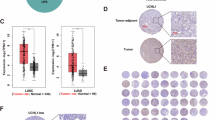
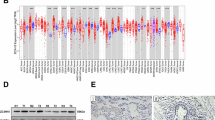
Abstract
Lung cancer persists as a major contributor to global cancer-related mortality, with metastasis, recurrence, and therapy resistance posing substantial barriers to effective disease management. CDYL has gained recognition as an epigenetic co-repressor involved in multiple dimensions of oncogenesis. However, its precise mechanistic contributions to non-small cell lung cancer (NSCLC) pathogenesis remain inadequately characterized. In this study, we observed pronounced CDYL overexpression in clinical NSCLC specimens, which exhibited a strong association with advanced disease staging and diminished patient survival. Functional profiling established that CDYL augments proliferative and migratory properties of NSCLC cells in vitro, whereas its genetic suppression markedly impaired tumor development in murine xenograft models. Mechanistically, we uncovered the deubiquitinating enzyme OTUB1 as a critical upstream effector that interacts with and stabilizes CDYL, thereby elevating its protein abundance. Further exploration demonstrated that CDYL confers cellular resistance to cuproptosis, a recently delineated copper-induced modality of regulated cell death. Through integrated transcriptomic and epigenomic interrogation, we elucidated that CDYL collaborates with EZH2 to promote H3K27me3 enrichment at the promoter region of the transcription factor SOX18, resulting in its transcriptional repression. Subsequent investigations revealed that SOX18 transcriptionally activates FDX1, a central regulator of cuproptosis. Consequently, CDYL-driven SOX18 repression leads to attenuated FDX1 expression, suppression of cuproptosis, and accelerated tumor progression. Importantly, administration of the copper chelator tetrathiomolybdate (TTM) counteracted the tumor-restraining consequences of CDYL ablation in vivo. Collectively, our findings unveil the OTUB1/CDYL/SOX18/FDX1 signaling cascade as a previously uncharacterized regulatory circuit that facilitates lung cancer progression through cuproptosis inhibition, providing new insights into the epigenetic regulation of cuproptosis and identifying potential therapeutic targets for NSCLC.

Proposed molecular mechanism: OTUB1-mediated deubiquitination stabilizes CDYL, which recruits EZH2 to deposit H3K27me3 at the SOX18 promoter, thereby repressing SOX18 expression and subsequent FDX1 transactivation, leading to cuproptosis suppression and lung cancer progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Data availability
Data will be made available on request.
References
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229–63.
Debieuvre D, Molinier O, Falchero L, Locher C, Templement-Grangerat D, Meyer N, et al. Lung cancer trends and tumor characteristic changes over 20 years (2000–2020): results of three French consecutive nationwide prospective cohorts’ studies. Lancet Regional Health. 2022;22:100492
Dohopolski M, Gottumukkala S, Gomez D, Iyengar P. Radiation therapy in non-small-cell lung cancer. Cold Spring Harbor Perspect Med. 2021;11:a037713
Li Y, Yan B, He S. Advances and challenges in the treatment of lung cancer. Biomed Pharmacother. 2023;169:115891.
Majeed U, Manochakian R, Zhao Y, Lou Y. Targeted therapy in advanced non-small cell lung cancer: current advances and future trends. J Hematol Oncol. 2021;14:108
Su P-L, Furuya N, Asrar A, Rolfo C, Li Z, Carbone DP, et al. Recent advances in therapeutic strategies for non-small cell lung cancer. J Hematol Oncol. 2025;18:35
Chen R, Manochakian R, James L, Azzouqa A-G, Shi H, Zhang Y, et al. Emerging therapeutic agents for advanced non-small cell lung cancer. J Hematol Oncol. 2020;13:58.
Lahn BT, Page DC. Retroposition of autosomal mRNA yielded testis-specific gene family on human Y chromosome. Nat Genet. 1999;21:429–33.
Shi Y, Sawada J, Sui G, Affar el B, Whetstine JR, Lan F, et al. Coordinated histone modifications mediated by a CtBP co-repressor complex. Nature. 2003;422:735–8.
Liu S, Yu H, Liu Y, Liu X, Zhang Y, Bu C, et al. Chromodomain protein CDYL acts as a crotonyl-CoA hydratase to regulate histone crotonylation and spermatogenesis. Mol Cell. 2017;67:853–66.e5.
Caron C, Pivot-Pajot C, van Grunsven LA, Col E, Lestrat C, Rousseaux S, et al. Cdyl: a new transcriptional co-repressor. EMBO Rep. 2003;4:877–82.
