Abstract
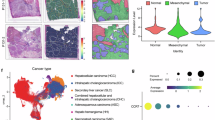
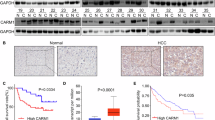
Cancer-associated hyperfibrinolysis are potentially fatal outcomes of advanced cancer. Unfortunately, this knowledge has not yet led to any breakthrough in cancer therapy. Plasmin is the key enzyme of fibrinolytic system. The identification of a direct link between plasmin and the tumor progression remains unknown. Here, we demonstrated that plasmin expression was significantly upregulated in HCC tissue, especially tumor tissues from patients who had received chemotherapy. Through in vitro and in vivo models, we showed that plasmin enhanced HCC cell invasion and migration. Pharmacological inhibition of plasmin with tranexamic acid (TXA) suppressed tumor invasion and metastasis. Mechanistically, we identified plasmin could directly bind to CXCR4, which potentiated the CXCL12/CXCR4 interaction and activated the PI3K/AKT/mTOR signaling pathway. Moreover, CDDP-resistant cells had higher expression of plasmin and stronger invasion and metastasis ability, which were effectively suppressed by plasmin inhibitor, and combining TXA with CDDP synergistically inhibited tumor progression and metastasis in mouse model and human HCC organoids. Our findings reveal a novel plasmin-CXCR4 axis driving HCC dissemination and suggest that targeting plasmin may enhance the efficacy of conventional chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The datasets utilized and/or analyzed during this study are available from the corresponding author upon reasonable request.
References
Li X, Ramadori P, Pfister D, Seehawer M, Zender L, Heikenwalder M. The immunological and metabolic landscape in primary and metastatic liver cancer. Nat Rev Cancer. 2021;21:541–57.
Llovet JM, Pinyol R, Yarchoan M, Singal AG, Marron TU, Schwartz M, et al. Adjuvant and neoadjuvant immunotherapies in hepatocellular carcinoma. Nat Rev Clin Oncol. 2024;21:294–311.
Zhang H, Yang Z, Jiang Z, Zhao Z, Chen X, Wan J, et al. Aberrant angiogenic signaling in HCC: therapeutic targeting and drug resistance. Front Oncol. 2025;15:1595195.
Wang Y, Deng B. Hepatocellular carcinoma: molecular mechanism, targeted therapy, and biomarkers. Cancer Metastasis Rev. 2023;42:629–52.
Sun J, Mao F, Liu C, Zhang F, Jiang D, Guo W, et al. Combined FOLFOX4 with all-trans retinoic acid versus FOLFOX4 with placebo in treatment of advanced hepatocellular carcinoma with extrahepatic metastasis: a randomized, double-blind comparative study. Signal Transduct Target Ther. 2023;8:368.
Weiss F, Lauffenburger D, Friedl P. Towards targeting of shared mechanisms of cancer metastasis and therapy resistance. Nat Rev Cancer. 2022;22:157–73.
Mutch NJ, Medcalf RL. The fibrinolysis renaissance. J Thromb Haemost. 2023;21:3304–16.
Hisada Y, Geddings JE, Ay C, Mackman N. Venous thrombosis and cancer: from mouse models to clinical trials. J Thromb Haemost. 2015;13:1372–82.
Heissig B, Eiamboonsert S, Salama Y, Shimazu H, Dhahri D, Munakata S, et al. Cancer therapy targeting the fibrinolytic system. Adv Drug Deliv Rev. 2016;99:172–9.
Ismail AA, Shaker BT, Bajou K. The plasminogen-activator plasmin system in physiological and pathophysiological angiogenesis. Int J Mol Sci 2021;23:337.
Kubala MH, Punj V, Placencio-Hickok VR, Fang H, Fernandez GE, Sposto R, et al. Plasminogen activator inhibitor-1 promotes the recruitment and polarization of macrophages in cancer. Cell Rep. 2018;25:2177–2191 e2177.
