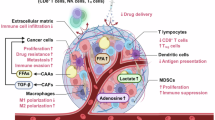
Abstract
Solid tumors arise within hostile microenvironments shaped by hypoxia, nutrient deprivation, acidosis, mechanical stress, immune pressure, and therapy-induced insults. Beyond malignant cells, stromal and immune components, including cancer-associated fibroblasts, endothelial cells, and infiltrating leukocytes, co-evolve with tumor cells to regulate tumor initiation, progression, and therapeutic response. Rather than passively tolerating stress, tumor cells actively reprogram signaling and transcriptional networks, frequently through the induction of stress-responsive membrane proteins. These proteins are typically low or absent under homeostatic conditions but are upregulated by hypoxic, metabolic, inflammatory, or therapeutic stress. By coordinating nutrient uptake, metabolite exchange, cell-matrix interactions, survival signaling, invasion, and immune evasion, stress-responsive membrane proteins function as critical execution nodes that support tumor stress tolerance and evolutionary fitness. In this Review, we summarize major microenvironmental stresses in solid tumors, delineate membrane protein-mediated stress-adaptation mechanisms, and discuss clinically approved and emerging strategies targeting stress-responsive membrane proteins to enhance treatment efficacy and overcome therapeutic resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
de Visser KE, Joyce JA. The evolving tumor microenvironment: from cancer initiation to metastatic outgrowth. Cancer Cell. 2023;41:374–403.
Hanahan D, Coussens LM. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell. 2012;21:309–22.
Sahai E, Astsaturov I, Cukierman E, DeNardo DG, Egeblad M, Evans RM, et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer. 2020;20:174–86.
Huang J, Zhang L, Wan D, Zhou L, Zheng S, Lin S, et al. Extracellular matrix and its therapeutic potential for cancer treatment. Signal Transduct Target Ther. 2021;6:153.
Hinshaw DC, Shevde LA. The tumor microenvironment innately modulates cancer progression. Cancer Res. 2019;79:4557–66.
Chen Z, Han F, Du Y, Shi H, Zhou W. Hypoxic microenvironment in cancer: molecular mechanisms and therapeutic interventions. Signal Transduction Targeted Ther. 2023;8:70.
Corbet C, Feron O. Tumour acidosis: from the passenger to the driver’s seat. Nat Rev Cancer. 2017;17:577–93.
Cheng H, Qiu Y, Xu Y, Chen L, Ma K, Tao M, et al. Extracellular acidosis restricts one-carbon metabolism and preserves T cell stemness. Nat Metab. 2023;5:314–30.
Chang C-H, Qiu J, O’Sullivan D, Buck MD, Noguchi T, Curtis JD, et al. Metabolic competition in the tumor microenvironment is a driver of cancer progression. Cell. 2015;162:1229–41.
Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005;307:58–62.
Salgia R, Kulkarni P. The genetic/non-genetic duality of drug ‘resistance’ in cancer. Trends Cancer. 2018;4:110–8.
Hanahan D. Hallmarks of cancer: new dimensions. Cancer Discov. 2022;12:31–46.
Kim J, Tchernyshyov I, Semenza GL, Dang CV. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 2006;3:177–85.
Forsythe JA, Jiang BH, Iyer NV, Agani F, Leung SW, Koos RD, et al. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol Cell Biol. 1996;16:4604–13.
Jiang X-Y, Guo Q-Q, Wang S-S, Guo R, Zou Y, Liu J-W, et al. DNA damage response pathway regulates Nrf2 in response to oxidative stress. Sci Adv. 2025;11:eadu9555.
Alam S, Doherty E, Ortega-Prieto P, Arizanova J, Fets L. Membrane transporters in cell physiology, cancer metabolism and drug response. Dis Model Mech. 2023;16:dmm050404.
Yu Y, Sun R, Hu F, Ding Z, Li X, Han J, et al. Hypoxia upregulates the expression of PD-L1 via NPM1 in breast cancer. J Immunother Cancer. 2025;13:e010151.
Godet I, Oza HH, Shi Y, Joe NS, Weinstein AG, Johnson J, et al. Hypoxia induces ROS-resistant memory upon reoxygenation in vivo promoting metastasis in part via MUC1-C. Nat Commun. 2024;15:8416.
Nam A, Jain S, Wu C, Campos A, Shepard RM, Yu Z, et al. Integrin αvβ3 upregulation in response to nutrient stress promotes lung cancer cell metabolic plasticity. Cancer Res. 2024;84:1630–42.
