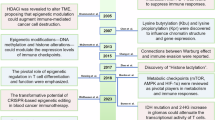
Abstract
Cancer remains a leading cause of human mortality worldwide, imposing a substantial public health burden. A deep understanding of the tumor microenvironment (TME) is essential for improving cancer care. Erythroid progenitor cells (EPCs) were traditionally viewed solely as intermediates in erythropoiesis; however, growing evidence indicates their active involvement in cancer progression and immune evasion. Research on EPCs increasingly utilizes omics sequencing technologies. Multi-omics strategies in particular enable in-depth investigation of the functional mechanisms of EPCs and their interactions with tumor and immune cells. This review examines various omics methodologies applied to EPCs from an oncology perspective, including transcriptomics, proteomics, epigenomics, and metabolomics, while critically assessing the advantages and limitations of each approach. Furthermore, it synthesizes how the integration of multiple omics technologies provides a more comprehensive view of EPC biology, particularly through complementary data modalities. This review also discusses artificial intelligence (AI)-powered multi-omics integration strategies and explore the translational potential of EPC-focused research in advancing cancer therapeutics from bench to bedside.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Siegel RL, Kratzer TB, Giaquinto AN, Sung H, Jemal A. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10–45.
Filho AM, Laversanne M, Ferlay J, Colombet M, Pineros M, Znaor A, et al. The GLOBOCAN 2022 cancer estimates: Data sources, methods, and a snapshot of the cancer burden worldwide. Int J Cancer. 2025;156:1336–46.
Arafat Hossain M. A comprehensive review of immune checkpoint inhibitors for cancer treatment. Int Immunopharmacol. 2024;143:113365.
Wagle NS, Nogueira L, Devasia TP, Mariotto AB, Yabroff KR, Islami F, et al. Cancer treatment and survivorship statistics, 2025. CA Cancer J Clin. 2025;75:308–40.
Zhang C, Zhang C, Wang H. Immune-checkpoint inhibitor resistance in cancer treatment: Current progress and future directions. Cancer Lett. 2023;562:216182.
Tang T, Huang X, Zhang G, Hong Z, Bai X, Liang T. Advantages of targeting the tumor immune microenvironment over blocking immune checkpoint in cancer immunotherapy. Signal Transduct Target Ther. 2021;6:72.
Li ZZ, Zhou K, Wu Q, Liu B, Bu LL. Lymph node metastasis in cancer: clearing the clouds to see the dawn. Crit Rev Oncol Hematol. 2024;204:104536.
Bader JE, Voss K, Rathmell JC. Targeting metabolism to improve the tumor microenvironment for cancer immunotherapy. Mol Cell. 2020;78:1019–33.
Li ZZ, Wu TF, Sun ZJ. Harnessing cuproptosis resistance to advance cancer therapeutics. Apoptosis. 2026;31:50.
Li SR, Wu ZZ, Yu HJ, Sun ZJ. Targeting erythroid progenitor cells for cancer immunotherapy. Int J Cancer. 2024;155:1928–38.
He X, Liu X, Zuo F, Shi H, Jing J. Artificial intelligence-based multi-omics analysis fuels cancer precision medicine. Semin Cancer Biol. 2023;88:187–200.
Szeto GL, Finley SD. Integrative approaches to cancer immunotherapy. Trends Cancer. 2019;5:400–10.
Donisi C, Pretta A, Pusceddu V, Ziranu P, Lai E, Puzzoni M, et al. Immunotherapy and cancer: the multi-omics perspective. Int J Mol Sci. 2024;25:3563.
Hernandez-Lemus E, Ochoa S. Methods for multi-omic data integration in cancer research. Front Genet. 2024;15:1425456.
Wang YY, Wu ZZ, Huang CF, Sun ZJ. Tumor-host colluding through erythroid progenitor cells: Mechanisms and opportunities. Cancer Lett. 2023;563:216193.
