s resource allocation and planning, and enhance patient counseling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Huang J, Chan WC, Ngai CH, Lok V, Zhang L, Lucero-Prisno DE 3rd, et al. Worldwide burden, risk factors, and temporal trends of ovarian cancer: a global study. Cancers. 2022;14:2230.
Smith LH, Morris CR, Yasmeen S, Parikh-Patel A, Cress RD, Romano PS. Ovarian cancer: can we make the clinical diagnosis earlier? Cancer. 2005;104:1398–407.
Torre LA, Trabert B, DeSantis CE, Miller KD, Samimi G, Runowicz CD, et al. Ovarian cancer statistics, 2018. CA Cancer J Clin. 2018;68:284–96.
Aletti GD, Dowdy SC, Gostout BS, Jones MB, Stanhope CR, Wilson TO, et al. Aggressive surgical effort and improved survival in advanced-stage ovarian cancer. Obstet Gynecol. 2006;107:77–85.
Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, Deutsche Krebshilfe, AWMF). S3-Leitlinie Diagnostik, Therapie und Nachsorge maligner Ovarialtumoren. version 5.1, AWMF-Registernummer: 032/035OL. AWMF online. Accessible from https://www.leitlinienprogramm-onkologie.de/leitlinien/ovarialkarzinom (2022).
Lin Q, Liu W, Xu S, Li J, Tong J. The value of systematic lymphadenectomy during debulking surgery in the treatment of ovarian cancer: a meta-analysis of randomized controlled trials. J Ovar Res. 2020;13:56.
Iida Y, Kobayashi-Kato M, Komatsu H, Ishikawa M, Satoh TGynecologic Cancer Study Group of the Japan Clinical Oncology Group Questionnaire-based survey on the extent of lymph node dissection during interval debulking surgery after neoadjuvant chemotherapy for patients with advanced ovarian cancer in the Gynecologic Cancer Study Group of JCOG. Int J Clin Oncol. 2025;30:1040–7.
Colombo N, Sessa C, du Bois A, Ledermann J, McCluggage WG, McNeish I, et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Ann Oncol. 2019;30:672–705.
Xu Z, Becerra AZ, Justiniano CF, Aquina CT, Fleming FJ, Boscoe FP, et al. Complications and survivorship trends after primary debulking surgery for ovarian cancer. J Surg Res. 2020;246:34–41.
National Institute for Health and Care Excellence (NICE). Interventional procedure overview of maximal cytoreductive surgery for advanced ovarian cancer. NICE. Accessible from: https://www.nice.org.uk/consultations/1769/6/summary-of-key-evidence-on-maximal-effort-cytoreductive-surgery-for-advanced-ovarian-cancer (2022).
Gerestein CG, Damhuis RA, de Vries M, Reedijk A, Burger CW, Kooi GS. Causes of postoperative mortality after surgery for ovarian cancer. Eur J Cancer. 2009;45:2799–803.
Bidzinski M, Derlatka P, Kubik P, Ziolkowska-Seta I, Danska-Bidzinska A, Gmyrek L, et al. The evaluation of intra- and postoperative complications related to debulking surgery with bowel resection in patients with FIGO stage III-IV ovarian cancer. Int J Gynecol Cancer. 2007;17:993–7.
Ledermann JA, Raja FA, Fotopoulou C, Gonzalez-Martin A, Colombo N, Sessa C, et al. Newly diagnosed and relapsed epithelial ovarian carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24:vi24–32.
Lowe KA, Chia VM, Taylor A, O’Malley C, Kelsh M, Mohamed M, et al. An international assessment of ovarian cancer incidence and mortality. Gynecol Oncol. 2013;130:107–14.
Kang S, Kim TJ, Nam BH, Seo SS, Kim BG, Bae DS, et al. Preoperative serum CA-125 levels and risk of suboptimal cytoreduction in ovarian cancer: a meta-analysis. J Surg Oncol. 2010;101:13–7.
Vorgias G, Iavazzo C, Savvopoulos P, Myriokefalitaki E, Katsoulis M, Kalinoglou N, et al. Can the preoperative Ca-125 level predict optimal cytoreduction in patients with advanced ovarian carcinoma? A single institution cohort study. Gynecol Oncol. 2009;112:11–5.