Molina JJ, Flores-Mireles AL. Role of clot formation and plasminogen activation in lower urinary tract pathologies. Arterioscler Thromb Vasc Biol. 2025;45:1471–8.
Uhl B, Mittmann LA, Dominik J, Hennel R, Smiljanov B, Haring F, et al. uPA-PAI-1 heteromerization promotes breast cancer progression by attracting tumorigenic neutrophils. EMBO Mol Med. 2021;13:e13110.
Wang B, Gu B, Zhang T, Li X, Wang N, Ma C, et al. Good or bad: Paradox of plasminogen activator inhibitor 1 (PAI-1) in digestive system tumors. Cancer Lett. 2023;559:216117.
Hisada Y, Mackman N. Profibrinolytic factors and cancer progression, metastasis, and survival. Arterioscler Thromb Vasc Biol. 2025;45:1732–41.
Bonlokke ST, Fenger-Eriksen C, Ommen HB, Hvas AM. Impaired fibrinolysis and increased clot strength are potential risk factors for thrombosis in lymphoma. Blood Adv. 2023;7:7056–66.
Loosen SH, Gorgulho J, Jordens MS, Schulze-Hagen M, Beier F, Vucur M, et al. Serum levels of soluble urokinase plasminogen activator receptor predict tumor response and outcome to immune checkpoint inhibitor therapy. Front Oncol. 2021;11:646883.
Keragala CB, Medcalf RL. Plasminogen: an enigmatic zymogen. Blood. 2021;137:2881–9.
Liu RZ, Hu YJ, Wang TF, Yu JC, Wang YX, Zhang YL et al. A novel recombinant human microplasminogen induced complete posterior vitreous detachment without morphological change of retina in juvenile rabbits. Exp Eye Res. 2023;230:109465.
Law RHP, Abu-Ssaydeh D, Whisstock JC. New insights into the structure and function of the plasminogen/plasmin system. Curr Opin Struc Biol. 2013;23:836–41.
O’Connell PA, Surette AP, Liwski RS, Svenningsson P, Waisman DM. S100A10 regulates plasminogen-dependent macrophage invasion. Blood. 2010;116:1136–46.
Bharadwaj AG, Kempster E, Waisman DM. The ANXA2/S100A10 complex-regulation of the oncogenic plasminogen receptor. Biomolecules. 2021;11:1772.
Phipps KD, Surette AP, O’Connell PA, Waisman DM. Plasminogen receptor S100A10 is essential for the migration of tumor-promoting macrophages into tumor sites. Cancer Res. 2011;71:6676–83.
Heissig B, Salama Y, Osada T, Okumura K, Hattori K. The multifaceted role of plasminogen in cancer. Int J Mol Sci. 2021;22:2304.
Bao SS, Darvishi M, Amin A, Al-Haideri MT, Patra I, Kashikova K, et al. CXC chemokine receptor 4 (CXCR4) blockade in cancer treatment. J Cancer Res Clin. 2023;149:7945–68.
Chatterjee S, Behnam Azad B, Nimmagadda S. The intricate role of CXCR4 in cancer. Adv Cancer Res. 2014;124:31–82.
Xin Y, Hu B, Li KM, Hu GS, Zhang CY, Chen X et al. Circulating tumor cells with metastasis-initiating competence survive fluid shear stress during hematogenous dissemination through CXCR4-PI3K/ AKT signaling. Cancer Lett. 2024;590:216870.
Teicher BA, Fricker SP. CXCL12 (SDF-1)/CXCR4 pathway in cancer. Clin Cancer Res. 2010;16:2927–31.
Yang YR, Li JY, Lei WR, Wang HY, Ni YF, Liu YQ, et al. CXCL12-CXCR4/CXCR7 axis in cancer: from mechanisms to clinical applications. Int J Biol Sci. 2023;19:3341–59.
