Abstract
Background
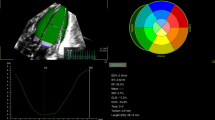
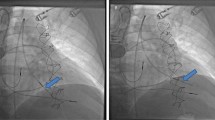
Definitive closure of the patent ductus arteriosus (PDA) is associated with significant changes in the loading conditions of the left ventricle (LV), which may lead to cardiovascular and respiratory instability. The objective of the study was to evaluate targeted neonatal echocardiography (TnECHO) characteristics and the clinical course of preterm infants ≤2 kg undergoing percutaneous PDA closure.
Methods
Retrospective cohort study of prospectively acquired pre- and post-closure TnECHOs to assess hemodynamic changes. Cardiorespiratory parameters in the first 24 h following PDA closure were also evaluated.
Results
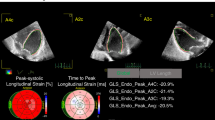
Fifty patients were included with a mean age of 30.6 ± 9.6 days and weight of 1188 ± 280 g. LV global longitudinal strain decreased from −20.6 ± 2.6 to −14.9 ± 2.9% (p < 0.001) after 1 h. There was a decrease in LV volume loading, left ventricular output, LV systolic and diastolic parameters. Cardiorespiratory instability occurred in 24 (48%) [oxygenation failure in 44%] but systolic hypotension and/or need for cardiovascular medications was only seen in 6 (12%). Patients with instability had worse baseline respiratory severity score and lower post-closure early diastolic strain rates.
Conclusions
Percutaneous PDA closure leads to a reduction in echocardiography markers of LV systolic/diastolic function. Post-closure cardiorespiratory instability is characterized primarily by oxygenation failure and may relate to impaired diastolic performance.
Impact
-
Percutaneous patent ductus arteriosus closure leads to a reduction in echocardiography markers of left ventricular volume loading, cardiac output, and left ventricular systolic/diastolic function.
-
Post-procedural cardiorespiratory instability is characterized primarily by oxygenation failure.
-
Post-procedural cardiorespiratory instability may relate to impaired diastolic performance.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Backes, C. H. et al. Percutaneous closure of the patent ductus arteriosus in very low weight infants: considerations following US Food and Drug Administration approval of a novel device. J. Pediatr. 213, 218–221 (2019).
Bischoff, A. R. et al. Percutaneous closure of patent ductus arteriosus in infants </=1.5 Kg: a meta-analysis. J. Pediatr. 230, 84.e14–92.e14 (2020).
Apalodimas, L. et al. A comprehensive program for preterm infants with patent ductus arteriosus. Congenit. Heart Dis. 14, 90–94 (2019).
El-Khuffash, A. F., Jain, A. & McNamara, P. J. Ligation of the patent ductus arteriosus in preterm infants: understanding the physiology. J. Pediatr. 162, 1100–1106 (2013).
El-Khuffash, A. F., Jain, A., Dragulescu, A., McNamara, P. J. & Mertens, L. Acute changes in myocardial systolic function in preterm infants undergoing patent ductus arteriosus ligation: a tissue Doppler and myocardial deformation study. J. Am. Soc. Echocardiogr. 25, 1058–1067 (2012).
Jain, A. et al. Use of targeted neonatal echocardiography to prevent postoperative cardiorespiratory instability after patent ductus arteriosus ligation. J. Pediatr. 160, 584–589 e581 (2012).
Giesinger, R. E., Bischoff, A. R. & McNamara, P. J. Anticipatory perioperative management for patent ductus arteriosus surgery: understanding postligation cardiac syndrome. Congenit. Heart Dis. 14, 311–316 (2019).
Teixeira, L. S., Shivananda, S. P., Stephens, D., Van Arsdell, G. & McNamara, P. J. Postoperative cardiorespiratory instability following ligation of the preterm ductus arteriosus is related to early need for intervention. J. Perinatol. 28, 803–810 (2008).
Weisz, D. E., Jain, A., Ting, J., McNamara, P. J. & El-Khuffash, A. Non-invasive cardiac output monitoring in preterm infants undergoing patent ductus arteriosus ligation: a comparison with echocardiography. Neonatology 106, 330–336 (2014).
Ting, J. Y. et al. Predictors of respiratory instability in neonates undergoing patient ductus arteriosus ligation after the introduction of targeted milrinone treatment. J. Thorac. Cardiovasc. Surg. 152, 498–504 (2016).
Clyman, R. I. et al. Hypotension following patent ductus arteriosus ligation: the role of adrenal hormones. J. Pediatr. 164, 1449.e1–1455.e1 (2014).