Holzgruber J, Martins C, Kulcsar Z, Duplaine A, Rasbach E, Migayron L, et al. Type I interferon signaling induces melanoma cell-intrinsic PD-1 and its inhibition antagonizes immune checkpoint blockade. Nat Commun. 2024;15:7165.
Hwang HJ, Kang D, Shin J, Jung J, Ko S, Jung KH, et al. Therapy-induced senescent cancer cells contribute to cancer progression by promoting ribophorin 1-dependent PD-L1 upregulation. Nat Commun. 2025;16:353.
Wu C, Weis SM, Cheresh DA. Tumor-initiating cells establish a niche to overcome isolation stress. Trends Cell Biol. 2024;34:380–7.
Wu C, Rakhshandehroo T, Wettersten HI, Campos A, von Schalscha T, Jain S, et al. Pancreatic cancer cells upregulate LPAR4 in response to isolation stress to promote an ECM-enriched niche and support tumour initiation. Nat Cell Biol. 2023;25:309–22.
Bakırdöğen D, Görgülü K, Xin J, Richter L, Alcalá S, Ruiz-Cañas L, et al. c-rel drives pancreatic cancer metastasis through fibronectin-integrin signaling-induced isolation stress resistance and EMT. Mol Cancer. 2025;25:16.
Chen X, Song E. The theory of tumor ecosystem. Cancer Commun. 2022;42:587–608.
Munne PM, Martikainen L, Räty I, Bertula K, Nonappa J, Ruuska, et al. Compressive stress-mediated p38 activation required for ERα + phenotype in breast cancer. Nat Commun. 2021;12:6967.
Guzelsoy G, Elorza SD, Ros M, Schachtner LT, Hayashi M, Hobson-Gutierrez S, et al. Cooperative nutrient scavenging is an evolutionary advantage in cancer. Nature. 2025;640:534–42.
Michl J, Monterisi S, White B, Blaszczak W, Hulikova A, Abdullayeva G, et al. Acid-adapted cancer cells alkalinize their cytoplasm by degrading the acid-loading membrane transporter anion exchanger 2, SLC4A2. Cell Rep. 2023;42:112601.
Oren Y, Tsabar M, Cuoco MS, Amir-Zilberstein L, Cabanos HF, Hütter J-C, et al. Cycling cancer persister cells arise from lineages with distinct programs. Nature. 2021;596:576–82.
Isozaki H, Sakhtemani R, Abbasi A, Nikpour N, Stanzione M, Oh S, et al. Therapy-induced APOBEC3A drives evolution of persistent cancer cells. Nature. 2023;620:393–401.
Luo Y-W, Fang Y, Zeng H-X, Ji Y-C, Wu M-Z, Li H, et al. HIF1α counteracts TGFβ1-driven TSP1 expression in endothelial cells to stimulate angiogenesis in the hypoxic tumor microenvironment. Cancer Res. 2025;85:69–83.
Yang Y, Zuo Q, Ramu V, Talwar V, Lyu Y, Chen C, et al. HIF-1-mediated ISG20 expression promotes breast cancer stemness and immune evasion. J Exp Med. 2026;223:e20250777.
Cognet G, Muir A. Identifying metabolic limitations in the tumor microenvironment. Sci Adv. 2024;10:eadq7305.
Wang Z, Shang J, Qiu Y, Cheng H, Tao M, Xie E, et al. Suppression of the METTL3-m6A-integrin β1 axis by extracellular acidification impairs T cell infiltration and antitumor activity. Cell Rep. 2024;43:113796.
Enjalbert R, Krüger T, Bernabeu MO. Effect of vessel compression on blood flow in microvascular networks and its implications for tumour tissue hypoxia. Commun Phys. 2024;7:49.
Weeden CE, Gayevskiy V, Marceaux C, Batey D, Tan T, Yokote K, et al. Early immune pressure initiated by tissue-resident memory T cells sculpts tumor evolution in non-small cell lung cancer. Cancer Cell. 2023;41:837–52.e6.
Lior C, Barki D, Halperin C, Iacobuzio-Donahue CA, Kelsen D, Shouval RS-. Mapping the tumor stress network reveals dynamic shifts in the stromal oxidative stress response. Cell Rep. 2024;43:114236.
Harris AL. Hypoxia-a key regulatory factor in tumour growth. Nat Rev Cancer. 2002;2:38–47.
Wang GL, Jiang BH, Rue EA, Semenza GL. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci USA. 1995;92:5510–4.
Maxwell PH, Dachs GU, Gleadle JM, Nicholls LG, Harris AL, Stratford IJ, et al. Hypoxia-inducible factor-1 modulates gene expression in solid tumors and influences both angiogenesis and tumor growth. Proc Natl Acad Sci USA. 1997;94:8104–9.