Merlo S, Besic N, Drmota E, Kovacevic N. Preoperative serum CA-125 level as a predictor for the extent of cytoreduction in patients with advanced stage epithelial ovarian cancer. Radio Oncol. 2021;55:341–6.
Joosse SA, Gorges TM, Pantel K. Biology, detection, and clinical implications of circulating tumor cells. EMBO Mol Med. 2015;7:1–11.
He X, Li S, Ni Y, Jin M, Fu X. A meta-analysis of the prognostic value of circulating tumor cells in ovarian cancer. Am J Transl Res. 2022;14:3574–83.
Wang T, Gao Y, Wang X, Tian J, Li Y, Yu B, et al. Establishment of an optimized CTC detection model consisting of EpCAM, MUC1 and WT1 in epithelial ovarian cancer and its correlation with clinical characteristics. Chin J Cancer Res. 2022;34:95–108.
Kuhlmann JD, Wimberger P, Bankfalvi A, Keller T, Scholer S, Aktas B, et al. ERCC1-positive circulating tumor cells in the blood of ovarian cancer patients as a predictive biomarker for platinum resistance. Clin Chem. 2014;60:1282–9.
Chebouti I, Kuhlmann JD, Buderath P, Weber S, Wimberger P, Bokeloh Y, et al. ERCC1-expressing circulating tumor cells as a potential diagnostic tool for monitoring response to platinum-based chemotherapy and for predicting post-therapeutic outcome of ovarian cancer. Oncotarget. 2017;8:24303–13.
Lee M, Kim EJ, Cho Y, Kim S, Chung HH, Park NH, et al. Predictive value of circulating tumor cells (CTCs) captured by microfluidic device in patients with epithelial ovarian cancer. Gynecol Oncol. 2017;145:361–5.
Elazezy M, Prieske K, Kluwe L, Oliveira-Ferrer L, Peine S, Muller V, et al. BRCA1 promoter hypermethylation on circulating tumor DNA correlates with improved survival of patients with ovarian cancer. Mol Oncol. 2021;15:3615–25.
Lou E, Vogel RI, Hoostal S, Wong P, Grad A, Monu M, et al. Analysis of differentially expressed MicroRNAs and circulating tumor cells as predictive biomarkers of platinum chemoresistance in primary ovarian carcinomas: a prospective study. Oncologist. 2019;24:1422–e013.
Stuart GC, Kitchener H, Bacon M, duBois A, Friedlander M, Ledermann J, et al. 2010 Gynecologic Cancer InterGroup (GCIG) consensus statement on clinical trials in ovarian cancer: report from the Fourth Ovarian Cancer Consensus Conference. Int J Gynecol Cancer. 2011;21:750–5.
Smerage JB, Barlow WE, Hortobagyi GN, Winer EP, Leyland-Jones B, Srkalovic G, et al. Circulating tumor cells and response to chemotherapy in metastatic breast cancer: SWOG S0500. J Clin Oncol. 2014;32:3483–9.
Cohen SJ, Punt CJ, Iannotti N, Saidman BH, Sabbath KD, Gabrail NY, et al. Prognostic significance of circulating tumor cells in patients with metastatic colorectal cancer. Ann Oncol. 2009;20:1223–9.
de Bono JS, Scher HI, Montgomery RB, Parker C, Miller MC, Tissing H, et al. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res. 2008;14:6302–9.
Poveda A, Kaye SB, McCormack R, Wang S, Parekh T, Ricci D, et al. Circulating tumor cells predict progression-free survival and overall survival in patients with relapsed/recurrent advanced ovarian cancer. Gynecol Oncol. 2011;122:567–72.
Liu JF, Kindelberger D, Doyle C, Lowe A, Barry WT, Matulonis UA. Predictive value of circulating tumor cells (CTCs) in newly-diagnosed and recurrent ovarian cancer patients. Gynecol Oncol. 2013;131:352–6.
