Abstract
Background
Many premature infants with respiratory failure are deficient in surfactant, but the relationship to occurrence of bronchopulmonary dysplasia (BPD) is uncertain.
Methods
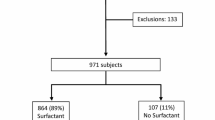
Tracheal aspirates were collected from 209 treated and control infants enrolled at 7–14 days in the Trial of Late Surfactant. The content of phospholipid, surfactant protein B, and total protein were determined in large aggregate (active) surfactant.
Results
At 24 h, surfactant treatment transiently increased surfactant protein B content (70%, p < 0.01), but did not affect recovered airway surfactant or total protein/phospholipid. The level of recovered surfactant during dosing was directly associated with content of surfactant protein B (r = 0.50, p < 0.00001) and inversely related to total protein (r = 0.39, p < 0.0001). For all infants, occurrence of BPD was associated with lower levels of recovered large aggregate surfactant, higher protein content, and lower SP-B levels. Tracheal aspirates with lower amounts of recovered surfactant had an increased proportion of small vesicle (inactive) surfactant.
Conclusions
We conclude that many intubated premature infants are deficient in active surfactant, in part due to increased intra-alveolar metabolism, low SP-B content, and protein inhibition, and that the severity of this deficit is predictive of BPD. Late surfactant treatment at the frequency used did not provide a sustained increase in airway surfactant.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Ehrenkranz, R. A. et al. Validation of the National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics 116, 1353–1360 (2005).
Natarajan, G. et al. Outcomes of extremely low birth weight infants with bronchopulmonary dysplasia: impact of the physiologic definition. Early Hum. Dev. 88, 509–515 (2012).
Jobe, A. H. & Bancalari, E. Bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 163, 1723–1729 (2001).
Coalson, J. J. Pathology of new bronchopulmonary dysplasia. Semin. Neonatol. 8, 73–81 (2003).
Gibson, A. M. et al. Lung function in adult survivors of very low birth weight, with and without bronchopulmonary dysplasia. Pediatr. Pulmonol. 50, 987–994 (2015).
Greenough, A. & Ahmed, N. Perinatal prevention of bronchopulmonary dysplasia. J. Perinat. Med. 41, 119–126 (2013).
Cole, F. S. et al. NIH Consensus Development Conference statement: inhaled nitric-oxide therapy for premature infants. Pediatrics 127, 363–369 (2011).
Askie, L. M. et al. Race effects of inhaled nitric oxide in preterm infants: an individual participant data meta-analysis. J. Pediatr. 193, 34–39 (2018).
Merrill, J. D. et al. Dysfunction of pulmonary surfactant in chronically ventilated premature infants. Pediatr. Res. 56, 1–9 (2004).
Merrill, J. D. et al. Pilot trial of late booster doses of surfactant for ventilated premature infants. J. Perinatol. 31, 599–606 (2011).
Keller, R. L. et al. Late administration of surfactant replacement therapy increases surfactant protein-B content: a randomized pilot study. Pediatr. Res. 72, 613–619 (2012).
Katz, L. A. & Klein, J. M. Repeat surfactant therapy for postsurfactant slump. J. Perinatol. 26, 414–422 (2006).
Bissinger, R., Carlson, C., Hulsey, T. & Eicher, D. Secondary surfactant deficiency in neonates. J. Perinatol. 24, 663–666 (2004).
Laughon, M. et al. A pilot randomized, controlled trial of later treatment with a peptide-containing, synthetic surfactant for the prevention of bronchopulmonary dysplasia. Pediatrics 123, 89–96 (2009).
Ballard, R. A. et al. Inhaled nitric oxide in preterm infants undergoing mechanical ventilation. N. Engl. J. Med. 355, 343–353 (2006).
Ballard, R. A. et al. Randomized Trial of Late Surfactant treatment in ventilated preterm infants receiving inhaled nitric oxide. J. Pediatr. 168, 23–9 e4 (2016).
Keller, R. L. et al. The randomized, controlled Trial of Late Surfactant: effects on respiratory outcomes at 1-year corrected age. J. Pediatr. 183, 19–25 e2 (2017).
Pryhuber, G. S. et al. Prematurity and respiratory outcomes program (PROP): study protocol of a prospective multicenter study of respiratory outcomes of preterm infants in the United States. BMC Pediatr. 15, 37 (2015).
Poindexter, B. B. et al. Comparisons and limitations of current definitions of bronchopulmonary dysplasia for the prematurity and respiratory outcomes program. Ann. Am. Thorac. Soc. 12, 1822–1830 (2015).
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Med. Sci. 37, 911–917 (1959).
Dittmer, J. C. & Wells, M. A. in Methods in Enzymology (ed. Lowenstein, J. M.) 482–487 (Academic Press Inc., New York, 1969).
Ballard, P. L. et al. Surfactant protein profile of pulmonary surfactant in premature infants. Am. J. Respir. Crit. Care Med. 168, 1123–1128 (2003).