Robey IF, Lien AD, Welsh SJ, Baggett BK, Gillies RJ. Hypoxia-inducible factor-1alpha and the glycolytic phenotype in tumors. Neoplasia. 2005;7:324–30.
Kim I, Choi S, Yoo S, Lee M, Kim I-S. Cancer-associated fibroblasts in the hypoxic tumor microenvironment. Cancers. 2022;14:3321.
Wang B, Zhao Q, Zhang Y, Liu Z, Zheng Z, Liu S, et al. Targeting hypoxia in the tumor microenvironment: a potential strategy to improve cancer immunotherapy. J Exp Clin Cancer Res. 2021;40:24.
Ciepła J, Smolarczyk R. Tumor hypoxia unveiled: Insights into microenvironment, detection tools and emerging therapies. Clin Exp Med. 2024;24:235.
Harris B, Saleem S, Cook N, Searle E. Targeting hypoxia in solid and haematological malignancies. J Exp Clin Cancer Res. 2022;41:318.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74.
Carmeliet P, Jain RK. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011;473:298–307.
Park J-M, Lee D-H, Kim D-H. Redefining the role of AMPK in autophagy and the energy stress response. Nat Commun. 2023;14:2994.
Li X, Yang Y, Zhang B, Lin X, Fu X, An Y, et al. Lactate metabolism in human health and disease. Signal Transduction Targeted Ther. 2022;7:305.
Chen J, Huang Z, Chen Y, Tian H, Chai P, Shen Y, et al. Lactate and lactylation in cancer. Signal Transduction Targeted Ther. 2025;10:38.
García-Cañaveras JC, Chen L, Rabinowitz JD. The tumor metabolic microenvironment: lessons from lactate. Cancer Res. 2019;79:3155–62.
Doherty JR, Yang C, Scott KEN, Cameron MD, Fallahi M, Li W, et al. Blocking lactate export by inhibiting the myc target MCT1 disables glycolysis and glutathione synthesis. Cancer Res. 2014;74:908–20.
Chiche J, Ilc K, Laferrière J, Trottier E, Dayan F, Mazure NM, et al. Hypoxia-inducible carbonic anhydrase IX and XII promote tumor cell growth by counteracting acidosis through the regulation of the intracellular pH. Cancer Res. 2009;69:358–68.
Boedtkjer E, Pedersen SF. The acidic tumor microenvironment as a driver of cancer. Annu Rev Physiol. 2020;82:103–26.
Jain RK, Martin JD, Stylianopoulos T. The role of mechanical forces in tumor growth and therapy. Annu Rev Biomed Eng. 2014;16:321–46.
Helmlinger G, Netti PA, Lichtenbeld HC, Melder RJ, Jain RK. Solid stress inhibits the growth of multicellular tumor spheroids. Nat Biotechnol. 1997;15:778–83.
Stylianopoulos T, Martin JD, Snuderl M, Mpekris F, Jain SR, Jain RK. Coevolution of solid stress and interstitial fluid pressure in tumors during progression: implications for vascular collapse. Cancer Res. 2013;73:3833–41.
Meli VS, Veerasubramanian PK, Downing TL, Wang W, Liu WF. Mechanosensation to inflammation: roles for YAP/TAZ in innate immune cells. Sci Signal. 2023;16:eadc9656.
Brusatin G, Panciera T, Gandin A, Citron A, Piccolo S. Biomaterials and engineered microenvironments to control YAP/TAZ-dependent cell behaviour. Nat Mater. 2018;17:1063–75.
Dupont S, Morsut L, Aragona M, Enzo E, Giulitti S, Cordenonsi M, et al. Role of YAP/TAZ in mechanotransduction. Nature. 2011;474:179–83.
Gao J, He L, Zhou L, Jing Y, Wang F, Shi Y, et al. Mechanical force regulation of YAP by F-actin and GPCR revealed by super-resolution imaging. Nanoscale. 2020;12:2703–14.
Seo J, Kim J. Regulation of hippo signaling by actin remodeling. BMB Rep. 2018;51:151–6.
Mo J-S, Meng Z, Kim YC, Park HW, Hansen CG, Kim S, et al. Cellular energy stress induces AMPK-mediated regulation of YAP and the hippo pathway. Nat Cell Biol. 2015;17:500–10.
Kurosawa S, Harada M, Matsuzaki G, Shinomiya Y, Terao H, Kobayashi N, et al. Early-appearing tumour-infiltrating natural killer cells play a crucial role in the generation of anti-tumour T lymphocytes. Immunology. 1995;85:338–46.
