Abstract
Background
Hyperoxia (HO) causes kidney injury in preterm infants; however, whether these effects are modifiable is unknown. We hypothesized that administration of exogenous soluble Klotho, a kidney-derived antioxidant, would attenuate HO-induced kidney injury during postnatal nephrogenesis in rats.
Methods
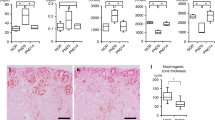
Sprague Dawley neonatal rats assigned to normoxia (21% O2) or HO (85% O2) groups from postnatal day (P) 1 to 21 were randomly assigned to receive alternate day intraperitoneal injections of recombinant Klotho or placebo for 3 weeks. They were recovered in normoxia for an additional 3 weeks and sacrificed at 6 weeks. Renal artery resistance and pulsatility indices, tubular injury scores, glomerular area, and renal antioxidant capacity were assessed.
Results
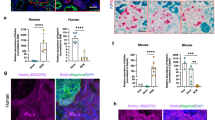
Rodents exposed to HO during postnatal nephrogenesis had reduced kidney Klotho expression, glomerulomegaly, and higher tubular injury scores. Exogenous Klotho administration improved renal perfusion as indicated by decreases in both resistance and pulsatility indices and increased antioxidant enzyme expression.
Conclusions
HO exposure during postnatal nephrogenesis in rodents results in a decline in kidney Klotho expression, decreased renal perfusion, enlarged glomerular size, and tubular injury. The exogenous administration of Klotho attenuated HO-induced kidney injury and augmented antioxidant capacity.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Rodriguez, M. M. et al. Histomorphometric analysis of postnatal glomerulogenesis in extremely preterm infants. Pediatr. Dev. Pathol. 7, 17–25 (2004).
Abitbol, C. L. & Rodriguez, M. M. The long-term renal and cardiovascular consequences of prematurity. Nat. Rev. Nephrol. 8, 265–274 (2012).
Crump, C. et al. Gestational age at birth and mortality in young adulthood. JAMA 306, 1233–1240 (2011).
Luyckx, V. A. et al. A developmental approach to the prevention of hypertension and kidney disease: a report from the Low Birth Weight and Nephron Number Working Group. Lancet 390, 424–428 (2017).
Abitbol, C. L., DeFreitas, M. J. & Strauss, J. Assessment of kidney function in preterm infants: lifelong implications. Pediatr. Nephrol. 31, 2213–2222 (2016).
Abitbol, C. L. & Moxey-Mims, M. Chronic kidney disease: low birth weight and the global burden of kidney disease. Nat. Rev. Nephrol. 12, 199–200 (2016).
Davis, J. M. & Auten, R. L. Maturation of the antioxidant system and the effects on preterm birth. Semin. Fetal Neonatal Med. 15, 191–195 (2010).
Vento, M. et al. Oxidative stress in asphyxiated term infants resuscitated with 100% oxygen. J. Pediatr. 142, 240–246 (2003).
Vento, M. et al. Resuscitation with room air instead of 100% oxygen prevents oxidative stress in moderately asphyxiated term neonates. Pediatrics 107, 642–647 (2001).
Georgeson, G. D. et al. Antioxidant enzyme activities are decreased in preterm infants and in neonates born via caesarean section. Eur. J. Obstet. Gynecol. Reprod. Biol. 103, 136–139 (2002).
Lee, Y. S. & Chou, Y. H. Antioxidant profiles in full term and preterm neonates. Chang Gung Med. J. 28, 846–851 (2005).
Hinchliffe, S. A., Sargent, P. H., Howard, C. V., Chan, Y. F. & van Velzen, D. Human intrauterine renal growth expressed in absolute number of glomeruli assessed by the disector method and Cavalieri principle. Lab. Invest. 64, 777–784 (1991).
Sutherland, M. R. et al. Accelerated maturation and abnormal morphology in the preterm neonatal kidney. J. Am. Soc. Nephrol. 22, 1365–1374 (2011).
Sutherland, M. R. et al. Neonatal hyperoxia: effects on nephrogenesis and long-term glomerular structure. Am. J. Physiol. Ren. Physiol. 304, F1308–F1316 (2013).
Bryan, C. et al. TNFalpha-stimulated protein 6 (TSG-6) reduces lung inflammation in an experimental model of bronchopulmonary dysplasia. Pediatr. Res. 85, 390–397 (2019).
Vaidya, R. et al. Recombinant CCN1 prevents hyperoxia induced lung injury in neonatal rats. Pediatr. Res. 82, 863–871 (2017).
Reiter, J. et al. Stromal derived factor-1 mediates the lung regenerative effects of mesenchymal stem cells in a rodent model of bronchopulmonary dysplasia. Respir. Res. 18, 137 (2017).
Jiang, J. S., Chou, H. C., Yeh, T. F. & Chen, C. M. Neonatal hyperoxia exposure induces kidney fibrosis in rats. Pediatr. Neonatol. 56, 235–241 (2015).
Kuro-o, M. et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 390, 45–51 (1997).
Hu, M. C., Matsumura, Y., Aizawa, H. & Moe, O. W. Fibroblast growth factor 23 and Klotho: physiology and pathophysiology of an endocrine network of mineral metabolism. Annu. Rev. Physiol. 75, 503–533 (2013).