R Development Core Team. R: A Language and Environment for Statistical Computing ISBN 3-900051-07-0, http://www.r-project.org/ (R Foundation for Statistical Computing, Vienna, Austria, 2008).
Venables, W. N. & Ripley, B. D. Modern Applied Statistics. 4th edn (Springer, New York, 2002).
Torresin, M. et al. Exogenous surfactant kinetics in infant respiratory distress syndrome: a novel method with stable isotopes. Am. J. Respir. Crit. Care Med. 161, 1584–1589 (2000).
Yamada, T., Ikegami, M. & Jobe, A. H. Effects of surfactant subfractions on preterm rabbit lung function. Pediatr. Res. 27, 592–598 (1990).
Wai, K. C. et al. Early cumulative supplemental oxygen predicts bronchopulmonary dysplasia in high risk extremely low gestational age newborns. J. Pediatr. 177, 97–102 e2 (2016).
Bhandari, A. & Bhandari, V. Biomarkers in bronchopulmonary dysplasia. Paediatr. Respir. Rev. 14, 173–179 (2013).
Ikegami, M., Whitsett, J. A., Martis, P. C. & Weaver, T. E. Reversibility of lung inflammation caused by SP-B deficiency. Am. J. Physiol. Lung Cell. Mol. Physiol. 289, L962–L970 (2005).
Verlato, G. et al. Surfactant status in preterm neonates recovering from respiratory distress syndrome. Pediatrics 122, 102–108 (2008).
Carnielli, V. P., Zimmermann, L. J., Hamvas, A. & Cogo, P. E. Pulmonary surfactant kinetics of the newborn infant: novel insights from studies with stable isotopes. J. Perinatol. 29(Suppl. 2), S29–S37 (2009).
Facco, M. et al. In vivo effect of pneumonia on surfactant disaturated-phosphatidylcholine kinetics in newborn infants. PLoS ONE 9, e93612 (2014).
Bohlin, K., Merchak, A., Spence, K., Patterson, B. W. & Hamvas, A. Endogenous surfactant metabolism in newborn infants with and without respiratory failure. Pediatr. Res. 54, 185–191 (2003).
Cogo, P. E. et al. Surfactant disaturated phosphatidylcholine kinetics in infants with bronchopulmonary dysplasia measured with stable isotopes and a two-compartment model. J. Appl. Physiol. (1985) 99, 323–329 (2005).
Ueda, T., Ikegami, M. & Jobe, A. Surfactant subtypes. In vitro conversion, in vivo function, and effects of serum proteins. Am. J. Respir. Crit. Care Med. 149, 1254–1259 (1994).
Veldhuizen, R. A. et al. Alveolar surfactant aggregate conversion in ventilated normal and injured rabbits. Am. J. Physiol. 270, L152–L158 (1996).
Veldhuizen, R. A., McCaig, L. A., Akino, T. & Lewis, J. F. Pulmonary surfactant subfractions in patients with the acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 152, 1867–1871 (1995).
Ballard, P. L. et al. Surfactant function and composition in premature infants treated with inhaled nitric oxide. Pediatrics 120, 346–353 (2007).
Yeh, T. F. et al. Intratracheal administration of budesonide/surfactant to prevent bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 193, 86–95 (2016).
Roberts, J. K. et al. Pharmacokinetics of budesonide administered with surfactant in premature lambs: implications for neonatal clinical trials. Curr. Clin. Pharmacol. 11, 53–61 (2016).
Acknowledgements
We thank the TOLSURF and PROP Investigators, study coordinators, physicians, nurses, respiratory therapists, and the families who participated in the studies. This study was supported by grants from the National Health, Lung, and Blood Institute (NHLBI, U01 HL094338, U01 HL094355, U01 HL101798). Ikaria Inc. and ONY Inc. provided drug for the conduct of the parent TOLSURF trial, but neither company had input into study design, data collection, analysis and interpretation, or manuscript preparation.
Author contributions
Study design: P.B., R.K., W.T., R.B.; sample and data acquisition: all authors; data interpretation: P.B., R.K., W.T., R.B.; draft manuscript: P.B.; statistics: M.S.; review and approve manuscript: all authors; accountability for work: all authors.