Banys-Paluchowski M, Fehm T, Neubauer H, Paluchowski P, Krawczyk N, Meier-Stiegen F, et al. Clinical relevance of circulating tumor cells in ovarian, fallopian tube and peritoneal cancer. Arch Gynecol Obstet. 2020;301:1027–35.
Miller MC, Doyle GV, Terstappen LW. Significance of circulating tumor cells detected by the CellSearch system in patients with metastatic breast colorectal and prostate cancer. J Oncol. 2010;2010:617421.
Gao Y, Fan WH, Song Z, Lou H, Kang X. Comparison of circulating tumor cell (CTC) detection rates with epithelial cell adhesion molecule (EpCAM) and cell surface vimentin (CSV) antibodies in different solid tumors: a retrospective study. PeerJ. 2021;9:e10777.
Allard WJ, Matera J, Miller MC, Repollet M, Connelly MC, Rao C, et al. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res. 2004;10:6897–904.
Biosystems MS. CellSearch Circulating Tumor Cell Kit (Epithelial) IVD REF 7900001. Menarini Silicon Biosystems Inc. 10355 Science Center Drive, Suite 210, San Diego, CA 92121, USA; 2017.
Joosse SA. In-Silico Online v2.4.0 [accessed on Oct 2024]. Available from: http://in-silico.online.
Joosse SA, Souche FR, Babayan A, Gasch C, Kerkhoven RM, Ramos J, et al. Chromosomal aberrations associated with sequential steps of the metastatic cascade in colorectal cancer patients. Clin Chem. 2018;64:1505–12.
Emurlai G, Pose RM, Kalra N, Coith C, Lenz S, Tennstedt P, et al. Comparative analysis of circulating tumor cells in prostatic plexus and peripheral blood of patients undergoing prostatectomy. J Exp Clin Cancer Res. 2025;44:143.
Olson MT, Ly QP, Mohs AM. Fluorescence guidance in surgical oncology: challenges, opportunities, and translation. Mol Imaging Biol. 2019;21:200–18.
Obermayr E, Castillo-Tong DC, Pils D, Speiser P, Braicu I, Van Gorp T, et al. Molecular characterization of circulating tumor cells in patients with ovarian cancer improves their prognostic significance - a study of the OVCAD consortium. Gynecol Oncol. 2013;128:15–21.
Lou E, Vogel RI, Teoh D, Hoostal S, Grad A, Gerber M, et al. Assessment of circulating tumor cells as a predictive biomarker of histology in women with suspected ovarian cancer. Lab Med. 2018;49:134–9.
Zhou Y, Bian B, Yuan X, Xie G, Ma Y, Shen L. Prognostic value of circulating tumor cells in ovarian cancer: a meta-analysis. PLoS One. 2015;10:e0130873.
Zhao H, Wang L, Fang C, Li C, Zhang L. Factors influencing the diagnostic and prognostic values of circulating tumor cells in breast cancer: a meta-analysis of 8,935 patients. Front Oncol. 2023;13:1272788.
Yao Y, Zhu X, Liu W, Jiang J, Jiang H. Meta-analysis of the prognostic value of circulating tumor cells in gastrointestinal cancer. Med (Baltim). 2022;101:e31099.
Kuo YC, Chuang CH, Kuo HC, Lin CT, Chao A, Huang HJ, et al. Circulating tumor cells help differentiate benign ovarian lesions from cancer before surgery: a literature review and proof of concept study using flow cytometry with fluorescence imaging. Oncol Lett. 2024;27:234.
Abreu M, Cabezas-Sainz P, Alonso-Alconada L, Ferreiros A, Mondelo-Macia P, Lago-Leston RM, et al. Circulating tumor cells characterization revealed timp1 as a potential therapeutic target in ovarian cancer. Cells. 2020;9:1218.
Obermayr E, Bednarz-Knoll N, Orsetti B, Weier HU, Lambrechts S, Castillo-Tong DC, et al. Circulating tumor cells: potential markers of minimal residual disease in ovarian cancer? a study of the OVCAD consortium. Oncotarget. 2017;8:106415–28.
Joosse SA, Pantel K. Biologic challenges in the detection of circulating tumor cells. Cancer Res. 2013;73:8–11.