Kuro-o, M. A potential link between phosphate and aging-lessons from Klotho-deficient mice. Mech. Ageing Dev. 131, 270–275 (2010).
Barker, S. L. et al. The demonstration of alphaKlotho deficiency in human chronic kidney disease with a novel synthetic antibody. Nephrol. Dial. Transpl. 30, 223–233 (2015).
Christov, M., Neyra, J. A., Gupta, S. & Leaf, D. E. Fibroblast growth factor 23 and Klotho in AKI. Semin. Nephrol. 39, 57–75 (2019).
Yamamoto, M. et al. Regulation of oxidative stress by the anti-aging hormone klotho. J. Biol. Chem. 280, 38029–38034 (2005).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Bresciani, G., da Cruz, I. B. & Gonzalez-Gallego, J. Manganese superoxide dismutase and oxidative stress modulation. Adv. Clin. Chem. 68, 87–130 (2015).
Ahn, J. et al. Associations between catalase phenotype and genotype: modification by epidemiologic factors. Cancer Epidemiol. Biomark. Prev. 15, 1217–1222 (2006).
Popescu, C. R. et al. Hyperoxia exposure impairs nephrogenesis in the neonatal rat: role of HIF-1alpha. PLoS ONE 8, e82421 (2013).
Kamlin, C. O., O’Donnell, C. P., Davis, P. G. & Morley, C. J. Oxygen saturation in healthy infants immediately after birth. J. Pediatr. 148, 585–589 (2006).
Rabi, Y., Yee, W., Chen, S. Y. & Singhal, N. Oxygen saturation trends immediately after birth. J. Pediatr. 148, 590–594 (2006).
Saugstad, O. D. Update on oxygen radical disease in neonatology. Curr. Opin. Obstet. Gynecol. 13, 147–153 (2001).
Marquez, M. G., Cabrera, I., Serrano, D. J. & Sterin-Speziale, N. Cell proliferation and morphometric changes in the rat kidney during postnatal development. Anat. Embryol. (Berl.) 205, 431–440 (2002).
Yzydorczyk, C. et al. Neonatal oxygen exposure in rats leads to cardiovascular and renal alterations in adulthood. Hypertension 52, 889–895 (2008).
Sutherland, M. R. et al. Age- and sex-related changes in rat renal function and pathology following neonatal hyperoxia exposure. Physiol. Rep. 4, e12887 (2016).
Puelles, V. G. et al. Estimating individual glomerular volume in the human kidney: clinical perspectives. Nephrol. Dial. Transpl. 27, 1880–1888 (2012).
Hu, M. C. et al. Recombinant alpha-Klotho may be prophylactic and therapeutic for acute to chronic kidney disease progression and uremic cardiomyopathy. Kidney Int. 91, 1104–1114 (2017).
Ravikumar, P. et al. alpha-Klotho protects against oxidative damage in pulmonary epithelia. Am. J. Physiol. Lung Cell. Mol. Physiol. 307, L566–L575 (2014).
Sakan, H. et al. Reduced renal α-Klotho expression in CKD patients and its effect on renal phosphate handline and vitamin D metabolism. PLoS ONE 23, e86301 (2014).
Wang, R. et al. Reduced NRF2 expression suppresses endothelial progenitor cell function and induces senescence during aging. Aging 11, 7021–7035 (2019).
Saleem, M., Wang, X., Pokkunuri, I. & Asghar, M. Superoxide via Sp3 mechanism increases renal renin activity, renal AT1 receptor function, and blood pressure in rats. Am. J. Physiol. Ren. Physiol. 315, F1478–F1483 (2018).
Acknowledgements
This work was supported by grant funding from the Batchelor Children’s Research Awards to C.L.A. and K.C.Y. L.A.S. is funded by grants from the NIH (1R01HL140468) and the Miami Heart Research Institute. K.Y. is a recipient of AHA predoctoral fellowship (18PRE33960070).
Author information
Authors and Affiliations
Contributions
All authors provided substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data. M.F.A., S.K.B.V., M.B., S.K., M.F., C.L.A., M.J.D., and K.C.Y. contributed to the drafting of the article and/or critical revision for important intellectual content, and all authors approved the final version of the submitted manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ali, M.F., Venkatarayappa, S.K.B., Benny, M. et al. Effects of Klotho supplementation on hyperoxia-induced renal injury in a rodent model of postnatal nephrogenesis. Pediatr Res 88, 565–570 (2020). https://doi.org/10.1038/s41390-020-0803-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-020-0803-z
This article is cited by
-
A novel mouse model for investigating the long-term impact of reduced nephron numbers on renal function and salt sensitivity
Pediatric Research (2025)
-
Klotho in aging and diseases: molecular features, expressions, and functional implications
Molecular and Cellular Biochemistry (2025)
-
Neonatal hyperoxia exposure leads to developmental programming of cardiovascular and renal disease in adult rats
Scientific Reports (2024)
-
From premature birth to premature kidney disease: does accelerated aging play a role?
Pediatric Nephrology (2024)
-
The danger of hyperoxia on the rat kidneys: is tadalafil a real shield?
International Urology and Nephrology (2022)