Abstract
Background
Late-Onset Neonatal Sepsis (LOS) is a rare condition, involving widespread infection, immune disruption, organ dysfunction, and often death. Because exposure to pathogens is not completely preventable, identifying susceptibility factors is critical to characterizing the pathophysiology and developing interventions. Prior studies demonstrated both genetics and infant sex influence susceptibility. Our study was designed to identify LOS associated genetic variants.
Methods
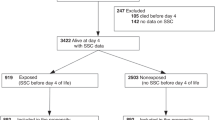
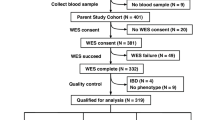
We performed an exploratory genome wide association study (GWAS) with 224 LOS cases and 273 controls from six European countries. LOS was defined as sepsis presenting from 3 to 90 days of age; diagnosis was established by clinical criteria consensus guidelines. We tested for association with both autosomal and X-chromosome variants in the total sample and in sex-stratified analyses.
Results
In total, 71 SNPs associated with neonatal sepsis at p < 1 × 10−4 in at least one analysis. Most importantly, sex-stratified analyses revealed associations with multiple SNPs (28 in males and 16 in females), but no variants from single-sex analyses associated with sepsis in the other sex. Pathway analyses showed NOTCH signaling is over-represented among genes linked to these SNPS.
Conclusion
Our results indicate genetic susceptibility to LOS is sexually dimorphic and corroborate that NOTCH signaling plays a role in determining risk.
Impact
-
Genes associate with late onset neonatal sepsis.
-
Notch pathway genes are overrepresented in associations with sepsis.
-
Genes associating with sepsis do not overlap between males and females.
-
Sexual dimorphism can lead to sex specific treatment of sepsis.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Lutsar, I. et al. Meropenem vs standard of care for treatment of late onset sepsis in children of less than 90 days of age: study protocol for a randomised controlled trial. Trials 12, 215 (2011).
Rossi, P. et al. Report on the Expert Meeting on Neonatal and Paediatric Sepsis (European Medicines Agency 2010).
Goldstein, B., Giroir, B. & Randolph, A. International pediatric sepsis consensus conference: definitions for sepsis and organ dysfunction in pediatrics. Pediatr. Crit. Care Med. 6, 2–8 (2005).
Baltimore, R. S. Neonatal sepsis: epidemiology and management. Paediatr. Drugs 5, 723–740 (2003).
Zaidi, A. K. et al. Hospital-acquired neonatal Infections in developing countries. Lancet 365, 1175–1188 (2005).
Qazi, S. A. & Stoll, B. J. Neonatal sepsis: a major global public health challenge. Pediatr. Infect. Dis. J. 28, S1–S2 (2009).
Garland, J. S. & Uhing, M. R. Strategies to prevent bacterial and fungal infection in the neonatal intensive care unit. Clin. Perinatol. 36, 1–13 (2009).
Washburn, T. C., Medearis, D. N. Jr. & Childs, B. Sex differences in susceptibility to infections. Pediatrics 35, 57–64 (1965).
Dutta, S. et al. Intrapartum antibiotics and risk factors for early onset sepsis. Arch. Dis. Child. Fetal Neonatal Ed. 95, F99–F103 (2010).
Jaillon, S., Berthenet, K. & Garlanda, C. Sexual dimorphism in innate immunity. Clin. Rev. Allergy Immunol. 56, 308–321 (2019).
van Lunzen, J. & Altfeld, M. Sex differences in infectious diseases-common but neglected. J. Infect. Dis. 209, S79–S80 (2014).
Muenchhoff, M. & Goulder, P. J. Sex differences in pediatric infectious diseases. J. Infect. Dis. 209, S120–S126 (2014).
Bizzarro, M. J. et al. The impact of environmental and genetic factors on neonatal late-onset sepsis. J. Pediatrics 158, 234–238.e231 (2011).
Klein, S. L. & Flanagan, K. L. Sex differences in immune responses. Nat. Rev. Immunol. 16, 626–638 (2016).
Koch, M. A. Sex bias in sepsis. Cell Host Microbe 24, 613–615 (2018).
Powers, M. S., Smith, P. H., McKee, S. A. & Ehringer, M. A. From sexless to sexy: why it is time for human genetics to consider and report analyses of sex. Biol. Sex. Differences 8, 15 (2017).
Accounting for sex in the genome. Nat. Med. 23, 1243 (2017).