Liu DQ, Liu L, Zhang XM, Zhao XM, Li XR, Che XY et al. Decoding driver and phenotypic genes in cancer: Unveiling the essence behind the phenomenon. Mol Aspects Med. 2025;103:101358.
Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA. 2005;293:715–22.
Didiasova M, Wujak L, Wygrecka M, Zakrzewicz D. From plasminogen to plasmin: role of plasminogen receptors in human cancer. Int J Mol Sci. 2014;15:21229–52.
Sharma MC, Sharma M. The role of annexin II in angiogenesis and tumor progression: a potential therapeutic target. Curr Pharm Des. 2007;13:3568–75.
Chowdhury NN, Yang Y, Dutta A, Luo M, Wei Z, Abrahams SR, et al. Plasminogen deficiency suppresses pancreatic ductal adenocarcinoma disease progression. Mol Oncol. 2024;18:113–35.
Law ME, Davis BJ, Ghilardi AF, Yaaghubi E, Dulloo ZM, Wang MX et al. Repurposing tranexamic acid as an anticancer agent. Front Pharmacol. 2022;12:792600.
Jaffer AA, Karanicolas PJ, Davis LE, Behman R, Hanna SS, Law CH, et al. The impact of tranexamic acid on administration of red blood cell transfusions for resection of colorectal liver metastases. HPB Oxf. 2021;23:245–52.
Lohani KR, Kumar C, Kataria K, Srivastava A, Ranjan P, Dhar A. Role of tranexamic acid in axillary lymph node dissection in breast cancer patients: does it help in reducing lymphedema?. Breast J. 2021;27:502–502.
Wu CC, Ho WM, Cheng SB, Yeh DC, Wen MC, Liu TJ, et al. Perioperative parenteral tranexamic acid in liver tumor resection: a prospective randomized trial toward a “blood transfusion”-free hepatectomy. Ann Surg. 2006;243:173–80.
Bobkov V, Arimont M, Zarca A, De Groof TWM, van der Woning B, de Haard H, et al. Antibodies targeting chemokine receptors CXCR4 and ACKR3. Mol Pharm. 2019;96:753–64.
Lu LX, Li JJ, Jiang XY, Bai RR. CXCR4/CXCL12 axis: “old” pathway as “novel” target for anti-inflammatory drug discovery. Med Res Rev. 2024;44:1189–220.
Khan AB, Lee S, Harmanci AS, Patel R, Latha K, Yang YH, et al. CXCR4 expression is associated with proneural-to-mesenchymal transition in glioblastoma. Int J Cancer. 2023;152:713–24.
Hamaya S, Oura K, Morishita A, Masaki T. Cisplatin in liver cancer therapy. Int J Mol Sci. 2023;24:10858.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (NSFC 82273293), Shanghai Municipal Health Commission Health Industry Clinical Research Project (20224Y0120).
Author information
Authors and Affiliations
Contributions
ZB and SZ and YM performed all of the experiments. YY, MT, and YL participated in the research. ZB and SZ designed experiments, analyzed data, and wrote the paper. WF and LF collected the clinical samples. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The animal experiments were conducted in strict accordance with the guidelines of the Institutional Animal Care and Use Committee (IACUC) of Fudan University Shanghai Cancer Center. Approval for these experiments was obtained under the following permission numbers: FUSCC-IACUC-20220228-169 (Shanghai, China). All experiments using human tissues were performed in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Fudan University, Approval for these experiments was obtained under the following permission numbers: FUSCC-IACUC-2022-C016 (Shanghai, China). All methods were performed in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Fudan University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shen, Z., Yang, Y., Mao, T. et al. Plasmin promotes hepatocellular carcinoma invasion and metastasis via CXCR4-mediated activation of PI3K/AKT/mTOR signaling. Oncogene (2026). https://doi.org/10.1038/s41388-026-03775-z
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41388-026-03775-z