Tang P, Wang H. Regulation of erythropoiesis: emerging concepts and therapeutic implications. Hematology. 2023;28:2250645.
Schippel N, Sharma S. Dynamics of human hematopoietic stem and progenitor cell differentiation to the erythroid lineage. Exp Hematol. 2023;123:1–17.
Nandakumar SK, Ulirsch JC, Sankaran VG. Advances in understanding erythropoiesis: evolving perspectives. Br J Haematol. 2016;173:206–18.
Hewitt KJ, Johnson KD, Gao X, Keles S, Bresnick EH. The hematopoietic stem and progenitor cell cistrome: GATA factor-dependent Cis-regulatory mechanisms. Curr Top Dev Biol. 2016;118:45–76.
Sincennes MC, Humbert M, Grondin B, Lisi V, Veiga DF, Haman A, et al. The LMO2 oncogene regulates DNA replication in hematopoietic cells. Proc Natl Acad Sci USA. 2016;113:1393–8.
Wang C, Hu M, Yu K, Liu W, Hu A, Kuang Y, et al. An intricate regulatory circuit between FLI1 and GATA1/GATA2/LDB1/ERG dictates erythroid vs. megakaryocytic differentiation. Mol Med Rep. 2024;29:107.
Adlung L, Stapor P, Tonsing C, Schmiester L, Schwarzmuller LE, Postawa L, et al. Cell-to-cell variability in JAK2/STAT5 pathway components and cytoplasmic volumes defines survival threshold in erythroid progenitor cells. Cell Rep. 2021;36:109507.
Feng Y, Borosha S, Ratri A, Lee EB, Wang H, Fields TA, et al. DOT1L methyltransferase regulates calcium influx in erythroid progenitor cells in response to erythropoietin. Int J Mol Sci. 2022;23:5137.
Li J, Hale J, Bhagia P, Xue F, Chen L, Jaffray J, et al. Isolation and transcriptome analyses of human erythroid progenitors: BFU-E and CFU-E. Blood. 2014;124:3636–45.
Barisas DAG, Choi K. Extramedullary hematopoiesis in cancer. Exp Mol Med. 2024;56:549–58.
Zhao L, He R, Long H, Guo B, Jia Q, Qin D, et al. Late-stage tumors induce anemia and immunosuppressive extramedullary erythroid progenitor cells. Nat Med. 2018;24:1536–44.
Chen Z, Cheng X, Yang L, Cheng X, Zhu B, Long H. Mechanism and effects of extramedullary hematopoiesis on anti-tumor immunity. Cancer Biol Med. 2023;20:477–82.
Li ZZ, Su XY, Zhou CK, Li SR, Sun ZJ. Epigenetic reprogramming of erythroid progenitor cells: insights for enhancing cancer immunotherapy. Int J Biol Sci. 2026;22:1997–2011.
Long H, Jia Q, Wang L, Fang W, Wang Z, Jiang T, et al. Tumor-induced erythroid precursor-differentiated myeloid cells mediate immunosuppression and curtail anti-PD-1/PD-L1 treatment efficacy. Cancer Cell. 2022;40:674–693 e677.
Wu ZZ, Deng WW, Zhu SW, Wang WD, Wang S, Yang QC, et al. Erythroid progenitor cell-mediated spleen-tumor interaction deteriorates cancer immunity. Proc Natl Acad Sci USA. 2025;122:e2417473122.
Han Y, Liu Q, Hou J, Gu Y, Zhang Y, Chen Z, et al. Tumor-induced generation of splenic erythroblast-like Ter-cells promotes tumor progression. Cell. 2018;173:634–648.
Mamand DR, Bazaz S, Mohammad DK, Liang X, Pavlova S, Mim C, et al. Extracellular vesicles originating from melanoma cells promote dysregulation in haematopoiesis as a component of cancer immunoediting. J Extracell Vesicles. 2024;13:e12471.