Seliger B, Harders C, Wollscheid U, Staege MS, Reske-Kunz AB, Huber C. Suppression of MHC class I antigens in oncogenic transformants: association with decreased recognition by cytotoxic T lymphocytes. Exp Hematol. 1996;24:1275–9.
Dunn GP, Old LJ, Schreiber RD. The three es of cancer immunoediting. Annu Rev Immunol. 2004;22:329–60.
Yang J, Tang S, Saba NF, Shay C, Teng Y. Tumor secretome shapes the immune landscape during cancer progression. J Exp Clin Cancer Res. 2025;44:47.
Jenkins L, Jungwirth U, Avgustinova A, Iravani M, Mills A, Haider S, et al. Cancer-associated fibroblasts suppress CD8+ T-cell infiltration and confer resistance to immune-checkpoint blockade. Cancer Res. 2022;82:2904–17.
Mai Z, Lin Y, Lin P, Zhao X, Cui L. Modulating extracellular matrix stiffness: A strategic approach to boost cancer immunotherapy. Cell Death Dis. 2024;15:307.
Mpekris F, Panagi M, Charalambous A, Voutouri C, Stylianopoulos T. Modulating cancer mechanopathology to restore vascular function and enhance immunotherapy. Cell Rep Med. 2024;5:101626.
Bukowski K, Kciuk M, Kontek R. Mechanisms of multidrug resistance in cancer chemotherapy. Int J Mol Sci. 2020;21:3233.
Park SS, Choi YW, Kim J-H, Kim HS, Park TJ. Senescent tumor cells: an overlooked adversary in the battle against cancer. Exp Mol Med. 2021;53:1834–41.
Zhang D, Zhang J-W, Xu H, Chen X, Gao Y, Jiang H-G, et al. Therapy-induced senescent tumor cell-derived extracellular vesicles promote colorectal cancer progression through SERPINE1-mediated NF-κB p65 nuclear translocation. Mol Cancer. 2024;23:70.
Kwong TT, Wong CH, Zhou JY, Cheng ASL, Sung JJY, Chan AWH, et al. Chemotherapy-induced recruitment of myeloid-derived suppressor cells abrogates efficacy of immune checkpoint blockade. JHEP Rep. 2021;3:100224.
Nallasamy P, Nimmakayala RK, Parte S, Are AC, Batra SK, Ponnusamy MP. Tumor microenvironment enriches the stemness features: the architectural event of therapy resistance and metastasis. Mol Cancer. 2022;21:225.
Hanahan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell. 1996;86:353–64.
Brown JM, Giaccia AJ. The unique physiology of solid tumors: opportunities (and problems) for cancer therapy. Cancer Res. 1998;58:1408–16.
Goldie JH, Coldman AJ. The genetic origin of drug resistance in neoplasms: Implications for systemic therapy. Cancer Res. 1984;44:3643–53.
Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3:721–32.
Wouters BG, Koritzinsky M. Hypoxia signalling through mTOR and the unfolded protein response in cancer. Nat Rev Cancer. 2008;8:851–64.
Karin M, Greten FR. NF-kappaB: Linking inflammation and immunity to cancer development and progression. Nat Rev Immunol. 2005;5:749–59.
Pickup MW, Mouw JK, Weaver VM. The extracellular matrix modulates the hallmarks of cancer. EMBO Rep. 2014;15:1243–53.
Yu F-X, Zhao B, Guan K-L. Hippo pathway in organ size control, tissue homeostasis, and cancer. Cell. 2015;163:811–28.
Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19:1423–37.
Topalian SL, Taube JM, Anders RA, Pardoll DM. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer. 2016;16:275–87.
Chari RVJ, Miller ML, Widdison WC. Antibody-drug conjugates: an emerging concept in cancer therapy. Angew Chem. 2014;53:3796–827.
Semenza GL. Hypoxia-inducible factors in physiology and medicine. Cell. 2012;148:399–408.
Jaakkola P, Mole DR, Tian YM, Wilson MI, Gielbert J, Gaskell SJ, et al. Targeting of HIF-alpha to the von hippel-lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science. 2001;292:468–72.
Kshitiz, Afzal J, Suhail Y, Chang H, Hubbi ME, Hamidzadeh A, et al. Lactate-dependent chaperone-mediated autophagy induces oscillatory HIF-1α activity promoting proliferation of hypoxic cells. Cell Syst. 2022;13:1048–1064.e7.
Yeh Y-H, Hsiao H-F, Yeh Y-C, Chen T-W, Li T-K. Inflammatory interferon activates HIF-1α-mediated epithelial-to-mesenchymal transition via PI3K/AKT/mTOR pathway. J Exp Clin Cancer Res. 2018;37:70.