Liu Y, Lai S, Ma W, Ke W, Zhang C, Liu S, et al. CDYL suppresses epileptogenesis in mice through repression of axonal Nav1.6 sodium channel expression. Nat Commun. 2017;8:355.
Qin R, Cao S, Lyu T, Qi C, Zhang W, Wang Y. CDYL deficiency disrupts neuronal migration and increases susceptibility to epilepsy. Cell Rep. 2017;18:380–90.
Qi C, Liu S, Qin R, Zhang Y, Wang G, Shang Y, et al. Coordinated regulation of dendrite arborization by epigenetic factors CDYL and EZH2. J Neurosci. 2014;34:4494–508.
Mulligan P, Westbrook TF, Ottinger M, Pavlova N, Chang B, Macia E, et al. CDYL bridges REST and histone methyltransferases for gene repression and suppression of cellular transformation. Mol Cell. 2008;32:718–26.
Kong J-G, Mei Z, Zhang Y, Xu L-Z, Zhang J, Wang Y. CDYL knockdown reduces glioma development through an antitumor immune response in the tumor microenvironment. Cancer Lett. 2023;567:216265.
Cui Y, Zhao Y, Shen G, Lv Q, Ma L. CDYL loss promotes cervical cancer aggression by increasing PD-L1 expression via the suppression of IRF2BP2 transcription. Transl Oncol. 2024;47:102038.
Qiu Z, Zhu W, Meng H, Tong L, Li X, Luo P, et al. CDYL promotes the chemoresistance of small cell lung cancer by regulating H3K27 trimethylation at the CDKN1C promoter. Theranostics. 2019;9:4717–29.
Tsvetkov P, Coy S, Petrova B, Dreishpoon M, Verma A, Abdusamad M, et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022;375:1254–61.
Ge EJ, Bush AI, Casini A, Cobine PA, Cross JR, DeNicola GM, et al. Connecting copper and cancer: from transition metal signalling to metalloplasia. Nat Rev Cancer. 2021;22:102–13.
Lutsenko S. Human copper homeostasis: a network of interconnected pathways. Curr Opin Chem Biol. 2010;14:211–7.
Rae TD, Schmidt PJ, Pufahl RA, Culotta VC, O’Halloran TV. Undetectable intracellular free copper: the requirement of a copper chaperone for superoxide dismutase. Science. 1999;284:805–8.
Jawed R, Bhatti H. Cuproptosis in lung cancer: therapeutic options and prognostic models. Apoptosis. 2024;29:1393–8.
Kahlson MA, Dixon SJ. Copper-induced cell death. Science. 2022;375:1231–2.
Chen L, Min J, Wang F. Copper homeostasis and cuproptosis in health and disease. Signal Transduct Target Ther. 2022;7:378.
Xie J, Yang Y, Gao Y, He J. Cuproptosis: mechanisms and links with cancers. Mol Cancer. 2023;22:46.
Wang W, Wang X, Luo J, Chen X, Ma K, He H, et al. Serum copper level and the copper-to-zinc ratio could be useful in the prediction of lung cancer and its prognosis: a case-control study in Northeast China. Nutr Cancer. 2020;73:1908–15.
Saleh SAK, Adly HM, Abdelkhaliq AA, Nassir AM. Serum levels of selenium, zinc, copper, manganese, and iron in prostate cancer patients. Curr Urol. 2020;14:44–9.
Pavithra V, Sathisha TG, Kasturi K, Mallika DS, Amos SJ, Ragunatha S. Serum levels of metal ions in female patients with breast cancer. J Clin Diagn Res. 2015;9:BC25-c27.
Basu S, Singh MK, Singh TB, Bhartiya SK, Singh SP, Shukla VK. Heavy and Trace Metals in Carcinoma of the Gallbladder. World J Surg. 2013;37:2641–6.
Yaman M, Kaya G, Yekeler H. Distribution of trace metal concentrations in paired cancerous and non-cancerous human stomach tissues. World J Gastroenterol. 2007;13:612–8.
Kosova F, Cetin B, Akinci M, Aslan S, Seki A, Pirhan Y, et al. Serum copper levels in benign and malignant thyroid diseases. Bratisl Med J. 2012;113:718–20.
Xue R, Qin C, Li L, Huang L, Tang K, Chen J, et al. SRF/SLC31A1 signaling promotes cuproptosis induced by celastrol in NSCLC. Int Immunopharmacol. 2025;148:114165.