Mo WT, Huang CF, Sun ZJ. Erythroid progenitor cell modulates cancer immunity: insights and implications. Biochim Biophys Acta Rev Cancer. 2024;1879:189209.
Ediriwickrema A, Nakauchi Y, Fan AC, Kohnke T, Hu X, Luca BA, et al. A single-cell framework identifies functionally and molecularly distinct multipotent progenitors in adult human hematopoiesis. Cell Rep. 2025;44:116236.
Song Q, Liu L. Single-cell RNA-Seq technologies and computational analysis tools: application in cancer research. Methods Mol Biol. 2022;2413:245–55.
Zhang Y, Wang D, Peng M, Tang L, Ouyang J, Xiong F, et al. Single-cell RNA sequencing in cancer research. J Exp Clin Cancer Res. 2021;40:81.
Wen JY, Li X, Chen JN, Chen J, Zhang JY, Du Y, et al. CD45(-) erythroid progenitor cells promote lymph node metastasis in gastric cancer by inducing a hybrid epithelial/mesenchymal state in lymphatic endothelial cells. Gastric Cancer. 2023;26:918–33.
Chang X, Zheng Y, Xu K. Single-cell RNA sequencing: technological progress and biomedical application in cancer research. Mol Biotechnol. 2024;66:1497–519.
Conboy JG. RNA splicing during terminal erythropoiesis. Curr Opin Hematol. 2017;24:215–21.
Tusi BK, Wolock SL, Weinreb C, Hwang Y, Hidalgo D, Zilionis R, et al. Population snapshots predict early haematopoietic and erythroid hierarchies. Nature. 2018;555:54–60.
Xu C, He J, Wang H, Zhang Y, Wu J, Zhao L, et al. Single-cell transcriptomic analysis identifies an immune-prone population in erythroid precursors during human ontogenesis. Nat Immunol. 2022;23:1109–20.
Weiler P, Van den Berge K, Street K, Tiberi S. A guide to trajectory inference and RNA velocity. Methods Mol Biol. 2023;2584:269–92.
Ranzoni AM, Tangherloni A, Berest I, Riva SG, Myers B, Strzelecka PM, et al. Integrative single-cell RNA-Seq and ATAC-seq analysis of human developmental hematopoiesis. Cell Stem Cell. 2021;28:472–87.
Furtwangler B, Uresin N, Richter S, Schuster MB, Barmpouri D, Holze H, et al. Mapping early human blood cell differentiation using single-cell proteomics and transcriptomics. Science. 2025;390:eadr8785.
Trapnell C, Cacchiarelli D, Grimsby J, Pokharel P, Li S, Morse M, et al. The dynamics and regulators of cell fate decisions are revealed by pseudotemporal ordering of single cells. Nat Biotechnol. 2014;32:381–6.
Saelens W, Cannoodt R, Todorov H, Saeys Y. A comparison of single-cell trajectory inference methods. Nat Biotechnol. 2019;37:547–54.
Christodoulou MI, Zaravinos A single-cell analysis in immuno-oncology. Int J Mol Sci. 2023;24:8422.
Bai Z, Su G, Fan R. Single-cell analysis technologies for immuno-oncology research: from mechanistic delineation to biomarker discovery. Genomics Proteom Bioinforma. 2021;19:191–207.
Hou X, Wang YL, Shi W, Hu W, Zeng Z, Liu J, et al. Multiplexed analysis of gene expression and chromatin accessibility of human umbilical cord blood using scRNA-Seq and scATAC-Seq. Mol Immunol. 2022;152:207–14.
Luo C, Rivkin A, Zhou J, Sandoval JP, Kurihara L, Lucero J, et al. Robust single-cell DNA methylome profiling with snmC-seq2. Nat Commun. 2018;9:3824.
Nam AS, Dusaj N, Izzo F, Murali R, Myers RM, Mouhieddine TH, et al. Single-cell multi-omics of human clonal hematopoiesis reveals that DNMT3A R882 mutations perturb early progenitor states through selective hypomethylation. Nat Genet. 2022;54:1514–26.
