Abstract
Background
The aim of this study was to determine the relationship between iron exposure and the development of bronchopulmonary dysplasia (BPD).
Methods
A secondary analysis of the PENUT Trial dataset was conducted. The primary outcome was BPD at 36 weeks gestational age and primary exposures of interest were cumulative iron exposures in the first 28 days and through 36 weeks’ gestation. Descriptive statistics were calculated for study cohort characteristics with analysis adjusted for the factors used to stratify randomization.
Results
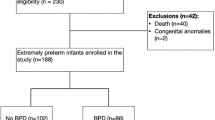
Of the 941 patients, 821 (87.2%) survived to BPD evaluation at 36 weeks, with 332 (40.4%) diagnosed with BPD. The median cohort gestational age was 26 weeks and birth weight 810 g. In the first 28 days, 76% of infants received enteral iron and 55% parenteral iron. The median supplemental cumulative enteral and parenteral iron intakes at 28 days were 58.5 and 3.1 mg/kg, respectively, and through 36 weeks’ 235.8 and 3.56 mg/kg, respectively. We found lower volume of red blood cell transfusions in the first 28 days after birth and higher enteral iron exposure in the first 28 days after birth to be associated with lower rates of BPD.
Conclusions
We find no support for an increased risk of BPD with iron supplementation.
Trial registration number
NCT01378273. https://clinicaltrials.gov/ct2/show/NCT01378273
Impact
-
Prior studies and biologic plausibility raise the possibility that iron administration could contribute to the pathophysiology of oxidant-induced lung injury and thus bronchopulmonary dysplasia in preterm infants.
-
For 24–27-week premature infants, this study finds no association between total cumulative enteral iron supplementation at either 28-day or 36-week postmenstrual age and the risk for developing bronchopulmonary dysplasia.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The datasets analyzed for this study are available through the NINDS Archived Clinical Research Datasets at https://www.ninds.nih.gov/Current-Research/Research-Funded-NINDS/Clinical-Research/Archived-Clinical-Research-Datasets.
References
Stoll, B. J. et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics 126, 443–456 (2010).
Shah, P. S. et al. Outcomes of preterm infants <29 weeks gestation over 10-year period in Canada: a cause for concern? J. Perinatol. 32, 132–138 (2012).
Stoll, B. J. et al. Trends in care practices, morbidity, and mortality of extremely preterm neonates, 1993-2012. JAMA 314, 1039–1051 (2015).
Jobe, A. H. Mechanisms of lung injury and bronchopulmonary dysplasia. Am. J. Perinatol. 33, 1076–1078 (2016).
Saugstad, O. D. Oxygen and oxidative stress in bronchopulmonary dysplasia. J. Perinat. Med. 38, 571–577 (2010).
Perrone, S. et al. The free radical diseases of prematurity: from cellular mechanisms to bedside. Oxid. Med. Cell Longev. 2018, 7483062 (2018).
Wang, J. & Dong, W. Oxidative stress and bronchopulmonary dysplasia. Gene 678, 177–183 (2018).
Lozoff, B. & Georgieff, M. K. Iron deficiency and brain development. Semin. Pediatr. Neurol. 13, 158–165 (2006).
Evstatiev, R. & Gasche, C. Iron sensing and signalling. Gut 61, 933–952 (2012).
Widdowson, E. M. & Spray, C. M. Chemical development in utero. Arch. Dis. Child. 26, 205–214 (1951).
Baker, R. D. & Greer, F. R. Committee on Nutrition American Academy of Pediatrics. Diagnosis and prevention of iron deficiency and iron-deficiency anemia in infants and young children (0-3 years of age). Pediatrics 126, 1040–1050 (2010).
Agostoni, C. et al. Enteral nutrient supply for preterm infants: commentary from the European Society of Paediatric Gastroenterology, Hepatology and Nutrition Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 50, 85–91 (2010).
Franz, A. R., Mihatsch, W. A., Sander, S., Kron, M. & Pohlandt, F. Prospective randomized trial of early versus late enteral iron supplementation in infants with a birth weight of less than 1301 grams. Pediatrics 106, 700–706 (2000).
Joy, R. et al. Early versus late enteral prophylactic iron supplementation in preterm very low birth weight infants: a randomised controlled trial. Arch. Dis. Child. Fetal Neonatal Ed. 99, F105–F109 (2014).
Mills, R. J. & Davies, M. W. Enteral iron supplementation in preterm and low birth weight infants. Cochrane Database Syst. Rev. 14, CD005095 (2012).
Siddappa, A. M. et al. High prevalence of iron deficiency despite standardized high-dose iron supplementation during recombinant erythropoietin therapy in extremely low gestational age newborns. J. Pediatr. 222, 98.e3–105.e3 (2020).
Patel, M. & Ramavataram, D. V. S. S. Non transferrin bound iron: nature, manifestations and analytical approaches for estimation. Ind. J. Clin. Biochem. 27, 322–332 (2012).