Loret N, Denys H, Tummers P, Berx G. The role of epithelial-to-mesenchymal plasticity in ovarian cancer progression and therapy resistance. Cancers. 2019;11:838.
Fang D, Chen H, Zhu JY, Wang W, Teng Y, Ding HF, et al. Epithelial-mesenchymal transition of ovarian cancer cells is sustained by Rac1 through simultaneous activation of MEK1/2 and Src signaling pathways. Oncogene. 2017;36:1546–58.
Xu L, Mao X, Guo T, Chan PY, Shaw G, Hines J, et al. The novel association of circulating tumor cells and circulating megakaryocytes with prostate cancer prognosis. Clin Cancer Res. 2017;23:5112–22.
Xu L, Mao X, Grey A, Scandura G, Guo T, Burke E, et al. Noninvasive detection of clinically significant prostate cancer using circulating tumor cells. J Urol. 2020;203:73–82.
Woo HJ, Rademacher PN, Shin HY, Lee J, Intisar A, Warkiani ME, et al. Robust automated separation of circulating tumor cells and cancer-associated fibroblasts for enhanced liquid biopsy in breast cancer. Anal Chem. 2025;97:17452–61.
Koch C, Joosse SA, Schneegans S, Wilken OJW, Janning M, Loreth D, et al. Pre-analytical and analytical variables of label-independent enrichment and automated detection of circulating tumor cells in cancer patients. Cancers. 2020;12:442.
Salmon C, Levermann J, Neves RPL, Liffers ST, Kuhlmann JD, Buderath P, et al. Image-based identification and genomic analysis of single circulating tumor cells in high-grade serous ovarian cancer patients. Cancers. 2021;13:3748.
Blassl C, Kuhlmann JD, Webers A, Wimberger P, Fehm T, Neubauer H. Gene expression profiling of single circulating tumor cells in ovarian cancer - Establishment of a multi-marker gene panel. Mol Oncol. 2016;10:1030–42.
Chebouti I, Kasimir-Bauer S, Buderath P, Wimberger P, Hauch S, Kimmig R, et al. EMT-like circulating tumor cells in ovarian cancer patients are enriched by platinum-based chemotherapy. Oncotarget. 2017;8:48820–31.
Obermayr E, Braicu EI, Polterauer S, Loverix L, Concin N, Woelber L, et al. Association of a combined cancer exhaustion score with circulating tumor cells and outcome in ovarian cancer—a study of the OVCAD Consortium. Cancers. 2021;13:5865.
Joosse SA, Pantel K. Genetic traits for hematogeneous tumor cell dissemination in cancer patients. Cancer Metastasis Rev. 2016;35:41–8.
Tan DS, Agarwal R, Kaye SB. Mechanisms of transcoelomic metastasis in ovarian cancer. Lancet Oncol. 2006;7:925–34.
Deng M, Yang R, Jiang J, Zhang J, He J, Miao J. The silent spread: exploring diverse metastatic pathways in high-grade serous ovarian cancer. Front Med (Lausanne). 2025;12:1539024.
Pradeep S, Kim SW, Wu SY, Nishimura M, Chaluvally-Raghavan P, Miyake T, et al. Hematogenous metastasis of ovarian cancer: rethinking mode of spread. Cancer cell. 2014;26:77–91.
Coffman LG, Burgos-Ojeda D, Wu R, Cho K, Bai S, Buckanovich RJ. New models of hematogenous ovarian cancer metastasis demonstrate preferential spread to the ovary and a requirement for the ovary for abdominal dissemination. Transl Res. 2016;175:92–102.e2.
Fan T, Zhao Q, Chen JJ, Chen WT, Pearl ML. Clinical significance of circulating tumor cells detected by an invasion assay in peripheral blood of patients with ovarian cancer. Gynecol Oncol. 2009;112:185–91.
Fehm T, Banys M, Rack B, Janni W, Marth C, Blassl C, et al. Pooled analysis of the prognostic relevance of disseminated tumor cells in the bone marrow of patients with ovarian cancer. Int J Gynecol Cancer. 2013;23:839–45.