Moin, F., Kennedy, K. A. & Moya, F. R. Risk factors predicting vasopressor use after patent ductus arteriosus ligation. Am. J. Perinatol. 20, 313–320 (2003).
Harting, M. T. et al. Acute hemodynamic decompensation following patent ductus arteriosus ligation in premature infants. J. Invest. Surg. 21, 133–138 (2008).
McNamara, P. J., Stewart, L., Shivananda, S. P., Stephens, D. & Sehgal, A. Patent ductus arteriosus ligation is associated with impaired left ventricular systolic performance in premature infants weighing less than 1000 g. J. Thorac. Cardiovasc. Surg. 140, 150–157 (2010).
Abu Hazeem, A. A. et al. Percutaneous closure of patent ductus arteriosus in small infants with significant lung disease may offer faster recovery of respiratory function when compared to surgical ligation. Catheter Cardiovasc. Inter. 82, 526–533 (2013).
Regan, W. et al. Improved ventilation in premature babies after transcatheter versus surgical closure of patent ductus arteriosus. Int. J. Cardiol. 311, 22–27 (2020).
Rodriguez Ogando, A. et al. Surgical ligation versus percutaneous closure of patent ductus arteriosus in very low-weight preterm infants: which are the real benefits of the percutaneous approach? Pediatr. Cardiol. 39, 398–410 (2018).
Kim, H. S. et al. Surgical versus percutaneous closure of pda in preterm infants: procedural charges and outcomes. J. Surg. Res. 243, 41–46 (2019).
Serrano, R. M., Madison, M., Lorant, D., Hoyer, M. & Alexy, R. Comparison of ‘post-patent ductus arteriosus ligation syndrome’ in premature infants after surgical ligation vs. percutaneous closure. J. Perinatol. 40, 324–329 (2019).
Bischoff, A. R., Stanford, A. H. & McNamara, P. J. Short-term ventriculo-arterial coupling and myocardial work efficiency in preterm infants undergoing percutaneous patent ductus arteriosus closure. Physiol. Rep. 9, e15108 (2021).
Bischoff, A. R. et al. Clinical and echocardiography predictors of response to inhaled nitric oxide in hypoxemic term and near-term neonates. Pediatr. Pulmonol. 56, 982–991 (2021).
Rios, D. R. et al. Early role of the atrial-level communication in premature infants with patent ductus arteriosus. J. Am. Soc. Echocardiogr. 34, 423.e1–432.e1 (2020).
El-Khuffash, A. F., Jain, A., Weisz, D., Mertens, L. & McNamara, P. J. Assessment and treatment of post patent ductus arteriosus ligation syndrome. J. Pediatr. 165, 46.e1–52.e1 (2014).
Konstam, M. A. & Abboud, F. M. Ejection fraction: misunderstood and overrated (changing the paradigm in categorizing heart failure). Circulation 135, 717–719 (2017).
Bussmann, N. & El-Khuffash, A. Future perspectives on the use of deformation analysis to identify the underlying pathophysiological basis for cardiovascular compromise in neonates. Pediatr. Res. 85, 591–595 (2019).
Rowland, D. G. & Gutgesell, H. P. Use of mean arterial pressure for noninvasive determination of left ventricular end-systolic wall stress in infants and children. Am. J. Cardiol. 74, 98–99 (1994).
Schmitz, L., Stiller, B., Koch, H., Koehne, P. & Lange, P. Diastolic left ventricular function in preterm infants with a patent ductus arteriosus: a serial Doppler echocardiography study. Early Hum. Dev. 76, 91–100 (2004).
Reller, M. D., Morton, M. J., Reid, D. L. & Thornburg, K. L. Fetal lamb ventricles respond differently to filling and arterial pressures and to in utero ventilation. Pediatr. Res. 22, 621–626 (1987).
de Waal, K., Phad, N. & Boyle, A. Left atrium function and deformation in very preterm infants with and without volume load. Echocardiography 35, 1818–1826 (2018).
Payne, R. M., Stone, H. L. & Engelken, E. J. Atrial function during volume loading. J. Appl. Physiol. 31, 326–331 (1971).
Blume, G. G. et al. Left atrial function: physiology, assessment, and clinical implications. Eur. J. Echocardiogr. 12, 421–430 (2011).
Prioli, A., Marino, P., Lanzoni, L. & Zardini, P. Increasing degrees of left ventricular filling impairment modulate left atrial function in humans. Am. J. Cardiol. 82, 756–761 (1998).
Sunagawa, K., Maughan, W. L., Burkhoff, D. & Sagawa, K. Left ventricular interaction with arterial load studied in isolated canine ventricle. Am. J. Physiol. 245, H773–H780 (1983).