Heidari, S., Babor, T. F., De Castro, P., Tort, S. & Curno, M. Sex and gender equity in research: rationale for the sager guidelines and Recommended Use. Res. Integr. peer Rev. 1, 2 (2016).
Khramtsova, E. A., Davis, L. K. & Stranger, B. E. The role of sex in the genomics of human complex traits. Nat. Rev. Genet. 20, 173–190 (2019).
Clayton, J. A. Applying the New Sabv (sex as a biological variable) policy to research and clinical care. Physiol. Behav. 187, 2–5 (2018).
Germovsek, E. et al. Plasma and Csf pharmacokinetics of meropenem in neonates and young infants: results from the neomero studies. J. Antimicrob. Chemother. 73, 1908–1916 (2018).
Lutsar, I. et al. Meropenem vs standard of care for treatment of neonatal late onset sepsis (Neomero1): a randomised controlled trial. PLoS ONE 15, e0229380 (2020).
Corte-Real, H. B. et al. Genetic diversity in the Iberian peninsula determined from mitochondrial sequence analysis. Ann. Hum. Genet. 60, 331–350 (1996).
Raveane, A. et al. Population structure of modern-day Italians reveals patterns of ancient and archaic ancestries in southern Europe. Sci. Adv. 5, eaaw3492 (2019).
Malferrari, G. et al. High-quality genomic DNA from human whole blood and mononuclear cells. Biotechniques 33, 1228–1230 (2002).
Srinivasan, L. et al. Genome-wide association study of sepsis in extremely premature infants. Arch. Dis. Child. Fetal neonatal Ed. 102, F439–f445 (2017).
Williams, S. M. & Haines, J. L. Correcting away the hidden heritability. Ann. Hum. Genet. 75, 348–350 (2011).
Bender, R. & Lange, S. Multiple test procedures other than Bonferroni’s deserve wider use. BMJ 318, 600–601 (1999).
Kraft, P. Curses-Winner’s and otherwise–in genetic epidemiology. Epidemiology 19, 649–651 (2008). discussion 657-648.
Sobota, R. S. et al. Addressing population-specific multiple testing burdens in genetic association studies. Ann. Hum. Genet. 79, 136–147 (2015).
Rothman, K. J. No adjustments are needed for multiple comparisons. Epidemiology 1, 43–46 (1990).
Ciesielski, T. H. et al. Diverse convergent evidence in the genetic analysis of complex disease: coordinating omic, informatic, and experimental evidence to better identify and validate risk factors. BioData Min. 7, 10 (2014).
Fabregat, A. et al. The reactome pathway knowledgebase. Nucleic acids Res. 44, D481–D487 (2016).
Fabregat, A. et al. Reactome pathway analysis: a high-performance in-memory approach. BMC Bioinforma. 18, 142 (2017).
Srinivasan, L., Swarr, D. T., Sharma, M., Cotten, C. M. & Kirpalani, H. Systematic review and meta-analysis: gene association studies in neonatal sepsis. Am. J. Perinatol. 34, 684–692 (2017).
Zaykin, D. V. & Zhivotovsky, L. A. Ranks of genuine associations in whole-genome scans. Genetics 171, 813–823 (2005).
Panagiotou, O. A. & Ioannidis, J. P. What should the genome-wide significance threshold be? Empirical replication of borderline genetic associations. Int. J. Epidemiol. 41, 273–286 (2012).
Lindenmann, J., Burke, D. C. & Isaacs, A. Studies on the production, mode of action and properties of interferon. Br. J. Exp. Pathol. 38, 551–562 (1957).
Stinebring, W. R. & Youngner, J. S. Patterns of interferon appearance in mice Injected with bacteria or bacterial endotoxin. Nature 204, 712–712 (1964).
Machado, J. R. et al. Neonatal sepsis and inflammatory mediators. Mediat. Inflammation 2014, 269681 (2014).
Hibbert, J. A.-O. et al. Plasma cytokine profiles in very preterm infants with late-onset sepsis. PLoS ONE 15, e0232933 (2020).
Boghossian, N. S. et al. Late-onset sepsis in very low birth weight infants from singleton and multiple-gestation births. J. Pediatrics 162, 1120–1124 (2013).
Saleem, S. J. & Conrad, D. H. Hematopoietic cytokine-induced transcriptional regulation and notch signaling as modulators of Mdsc expansion. Int. Immunopharmacol. 11, 808–815 (2011).