Brissot, P., Ropert, M., Le Lan, C. & Loreal, O. Non-transferrin bound iron: a key role in iron overload and iron toxicity. Biochem. Biophys. Acta 1820, 403–410 (2012).
Cooke, R. W., Drury, J. A., Yoxall, C. W. & James, C. Blood transfusion and chronic lung disease in preterm infants. Eur. J. Pediatr. 156, 47–50 (1997).
Collard, K. J. Is there a causal relationship between the receipt of blood transfusions and the development of chronic lung disease of prematurity? Med. Hypotheses 66, 355–364 (2006).
Valieva, O. A., Strandjord, T. P., Mayock, D. E. & Juul, S. E. Effects of transfusions in extremely low birth weight infants: a retrospective study. J. Pediatr. 155, 331.e1–37 (2009).
Zhang, Z., Huang, X. & Lu, H. Association between red blood cell transfusion and bronchopulmonary dysplasia in preterm infants. Sci. Rep. 4, 4340 (2014).
Ghirardello, S. et al. Effects of red blood cell transfusions on the risk of developing complications or death: an observational study of a cohort of very low birth weight infants. Am. J. Perinatol. 34, 88–95 (2017).
Braekke, K. et al. Oxidative stress markers and antioxidant status after oral iron supplementation to very low birth weight infants. J. Pediatr. 151, 23–28 (2007).
Miller, S. M., McPherson, R. J. & Juul, S. E. Iron sulfate supplementation decreases zinc protoporphyrin to heme ratio in premature infants. J. Pediatr. 148, 44–48 (2006).
Patel, R. M. et al. Enteral iron supplementation, red blood cell transfusion, and risk of bronchopulmonary dysplasia in very-low-birth-weight infants. Transfusion 59, 1675–1682 (2019).
Juul, S. E. et al. A randomized trial of erythropoietin for neuroprotection in preterm infants. N. Engl. J. Med. 382, 233–243 (2020).
Liang, K.-Y. & Zeger, S. L. Longitudinal data analysis using generalized linear models. Biometrika 73, 13–22 (1986).
Murphy, T., Chawla, A., Tucker, R. & Vohr, B. Impact of blood donor sex on transfusion-related outcomes in preterm infants. J. Pediatr. 201, 215–220 (2018).
Bell, E. F. et al. Randomized trial of liberal versus restrictive guidelines for red blood cell transfusion in preterm infants. Pediatrics 115, 1685–1691 (2005).
Kirpalani, H. et al. The Premature Infants in Need of Transfusion (PINT) study: a randomized, controlled trial of a restrictive (low) versus liberal (high) transfusion threshold for extremely low birth weight infants. J. Pediatr. 149, 301–307 (2006).
Franz, A. R. et al. Effects of liberal vs restrictive transfusion thresholds on survival and neurocognitive outcomes in extremely low-birth-weight infants: the ETTNO randomized clinical trial. JAMA 324, 560–570 (2020).
Kirpalani, H. et al. Higher or lower hemoglobin transfusion thresholds for preterm infants. N. Engl. J. Med. 383, 2639–2651 (2020).
Manuck, T. A., Levy, P. T., Gyamfi-Bannerman, C., Jobe, A. H. & Blaisdell, C. J. Prenatal and perinatal determinants of lung health and disease in early life: a National Heart, Lung, and Blood Institute Workshop Report. JAMA Pediatr. 170, e154577 (2016).
Rao, R. & Georgieff, M. K. Iron therapy for preterm infants. Clin. Perinatol. 36, 27–42 (2009).
Funding
This work was funded by NINDS U01NS077955 and U01NS077953.
Author information
Authors and Affiliations
Consortia
Contributions
M.R.G. contributed to study conception and design, interpretation of data, drafting the article, revising it critically for important intellectual content, and final approval of the version to be published. B.A.C. substantially contributed to study design, acquisition of data, analysis of data, interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published. R.M.P., V.N.T., and C.D.J. substantially contributed to study conception and design, interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published. M.K.G., R.R., and S.E.J. substantially contributed to study conception and design, acquisition of data, interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published. S.E.M. substantially contributed to study design, analysis and interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published. K.A.A. substantially contributed to study conception and design, acquisition of data, interpretation of data, drafting the article, revising the article critically for important intellectual content, and final approval of the version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Added individual patient consent was not required for this study, which utilized deidentified patient data. All patient families consented for enrollment in the PENUT Trial. Patient consent was obtained for the original PENUT Trial enrollment but was not required for this secondary study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Garcia, M.R., Comstock, B.A., Patel, R.M. et al. Iron supplementation and the risk of bronchopulmonary dysplasia in extremely low gestational age newborns. Pediatr Res 93, 701–707 (2023). https://doi.org/10.1038/s41390-022-02160-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-022-02160-2