Priego-Hernández VD, Arizmendi-Izazaga A, Soto-Flores DG, Santiago-Ramón N, Feria-Valadez MD, Navarro-Tito N, et al. Expression of HIF-1α and genes involved in glucose metabolism is increased in cervical cancer and HPV-16-positive cell lines. Pathogens. 2022;12:33.
Kato T, Sakurai M, Watanabe K, Mizukami Y, Nakagawa T, Baba K, et al. Identification of hypoxia-induced metabolism-associated genes in canine tumours. Vet Comp Oncol. 2024;22:367–76.
Nishioku T, Nakao S, Anzai R, Hiramatsu S, Momono A, Moriyama M, et al. HIF-1α stabilization in osteoclasts induces the expression of aerobic glycolysis-related proteins GLUT1, LDHA, and MCT4. J Pharmacol Sci. 2025;158:336–42.
Jin Z, Xiang R, Dai J, Wang Y, Xu Z. HIF-1α mediates CXCR4 transcription to activate the AKT/mTOR signaling pathway and augment the viability and migration of activated B cell-like diffuse large B-cell lymphoma cells. Mol Carcinog. 2023;62:676–84.
Guo M, Cai C, Zhao G, Qiu X, Zhao H, Ma Q, et al. Hypoxia promotes migration and induces CXCR4 expression via HIF-1α activation in human osteosarcoma. PLoS One. 2014;9:e90518.
Brooks DLP, Schwab LP, Krutilina R, Parke DN, Sethuraman A, Hoogewijs D, et al. ITGA6 is directly regulated by hypoxia-inducible factors and enriches for cancer stem cell activity and invasion in metastatic breast cancer models. Mol Cancer. 2016;15:26.
Gou S, Wu A, Luo Z. Integrins in cancer stem cells. Front Cell Dev Biol. 2024;12:1434378.
Yuan X, Liu X, Jiang D, Zheng Z, Ma X, Wu S, et al. Exosomal PD-L1 derived from hypoxia nasopharyngeal carcinoma cell exacerbates CD8+ T cell suppression by promoting PD-L1 upregulation in macrophages. Cancer Immunol Immunother. 2025;74:220.
Bailey CM, Liu Y, Liu M, Du X, Devenport M, Zheng P, et al. Targeting HIF-1α abrogates PD-L1-mediated immune evasion in tumor microenvironment but promotes tolerance in normal tissues. J Clin Invest. 2022;132:e150846.
Wang R, Cai X, Li X, Li J, Liu X, Wang J, et al. USP38 promotes deubiquitination of K11-linked polyubiquitination of HIF1α at Lys769 to enhance hypoxia signaling. J Biol Chem. 2024;300:105532.
Chen R, Lin Z, Shen S, Zhu C, Yan K, Suo C, et al. Citrullination modulation stabilizes HIF-1α to promote tumour progression. Nat Commun. 2024;15:7654.
Yang K, Zhang W, Zhong L, Xiao Y, Sahoo S, Fassan M, et al. Long non-coding RNA HIF1A-As2 and MYC form a double-positive feedback loop to promote cell proliferation and metastasis in KRAS-driven non-small cell lung cancer. Cell Death Differ. 2023;30:1533–49.
Ji Z, He L, Regev A, Struhl K. Inflammatory regulatory network mediated by the joint action of NF-kB, STAT3, and AP-1 factors is involved in many human cancers. Proc Natl Acad Sci USA. 2019;116:9453–62.
Chen Y-G, Rieser E, Bhamra A, Surinova S, Kreuzaler P, Ho M-H, et al. LUBAC enables tumor-promoting LTβ receptor signaling by activating canonical NF-κB. Cell Death Differ. 2024;31:1267–84.
Helbig G, Christopherson KW, Bhat-Nakshatri P, Kumar S, Kishimoto H, Miller KD, et al. NF-kappaB promotes breast cancer cell migration and metastasis by inducing the expression of the chemokine receptor CXCR4. J Biol Chem. 2003;278:21631–8.
Choi S-H, Kim M-Y, Yoon Y-S, Koh D-I, Kim M-K, Cho S-Y, et al. Hypoxia-induced RelA/p65 derepresses SLC16A3 (MCT4) by downregulating ZBTB7A. Biochim Biophys Acta Gene Regul Mech. 2019;1862:771–85.
Lim S-O, Li C-W, Xia W, Cha J-H, Chan L-C, Wu Y, et al. Deubiquitination and stabilization of PD-L1 by CSN5. Cancer Cell. 2016;30:925–39.