Tsao, P. N. et al. Lipopolysaccharide-induced notch signaling activation through Jnk-dependent pathway regulates inflammatory response. J. Biomed. Sci. 18, 56 (2011).
Pan, T. et al. Notch signaling pathway was involved in regulating programmed cell death 1 expression during sepsis-induced immunosuppression. Mediators Inflamm. 2015, 539841 (2015).
Skirecki, T. et al. Early severe impairment of hematopoietic stem and progenitor cells from the bone marrow caused by Clp sepsis and endotoxemia in a humanized mice model. Stem Cell Res. Ther. 6, 142 (2015).
Netzer, C. et al. Apoptotic diminution of immature single and double positive thymocyte subpopulations contributes to thymus involution during murine polymicrobial sepsis. Shock 48, 215–226 (2017).
Ge, Y., Huang, M. & Ma, Y. F. The effects of Microrna-34a regulating notch-1/Nf-Kappab signaling pathway on lipopolysaccharide-induced human umbilical vein endothelial cells. World J. Emerg. Med. 8, 292–296 (2017).
Yuyun, X. et al. Asiatic acid attenuates lipopolysaccharide-induced injury by suppressing activation of the notch signaling pathway. Oncotarget 9, 15036–15046 (2018).
Bai, X. et al. Acetylation-dependent regulation of notch signaling in macrophages by Sirt1 affects sepsis development. Front. Immunol. 9, 762 (2018).
Long, J. et al. Notch signaling protects Cd4 T cells from sting-mediated apoptosis during acute systemic inflammation. Sci Adv. 6, eabc5447 (2020).
Hotchkiss, R. S., Monneret, G. & Payen, D. Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nat. Rev. Immunol. 13, 862–874 (2013).
van der Poll, T., van de Veerdonk, F. L., Scicluna, B. P. & Netea, M. G. The immunopathology of sepsis and potential therapeutic targets. Nat. Rev. Immunol. 17, 407–420 (2017).
Boomer, J. S. et al. Immunosuppression in patients who die of sepsis and multiple organ failure. JAMA 306, 2594–2605 (2011).
Helbig, C. et al. Notch controls the magnitude of T helper cell responses by promoting cellular longevity. PNAS 109, 9041–9046 (2012).
Flannery, D. D. & Puopolo, K. M. The imperfect science of neonatal sepsis. Pediatric Res. 91, 733–734 (2021).
Kidszun, A. et al. Viral Infections in Neonates with Suspected Late-Onset Bacterial Sepsis-a Prospective Cohort Study. Am J Perinatol. 34, 1–7 (2017).
Wilcox, A. J., Weinberg, C. R. & Basso, O. On the pitfalls of adjusting for gestational age at birth. Am. J. Epidemiol. 174, 1062–1068 (2011).
Sirugo, G., Williams, S. M. & Tishkoff, S. A. The missing diversity in human genetic studies. Cell 177, 26–31 (2019).
Quinn, J. A. et al. Preterm birth: case definition & guidelines for data collection, analysis, and presentation of immunisation safety data. Vaccine 34, 6047–6056 (2016).
Funding
This work was supported by the European Commission under the FP7 (ID: 242146).
Author information
Authors and Affiliations
Consortia
Contributions
Substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data: T.H.C., X.Z., A.T., I.L., V.M.dC., E.R., C.C., P.B., W.K.S., S.M.W., G.S. Drafting the article or revising it critically for important intellectual content: T.H.C., X.Z., I.L., S.M.W., G.S. Final approval of the version to be published: All authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent to participate
Written consent was provided by the neonates’ parents for inclusion in this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ciesielski, T.H., Zhang, X., Tacconelli, A. et al. Late-onset neonatal sepsis: genetic differences by sex and involvement of the NOTCH pathway. Pediatr Res 93, 1085–1095 (2023). https://doi.org/10.1038/s41390-022-02114-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-022-02114-8
This article is cited by
-
Mind the “CRP gender gap”! sex differences in CRP evolution over time in neonatal sepsis: a monocentric retrospective cohort study
Biology of Sex Differences (2026)
-
Concordance between male- and female-specific GWAS results helps define underlying genetic architecture of complex traits
Nature Communications (2025)
-
Characterizing the pathogenicity of genetic variants: the consequences of context
npj Genomic Medicine (2024)
-
Association of red cell distribution width (RDW) with positive blood culture of neonatal sepsis in Khartoum state, Sudan
Discover Medicine (2024)