Kaya-Okur HS, Janssens DH, Henikoff JG, Ahmad K, Henikoff S. Efficient low-cost chromatin profiling with CUT&Tag. Nat Protoc. 2020;15:3264–83.
Kaya-Okur HS, Wu SJ, Codomo CA, Pledger ES, Bryson TD, Henikoff JG, et al. CUT&Tag for efficient epigenomic profiling of small samples and single cells. Nat Commun. 2019;10:1930.
Deng Y, Bartosovic M, Kukanja P, Zhang D, Liu Y, Su G, et al. Spatial-CUT&Tag: Spatially resolved chromatin modification profiling at the cellular level. Science. 2022;375:681–6.
Wu SJ, Furlan SN, Mihalas AB, Kaya-Okur HS, Feroze AH, Emerson SN, et al. Single-cell CUT&Tag analysis of chromatin modifications in differentiation and tumor progression. Nat Biotechnol. 2021;39:819–24.
Park PJ. ChIP-seq: advantages and challenges of a maturing technology. Nat Rev Genet. 2009;10:669–80.
Chen Y, Huo D, Meng Y, Zhang J, Huang M, Luo Q, et al. BRD4 acts as a transcriptional repressor of RhoB to inhibit terminal erythropoiesis. J Hematol Oncol. 2025;18:67.
Gautier EF, Ducamp S, Leduc M, Salnot V, Guillonneau F, Dussiot M, et al. Comprehensive proteomic analysis of human erythropoiesis. Cell Rep. 2016;16:1470–84.
Brand M, Ranish JA. Proteomic/transcriptomic analysis of erythropoiesis. Curr Opin Hematol. 2021;28:150–7.
Grzywa TM, Justyniarska M, Nowis D, Golab J. Tumor immune evasion induced by dysregulation of erythroid progenitor cells development. Cancers. 2021;13:870.
Hou Y, Liang HL, Yu X, Liu Z, Cao X, Rao E, et al. Radiotherapy and immunotherapy converge on elimination of tumor-promoting erythroid progenitor cells through adaptive immunity. Sci Transl Med. 2021;13:130.
Sasaki S, Inoguchi T, Muta K, Abe Y, Zhang M, Hiasa K, et al. Therapeutic angiogenesis by ex vivo expanded erythroid progenitor cells. Am J Physiol Heart Circ Physiol. 2007;292:H657–665.
Karayel O, Xu P, Bludau I, Velan Bhoopalan S, Yao Y, Ana Rita FC, et al. Integrative proteomics reveals principles of dynamic phosphosignaling networks in human erythropoiesis. Mol Syst Biol. 2020;16:e9813.
Danzi F, Pacchiana R, Mafficini A, Scupoli MT, Scarpa A, Donadelli M, et al. To metabolomics and beyond: a technological portfolio to investigate cancer metabolism. Signal Transduct Target Ther. 2023;8:137.
Schmidt DR, Patel R, Kirsch DG, Lewis CA, Vander Heiden MG, Locasale JW. Metabolomics in cancer research and emerging applications in clinical oncology. CA Cancer J Clin. 2021;71:333–58.
Fan X, Peng H, Wang X, Sun Y, Dong Y, Zhou J, et al. Tumor-associated CD8(+)T cell tolerance induced by erythroid progenitor cells. Front Immunol. 2024;15:1381919.
Argelaguet R, Arnol D, Bredikhin D, Deloro Y, Velten B, Marioni JC, et al. MOFA+: a statistical framework for comprehensive integration of multi-modal single-cell data. Genome Biol. 2020;21:111.
Ovaska K, Laakso M, Haapa-Paananen S, Louhimo R, Chen P, Aittomaki V, et al. Large-scale data integration framework provides a comprehensive view on glioblastoma multiforme. Genome Med. 2010;2:65.