Campos AD, Shepard RM, Ortega Z, Heumann I, Wilke AE, Nam A, et al. A STAT3/integrin axis accelerates pancreatic cancer initiation and progression. Cell Rep. 2025;44:116010.
Huang L, Zhu Y, Kong Q, Guan X, Lei X, Zhang L, et al. Inhibition of integrin αvβ3-FAK-MAPK signaling constrains the invasion of T-ALL cells. Cell Adhes Migr. 2023;17:1–14.
Nagesh R, Kiran Kumar KM, Naveen Kumar M, Patil RH, Sharma SC. Regulation of jun and fos AP-1 transcription factors by JNK MAPKs signaling cascade in areca nut extract treated KB cells. Biochem Biophys Rep. 2021;27:101090.
Zhang K, Myllymäki S-M, Gao P, Devarajan R, Kytölä V, Nykter M, et al. Oncogenic K-ras upregulates ITGA6 expression via FOSL1 to induce anoikis resistance and synergizes with αV-class integrins to promote EMT. Oncogene. 2017;36:5681–94.
Zhong Z, Wen Z, Darnell JE. Stat3: A STAT family member activated by tyrosine phosphorylation in response to epidermal growth factor and interleukin-6. Science. 1994;264:95–98.
Lee H-J, Zhuang G, Cao Y, Du P, Kim H-J, Settleman J. Drug resistance via feedback activation of Stat3 in oncogene-addicted cancer cells. Cancer Cell. 2014;26:207–21.
Tian Y, Qi H, Zhu N, Yan X, Guo L, Wang L, et al. An IL-6-induced STAT3-to-PI3K signaling switch potently drives PD-L1 transcription in cancer stem cells of colorectal cancer. Sci Bull. 2025;S2095-9273:00733–9.
Das SK, Lewis BA, Levens D. MYC: a complex problem. Trends Cell Biol. 2023;33:235–46.
Nie Z, Guo C, Das SK, Chow CC, Batchelor E, Simons SS, et al. Dissecting transcriptional amplification by MYC. Elife. 2020;9:e52483.
Dhara A, Aier I, Das S, Sarkar M, Kaur R, Varadwaj PK, et al. Enhanced ETS1 stability by DNAPKcs orchestrates transcriptional changes during chemoresistance in triple negative breast cancer. NPJ Breast Cancer. 2025;11:114.
Sun X, Teper Y, Sinnett-Smith J, Markarian M, Hines OJ, Li G, et al. Stress and obesity signaling converge on CREB phosphorylation to promote pancreatic cancer. Mol Cancer Res. 2025;23:236–49.
Dong Y, Tu R, Liu H, Qing G. Regulation of cancer cell metabolism: oncogenic MYC in the driver’s seat. Signal Transduction Targeted Ther. 2020;5:124.
Wang Z, Ouyang L, Liu N, Li T, Yan B, Mao C, et al. The DUBA-SLC7A11-c-myc axis is critical for stemness and ferroptosis. Oncogene. 2023;42:2688–2700.
Yin Y, Sun Y, Yao H, Yu F, Jia Q, Hu C, et al. TMEM105 modulates disulfidptosis and tumor growth in pancreatic cancer via the β-catenin-c-MYC-GLUT1 axis. Int J Biol Sci. 2025;21:1932–48.
Sobral LM, Sechler M, Parrish JK, McCann TS, Jones KL, Black JC, et al. KDM3A/Ets1/MCAM axis promotes growth and metastatic properties in rhabdomyosarcoma. Genes Cancer. 2020;11:53–65.
Fang L-W, Kao Y-H, Chuang Y-T, Huang H-L, Tai T-S. Ets-1 enhances tumor migration through regulation of CCR7 expression. BMB Rep. 2019;52:548–53.
Dai W, Xu Y, Mo S, Li Q, Yu J, Wang R, et al. GLUT3 induced by AMPK/CREB1 axis is key for withstanding energy stress and augments the efficacy of current colorectal cancer therapies. Signal Transduct Target Ther. 2020;5:177.
Chen J, Wang K, Ye S, Meng X, Jia X, Huang Y, et al. Tyrosine kinase receptor RON activates MAPK/RSK/CREB signal pathway to enhance CXCR4 expression and promote cell migration and invasion in bladder cancer. Aging. 2022;14:7093–108.
Kang J, Chun J, Hwang JS, Pan C, Li J, Boese AC, et al. EGFR-phosphorylated GDH1 harmonizes with RSK2 to drive CREB activation and tumor metastasis in EGFR-activated lung cancer. Cell Rep. 2022;41:111827.