James, A., Corcoran, J. D., Mertens, L., Franklin, O. & El-Khuffash, A. Left ventricular rotational mechanics in preterm infants less than 29 weeks’ gestation over the first week after birth. J. Am. Soc. Echocardiogr. 28, 808–817.e801 (2015).
Al-Naami, G. H. Torsion of young hearts: a speckle tracking study of normal infants, children, and adolescents. Eur. J. Echocardiogr. 11, 853–862 (2010).
Breatnach, C. R. et al. Left ventricular rotational mechanics in infants with hypoxic ischemic encephalopathy and preterm infants at 36 weeks postmenstrual age: a comparison with healthy term controls. Echocardiography 34, 232–239 (2017).
Wang, J., Khoury, D. S., Yue, Y., Torre-Amione, G. & Nagueh, S. F. Preserved left ventricular twist and circumferential deformation, but depressed longitudinal and radial deformation in patients with diastolic heart failure. Eur. Heart J. 29, 1283–1289 (2008).
Bischoff, A. R., Giesinger, R. E., Bell, E. F. & McNamara, P. J. Precision medicine in neonatal hemodynamics: need for prioritization of mechanism of illness and defining population of interest. J. Perinatol. 40, 1446–1449 (2020).
Sehgal, A. et al. The left heart, systemic circulation and bronchopulmonary dysplasia: relevance to pathophysiology and therapeutics. J. Pediatr. 225, 13.e2–22.e2 (2020).
Sehgal, A., Malikiwi, A., Paul, E., Tan, K. & Menahem, S. A new look at bronchopulmonary dysplasia: postcapillary pathophysiology and cardiac dysfunction. Pulm. Circ. 6, 508–515 (2016).
Mourani, P. M., Ivy, D. D., Rosenberg, A. A., Fagan, T. E. & Abman, S. H. Left ventricular diastolic dysfunction in bronchopulmonary dysplasia. J. Pediatr. 152, 291–293 (2008).
Mawad, W. & Friedberg, M. K. The continuing challenge of evaluating diastolic function by echocardiography in children: developing concepts and newer modalities. Curr. Opin. Cardiol. 32, 93–100 (2017).
LeJemtel, T. H., Scortichini, D., Levitt, B. & Sonnenblick, E. H. Effects of phosphodiesterase inhibition on skeletal muscle vasculature. Am. J. Cardiol. 63, 27A–30A (1989).
Silver, P. J. et al. Phosphodiesterase isozyme inhibition, activation of the camp system, and positive inotropy mediated by milrinone in isolated guinea pig cardiac muscle. J. Cardiovasc. Pharm. 13, 530–540 (1989).
Akita, T., Joyner, R. W., Lu, C., Kumar, R. & Hartzell, H. C. Developmental changes in modulation of calcium currents of rabbit ventricular cells by phosphodiesterase inhibitors. Circulation 90, 469–478 (1994).
Author information
Authors and Affiliations
Contributions
A.R.B. participated in the conception, design, acquisition, and analysis of data, drafted the article, and approved the final version. A.H.S. participated in acquisition of data, review of the manuscript for important intellectual content, and approved the final version. O.A., D.I.M., and B.M.N. participated in the acquisition of data, review of the manuscript for important intellectual content, and approved the final version. D.R.R. participated in the conception, acquisition of data, review of the manuscript for important intellectual content, and approved the final version. R.E.G. participated in the conception, design, analysis of data, review of the manuscript for important intellectual content, and approved the final version. P.J.M. participated in the conception, design, review of the manuscript for important intellectual content, and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Patient consent was not required. The study was approved by the University of Iowa Biomedical Institutional Board Review.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bischoff, A.R., Stanford, A.H., Aldoss, O. et al. Left ventricular function before and after percutaneous patent ductus arteriosus closure in preterm infants. Pediatr Res 94, 213–221 (2023). https://doi.org/10.1038/s41390-022-02372-6
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-022-02372-6
This article is cited by
-
Effect of Transcatheter Closure of the Ductus Arteriosus on Right Ventricular Function in Preterm Neonates
Pediatric Cardiology (2026)
-
Echocardiographic Predictors and Incidence of Left Ventricular Dysfunction Following Transcatheter Patent Ductus Arteriosus Closure in Extremely Premature Infants
Pediatric Cardiology (2026)
-
Early drug treatment in preterm patients with large patent ductus arteriosus at 28 weeks or less gestational age: systematic review and meta-analysis
Journal of Perinatology (2025)
-
Biventricular adaptation after patent ductus arteriosus ligation
Pediatric Research (2025)
-
Diastolic function in newborn infants: understanding pathophysiology, diagnosis and clinical relevance
Pediatric Research (2025)