Members of the TOLSURF Study Group
In addition to the authors, the following members of the TOLSURF Study Group participated in the parent trial: University of California San Francisco—Elizabeth E. Rogers, M.D., Dennis M. Black, Ph.D., Lisa Palermo, M.S., S. Hamilton Strong, J. Immamura-Ching, M. Orfanos-Villalobos, C. Williams; Alta Bates Summit Medical Center, Berkeley CA and Children’s Hospital and Research Center Oakland, Oakland CA - Jeffrey D. Merrill, M.D., David J. Durand, M.D, Jeanette M. Asselin R.T., D. Horton, L. Pacello, A. Willard; 4University of Texas Health Science Center – Houston, Houston, TX—Eric C. Eichenwald, C. Garcia M.D., G. McDavid, K. Burson, R. Hinojosa; C. Johnson, K. Miller, S. Rogers, S. Wright; Children’s Mercy Hospital, Kansas City MO—C. Gauldin, A. Holmes, P. Johnson, K. Meinert; Women and Children’s Hospital of Buffalo, Buffalo, NY—A.M. Reynolds, M.D., J. Lucie, P. Conway, M.l. Sacilowski, M. Leadersdorff, P. Orbank, K. Wynn; Anne and Robert H. Lurie Children’s Hospital/Northwestern University, Chicago IL—Robin H. Steinhorn, M.D., Nicolas F. Porta, M.D., M. deUngria, M.D., J. Khan, M.D., K. Hamann, M. Schau, B. Hopkins, J. Jenson; Stonybrook University Hospital, Stonybrook, NY—J. Shariff, R. McGovern, J. Adelman, A. Combs, M. Tjersland; University of Washington, Seattle, WA—Dennis E. Mayock, M.D., S. Walker, E. Howland, J. Longoria, H. Meo; University of Florida College of Medicine – Jacksonville, UF Jacksonville and Wolfson Children’s Hospital, Jacksonville, FL—Mark L. Hudak, M.D., K. Barnette, A. Kellum, M. Burke, C. Hayes, S. Chadwick, D. Howard, C. Kennedy, R. Prince; Wake Forest School of Medicine and Forsyth Medical Center, Winston-Salem, NC—B. Stefanescu, M.D., J. Helderman, M.D., K. Warden, P. Brown, J. Griffin, L. Conley; University of Minnesota Children’s Hospital, Minneapolis, MN—Catherine M. Bendel, M.D., M. Georgieff, M.D., B. Davern, M. Mills, S. Ritter; Medical University of South Carolina, Charleston, SC—Rita M. Ryan, M.D., Frances R. Koch, M.D., C. Wagner, M.D., D. Fanning, J. Roberson; Children’s Hospitals and Clinics of Minnesota, St. Paul, MN—Mark C. Mammel, M.D., A. Lampland, M.D., P. Meyers, A. Brey; Children’s Hospitals and Clinics of Minnesota, Minneapolis, MN—Ellen M. Bendel-Stenzel, M.D., C. Worwa, P. Dixon, G. Ebert, C. Hejl, M. Maxwell, K. McCullough; University of Tennessee Health Science Center, Memphis TN—Ramasubbareddy Dhanireddy, M.D., MT. El Abiad, M.D., A. Talati, M.D., S. Dempsey, K. Gammage, G. Gower, K. James, P. LeNoue; all Children’s Hospital, St. Petersburg, FL—Dennis E. Mayock, M.D., S. Bell, D. Bruton, M. Beaulieu, R. Williams; Florida Hospital for Children, Orlando FL—Rajan Wadhawan, M.D., T. Michael O’Shea, M.D., R. Barron-Nelson, S. Taylor; Arkansas Children’s Hospital and University of Arkansas Medical Sciences, Little Rock, AK—Sherry E. Courtney, M.D., N. C. Sikes, G. Lowe, B. Proffitt; Clinical Coordinating Center: University of California San Francisco Department of Pediatrics—C. Chapin, H. Horneman, K. Hamann, S. Kelley; Data Coordinating Center: University of California San Francisco, Department of Epidemiology and Biostatistics—E. Vittinghoff Ph.D., J. Hietpas, L. Denton, L. Wu; Data Safety Monitoring Board: Cincinnatti Children’s Hospital Medical Center, Cincinnati, OH—A. Jobe, M.D. (Chair 2009–2010); UH Rainbow Babies and Children’s Hospital, Cleveland, OH—A. Fanaroff, M.D. (Chair 2010–2014); EMMES Corporation, Rockville, MD—T. Clemons; Boston University School of Public Health, Boston, MA—L. Glantz; Wake Forest School of Medicine, Winston-Salem, NC—D. Reboussin; Stanford University, Stanford CA—K. Van Meurs MD (2009–2010); Johns Hopkins University, Baltimore MD—M. Allen, M.D. (2010–2014) Women and Infants Hospital, Providence, RI—B. Vohr MD; Clinical Steering Committee: R. Ballard, M.D., P. Ballard, M.D., Ph.D., C. Blaisdell, M.D., D. Durand, M.D., D. Black, Ph.D., E. Eichenwald, M.D., R. Keller, M.D., M. Mammel, M.D., J. Merrill, M.D., E. Rogers, M.D., R. Ryan, M.D., W. Truog, M.D., J. Asselin, N. Newton.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ballard, P.L., Keller, R.L., Truog, W.E. et al. Surfactant status and respiratory outcome in premature infants receiving late surfactant treatment. Pediatr Res 85, 305–311 (2019). https://doi.org/10.1038/s41390-018-0144-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-018-0144-3