Xu H, Zhao Q, Cai D, Chen X, Zhou X, Gao Y, et al. o8G-modified circKIAA1797 promotes lung cancer development by inhibiting cuproptosis. J Exp Clin Cancer Res. 2025;44:110
Wang Y, Yao X, Lu Y, Ruan J, Yang Z, Wang C, et al. A PROTAC-based cuproptosis sensitizer in lung cancer therapy. Adv Mater. 2025;37:e2501435.
Li D, Wang Y, Dong C, Chen T, Dong A, Ren J, et al. CST1 inhibits ferroptosis and promotes gastric cancer metastasis by regulating GPX4 protein stability via OTUB1. Oncogene. 2022;42:83–98.
Liu T, Jiang L, Tavana O, Gu W. The deubiquitylase OTUB1 mediates ferroptosis via stabilization of SLC7A11. Cancer Res. 2019;79:1913–24.
Han X, Ren C, Lu C, Qiao P, Yang T, Yu Z. Deubiquitination of MYC by OTUB1 contributes to HK2 mediated glycolysis and breast tumorigenesis. Cell Death Differ. 2022;29:1864–73.
Li P, Sun Q, Bai S, Wang H, Zhao L. Combination of the cuproptosis inducer disulfiram and anti‑PD‑L1 abolishes NSCLC resistance by ATP7B to regulate the HIF‑1 signaling pathway. Int J Mol Med. 2023;53:19.
Zhao J, Zhang W, Zeng Y, Lu D, Ma C, Zhang M, et al. Targeting ATP7A-dependent copper metabolic homeostasis induces cuproptosis and suppresses the progression of mutant KRAS-driven lung cancer. Cancer Res. 2025;85:3999–4017.
Sun Z, Xu H, Lu G, Yang C, Gao X, Zhang J, et al. AKT1 phosphorylates FDX1 to promote cuproptosis resistance in triple-negative breast cancer. Adv Sci. 2025;12:e2408106.
Sun L, Zhang Y, Yang B, Sun S, Zhang P, Luo Z, et al. Lactylation of METTL16 promotes cuproptosis via m6A-modification on FDX1 mRNA in gastric cancer. Nat Commun. 2023;14:6523.
Grimm D, Bauer J, Wise P, Krüger M, Simonsen U, Wehland M, et al. The role of SOX family members in solid tumours and metastasis. Semin Cancer Biol. 2020;67:122–53.
Chen J, Dang Y, Feng W, Qiao C, Liu D, Zhang T, et al. SOX18 promotes gastric cancer metastasis through transactivating MCAM and CCL7. Oncogene. 2020;39:5536–52.
Chen J, Feng W, Sun M, Huang W, Wang G, Chen X, et al. TGF-β1-induced SOX18 elevation promotes hepatocellular carcinoma progression and metastasis through transcriptionally upregulating PD-L1 and CXCL12. Gastroenterology. 2024;167:264–80.
Ling J, Wang S, Yi C, Zheng X, Zhou Y, Lou S, et al. PRMT1-mediated modification of H4R3me2a promotes liver cancer progression by enhancing the transcriptional activity of SOX18. Hepatol Commun. 2025;9:e0647.
Geng Q, Deng H, Fu J, Cui F. SOX18 exerts tumor-suppressive functions in papillary thyroid carcinoma through inhibition of Wnt/á-catenin signaling. Exp Cell Res. 2020;396:112249.
Funding
This study was supported by grants from the National Natural Science Foundation of China grants (Grant No. 82172351, Grant No. 82173018), and was sponsored by Natural Science Foundation of Henan Province (Grant No.232300421054), the Medical Science and Technology Provincial and Ministerial Co-construction Project of Henan province (Grant No. SBGJ202403034), the Henan Province Science and Technology Research and Development Joint Fund (242301420071).
Author information
Authors and Affiliations
Contributions
RZ, HYL, JW, and HG designed this study. RZ, YL, HSL, XW, YZ, and XZ performed the experiments. RZ, YL, KJ, LM, and ZX performed the data analysis. YL and JW reviewed the pathological data. RZ wrote the draft of the manuscript. YL, HG, and JW reviewed the manuscript. The authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was performed according to the ethical standards of the Declaration of Helsinki and received approval from the Ethics Committee at the First Affiliated Hospital of Zhengzhou University. Informed consent was obtained from all patients participating in this research.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, R., Li, Y., Lu, H. et al. OTUB1/CDYL axis-mediated epigenetic repression of SOX18 facilitates lung cancer progression by inhibiting FDX1-dependent cuproptosis. Oncogene (2026). https://doi.org/10.1038/s41388-026-03748-2
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41388-026-03748-2