Aktas B, Kasimir-Bauer S, Heubner M, Kimmig R, Wimberger P. Molecular profiling and prognostic relevance of circulating tumor cells in the blood of ovarian cancer patients at primary diagnosis and after platinum-based chemotherapy. Int J Gynecol Cancer. 2011;21:822–30.
Ponomarev AV, Shubina IZ. Insights into mechanisms of tumor and immune system interaction: association with wound healing. Front Oncol. 2019;9:1115.
Tai LH, de Souza CT, Belanger S, Ly L, Alkayyal AA, Zhang J, et al. Preventing postoperative metastatic disease by inhibiting surgery-induced dysfunction in natural killer cells. Cancer Res. 2013;73:97–107.
Lauber K, Ernst A, Orth M, Herrmann M, Belka C. Dying cell clearance and its impact on the outcome of tumor radiotherapy. Front Oncol. 2012;2:116.
Armstrong DK, Alvarez RD, Bakkum-Gamez JN, Barroilhet L, Behbakht K, Berchuck A, et al. Ovarian cancer, Version 2.2020, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw. 2021;19:191–226.
Vergote I, Trope CG, Amant F, Kristensen GB, Ehlen T, Johnson N, et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010;363:943–53.
Kehoe S, Hook J, Nankivell M, Jayson GC, Kitchener H, Lopes T, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet. 2015;386:249–57.
Mahner S, Heitz F, Salehi S, Reuss A, Guyon F, Du Bois A, et al. TRUST: trial of radical upfront surgical therapy in advanced ovarian cancer (ENGOT ov33/AGO-OVAR OP7). J Clin Oncol. 2025;43:Lba5500–Lba.
Filis P, Mauri D, Markozannes G, Tolia M, Filis N, Tsilidis K. Hyperthermic intraperitoneal chemotherapy (HIPEC) for the management of primary advanced and recurrent ovarian cancer: a systematic review and meta-analysis of randomized trials. ESMO Open. 2022;7:100586.
Della Corte L, Conte C, Palumbo M, Guerra S, Colacurci D, Riemma G, et al. Hyperthermic Intraperitoneal Chemotherapy (HIPEC): new approaches and controversies on the treatment of advanced epithelial ovarian cancer-systematic review and meta-analysis. J Clin Med. 2023;12:7012.
Lei Z, Wang Y, Wang J, Wang K, Tian J, Zhao Y, et al. Evaluation of cytoreductive surgery with or without hyperthermic intraperitoneal chemotherapy for stage III epithelial ovarian cancer. JAMA Netw Open. 2020;3:e2013940.
Acknowledgements
We would like to thank the patients for agreeing to participate in this study.
Funding
This work was supported by the Mildred Scheel Career Center HaTriCS4 (SAJ, KaP, MNH, AL) and University Cancer Center Hamburg (SAJ, JSS).
Author information
Authors and Affiliations
Contributions
Conceptualization: SAJ, KaP. Data curation: AL, JSS, MNH, PNR, MCW, PS. Formal analysis: AL, SAJ. Funding acquisition: SAJ, KaP. Investigation: AL, JSS, PNR, MCW, MNH, SL, SR, CC, SAJ. Methodology: AL, JSS, PNR, MCW, MNH, SL, SR, CC, SAJ. Project administration: SAJ, MNH, BS, LOF, LW, AJ, JSS, PNR, MCW, AL. Resources: SAJ, KaP, LOF, BS. Software: SAJ, AL. Supervision: SAJ, KaP, AL, BS. Validation: AL, SAJ, JSS, SR. Visualization: AL, SAJ. Writing - original draft: AL, SAJ. Writing - review and editing: AL, JSS, PNR, MCW, SL, CC, PS, AJ, LOF, LW, SR, KPa, BS, MNH, KaP, SAJ.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lobermeyer, A., Schöllhorn, J.S., Rademacher, P.N. et al. Pre-operative circulating tumor cells predict worse treatment outcome in patients with high-grade serous ovarian cancer. Oncogene (2026). https://doi.org/10.1038/s41388-026-03747-3
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41388-026-03747-3