Wang Y, Sparidans RW, Potters S, Lebre MC, Beijnen JH, Schinkel AHABCB1. and ABCG2, but not CYP3A4 limit oral availability and brain accumulation of the RET inhibitor pralsetinib. Pharmacol Res. 2021;172:105850.
Yan Y, Teng H, Hang Q, Kondiparthi L, Lei G, Horbath A, et al. SLC7A11 expression level dictates differential responses to oxidative stress in cancer cells. Nat Commun. 2023;14:3673.
Koppula P, Olszewski K, Zhang Y, Kondiparthi L, Liu X, Lei G, et al. KEAP1 deficiency drives glucose dependency and sensitizes lung cancer cells and tumors to GLUT inhibition. iScience. 2021;24:102649.
Li J, Wu Z, Chen G, Wang X, Zhu X, Zhang Y, et al. Formosanin C inhibits non-small-cell lung cancer progression by blocking MCT4/CD147-mediated lactate export. Phytomedicine. 2023;109:154618.
Diehl K, Dinges L-A, Helm O, Ammar N, Plundrich D, Arlt A, et al. Nuclear factor E2-related factor-2 has a differential impact on MCT1 and MCT4 lactate carrier expression in colonic epithelial cells: a condition favoring metabolic symbiosis between colorectal cancer and stromal cells. Oncogene. 2018;37:39–51.
Wang S, Zhang X, Chen Q, Wu H, Cao S, Zhao S, et al. FTO activates PD-L1 promotes immunosuppression in breast cancer via the m6A/YTHDF3/PDK1 axis under hypoxic conditions. J Adv Res. 2025;76:191–206.
Peuker K, Strigli A, Tauriello DVF, Hendricks A, von Schönfels W, Burmeister G, et al. Microbiota-dependent activation of the myeloid calcineurin-NFAT pathway inhibits B7H3- and B7H4-dependent anti-tumor immunity in colorectal cancer. Immunity. 2022;55:701–17.e7.
Guo X, Chang M, Wang Y, Xing B, Ma W. B7-H3 in brain malignancies: Immunology and immunotherapy. Int J Biol Sci. 2023;19:3762–80.
Ma Z, Yang J, Jia W, Li L, Li Y, Hu J, et al. Histone lactylation-driven B7-H3 expression promotes tumor immune evasion. Theranostics. 2025;15:2338–59.
Sutton MN, Glazer SE, Muzzioli R, Yang P, Gammon ST, Piwnica-Worms D. Dimerization of the 4Ig isoform of B7-H3 in tumor cells mediates enhanced proliferation and tumorigenic signaling. Commun. Biol. 2024;7:21.
Seo YR, Kim HB, Jung H, Kim EG, Huh S, Yi EC, et al. Unveiling transcriptional mechanisms of B7-H3 in breast cancer stem cells through proteomic approaches. iScience. 2025;28:112218.
Dong Y, Zhang Z, Luan S, Zheng M, Wang Z, Chen Y, et al. Novel bispecific antibody-drug conjugate targeting PD-L1 and B7-H3 enhances antitumor efficacy and promotes immune-mediated antitumor responses. J Immunother Cancer. 2024;12:e009710.
Ishii S, Hirane M, Fukushima K, Tomimatsu A, Fukushima N, Tsujiuchi T. Diverse effects of LPA4, LPA5 and LPA6 on the activation of tumor progression in pancreatic cancer cells. Biochem Biophys Res Commun. 2015;461:59–64.
Lee J-W, Lee C-S, Ryu Y-R, Lee J, Son H, Cho H-J, et al. Lysophosphatidic acid receptor 4 is transiently expressed during cardiac differentiation and critical for repair of the damaged heart. Mol Ther. 2021;29:1151–63.
Wang S, Jiang H, Liu J, Chen J, Hu S, Cui Y, et al. LPAR4-targeted dual-loaded liposome for synergistic lung cancer therapy. Eur J Pharm Sci. 2025;212:107176.
Chia K, Mazzolini J, Mione M, Sieger D. Tumor initiating cells induce Cxcr4-mediated infiltration of pro-tumoral macrophages into the brain. Elife. 2018;7:e31918.
Pal J, Patil V, Mondal B, Shukla S, Hegde AS, Arivazhagan A, et al. Epigenetically silenced GNG4 inhibits SDF1α/CXCR4 signaling in mesenchymal glioblastoma. Genes Cancer. 2016;7:136–47.
Weng J-R, Tsai C-H, Omar HA, Sargeant AM, Wang D, Kulp SK, et al. OSU-A9, a potent indole-3-carbinol derivative, suppresses breast tumor growth by targeting the Akt-NF-kappaB pathway and stress response signaling. Carcinogenesis. 2009;30:1702–9.