Schmidt D, Endres C, Hoefflin R, Andrieux G, Zwick M, Karantzelis N, et al. Oncogenic calreticulin induces immune escape by stimulating TGFbeta expression and regulatory T-cell expansion in the bone marrow microenvironment. Cancer Res. 2024;84:2985–3003.
Sarachakov A, Varlamova A, Svekolkin V, Polyakova M, Valencia I, Unkenholz C, et al. Spatial mapping of human hematopoiesis at single-cell resolution reveals aging-associated topographic remodeling. Blood. 2023;142:2282–95.
Jackson C, Cherry C, Bom S, Dykema AG, Wang R, Thompson E, et al. Distinct myeloid-derived suppressor cell populations in human glioblastoma. Science. 2025;387:eabm5214.
Han X, Wang B, Situ C, Qi Y, Zhu H, Li Y, et al. scapGNN: A graph neural network-based framework for active pathway and gene module inference from single-cell multi-omics data. PLoS Biol. 2023;21:e3002369.
Baek M, DiMaio F, Anishchenko I, Dauparas J, Ovchinnikov S, Lee GR, et al. Accurate prediction of protein structures and interactions using a three-track neural network. Science. 2021;373:871–6.
Baek M, McHugh R, Anishchenko I, Jiang H, Baker D, DiMaio F. Accurate prediction of protein-nucleic acid complexes using RoseTTAFoldNA. Nat Methods. 2024;21:117–21.
Kourou K, Exarchos TP, Exarchos KP, Karamouzis MV, Fotiadis DI. Machine learning applications in cancer prognosis and prediction. Comput Struct Biotechnol J. 2015;13:8–17.
Ahmed R, Augustine R, Valera E, Ganguli A, Mesaeli N, Ahmad IS, et al. Spatial mapping of cancer tissues by OMICS technologies. Biochim Biophys Acta Rev Cancer. 2022;1877:188663.
Zhou WM, Yan YY, Guo QR, Ji H, Wang H, Xu TT, et al. Microfluidics applications for high-throughput single cell sequencing. J Nanobiotechnol. 2021;19:312.
Li ZZ, Wei LY, Cao LM, Wang GR, Luo HY, Zhou K, et al. Emerging roles of microfluidics in oral cancer research and clinical translation. Mater Today Bio. 2026;37:102801.
Lin S, Liu Y, Zhang M, Xu X, Chen Y, Zhang H, et al. Microfluidic single-cell transcriptomics: moving towards multimodal and spatiotemporal omics. Lab Chip. 2021;21:3829–49.
Tyson JJ, Chen K, Novak B. Network dynamics and cell physiology. Nat Rev Mol Cell Biol. 2001;2:908–16.
Stahlberg EA, Abdel-Rahman M, Aguilar B, Asadpoure A, Beckman RA, Borkon LL, et al. Exploring approaches for predictive cancer patient digital twins: opportunities for collaboration and innovation. Front Digit Health. 2022;4:1007784.
Bjornsson B, Borrebaeck C, Elander N, Gasslander T, Gawel DR, Gustafsson M, et al. Digital twins to personalize medicine. Genome Med. 2019;12:4.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82273202); the Fundamental Research Funds for the Central Universities (2042022dx0003).
Author information
Authors and Affiliations
Contributions
Zi-Zhan Li: Writing – review & editing, Writing – original draft, Visualization, Validation, Software, Resources, Investigation. Cheng-Ke Zhou: Writing – original draft, Visualization, Software, Conceptualization, Writing – review & editing. Zhi-Jun Sun: Writing – review & editing, Conceptualization, Supervision, Investigation, Funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, ZZ., Zhou, CK. & Sun, ZJ. Multi-omics approaches reveal erythroid progenitor cell in cancer: from passive bystander to active player. Oncogene (2026). https://doi.org/10.1038/s41388-026-03758-0
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41388-026-03758-0