Rodriguez LR, Emblom-Callahan M, Chhina M, Bui S, Aljeburry B, Tran LH, et al. Global gene expression analysis in an in vitro fibroblast model of idiopathic pulmonary fibrosis reveals potential role for CXCL14/CXCR4. Sci Rep. 2018;8:3983.
Bergonzini C, Gregori A, Hagens TMS, van der Noord VE, van de Water B, Zweemer AJM, et al. ABCB1 overexpression through locus amplification represents an actionable target to combat paclitaxel resistance in pancreatic cancer cells. J Exp Clin Cancer Res. 2024;43:4.
Belli S, Esposito D, Ascione CM, Messina F, Napolitano F, Servetto A, et al. EGFR and HER2 hyper-activation mediates resistance to endocrine therapy and CDK4/6 inhibitors in ER+ breast cancer. Cancer Lett. 2024;593:216968.
Gupta A, Michelini F, Shao H, Yeh C, Drago JZ, Liu D, et al. EGFR-directed antibodies promote HER2 ADC internalization and efficacy. Cell Rep Med. 2024;5:101792.
Lu Y, Liu Y, Oeck S, Zhang GJ, Schramm A, Glazer PM. Hypoxia induces resistance to EGFR inhibitors in lung cancer cells via upregulation of FGFR1 and the MAPK pathway. Cancer Res. 2020;80:4655–67.
Kwon J, Lee T-S, Lee HW, Kang MC, Yoon H-J, Kim J-H, et al. Integrin alpha 6: A novel therapeutic target in esophageal squamous cell carcinoma. Int J Oncol. 2013;43:1523–30.
Cong F, Huang J, Wu C, Zhong H, Qiu G, Luo T, et al. Integrin α6 and integrin β4 in exosomes promote lung metastasis of colorectal cancer. J Cancer Res Ther. 2024;20:2082–93.
Zhao Z, Li Z-Q, Huang Y-B, Liu M-M, Cao F, Bu G-L, et al. An optimized integrin α6-targeted peptide capable of delivering toxins for melanoma treatment. J Transl Med. 2025;23:495.
Kowalski-Chauvel A, Modesto A, Gouaze-andersson V, et al. Alpha-6 integrin promotes radioresistance of glioblastoma by modulating DNA damage response and the transcription factor Zeb1. Cell Death Dis. 2018;9:872.
Okabe H, Ishimoto T, Mima K, Nakagawa S, Hayashi H, Kuroki H, et al. CD44s signals the acquisition of the mesenchymal phenotype required for anchorage-independent cell survival in hepatocellular carcinoma. Br J Cancer. 2014;110:958–6.
Jaggupilli A, Elkord E. Significance of CD44 and CD24 as cancer stem cell markers: An enduring ambiguity. Clin Dev Immunol. 2012;2012:708036.
Fletcher JI, Haber M, Henderson MJ, Norris MD. ABC transporters in cancer: more than just drug efflux pumps. Nat Rev Cancer. 2010;10:147–56.
Siebeneicher H, Cleve A, Rehwinkel H, Neuhaus R, Heisler I, Müller T, et al. Identification and optimization of the first highly selective GLUT1 inhibitor BAY-876. Chem Med Chem. 2016;11:2261–71.
Reckzeh ES, Karageorgis G, Schwalfenberg M, Ceballos J, Nowacki J, Stroet MCM, et al. Inhibition of glucose transporters and glutaminase synergistically impairs tumor cell growth. Cell Chem Biol. 2019;26:1214–28.e25.
Benjamin D, Robay D, Hindupur SK, Pohlmann J, Colombi M, El-Shemerly MY, et al. Dual inhibition of the lactate transporters MCT1 and MCT4 is synthetic lethal with metformin due to NAD⁺depletion incancer cells. Cell Rep. 2018;25:3047–58.e4.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82573393, CW), the Research Foundation of Chinese Academy of Sciences (CW), and the Starting Fund from the University of Chinese Academy of Sciences (CW). All the artwork in this manuscript was created with BioRender.com.
Author information
Authors and Affiliations
Contributions
XH and TYH drafted the manuscript. ZW and FB performed literature search and edited the manuscript. XC, MX and CW performed literature search, critically reviewed and finalized the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, X., Huang, T., Wang, Z. et al. Stress-responsive membrane proteins as execution nodes of tumor cell adaptation to microenvironmental stress. Oncogene (2026). https://doi.org/10.1038/s41388-026-03768-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41388-026-03768-y