Abstract
Background
Tobacco smoke exposure increases the risk and severity of lower respiratory tract infections in children, yet the mechanisms remain unclear. We hypothesized that tobacco smoke exposure would modify the lower airway microbiome.
Methods
Secondary analysis of a multicenter cohort of 362 children between ages 31 days and 18 years mechanically ventilated for >72 h. Tracheal aspirates from 298 patients, collected within 24 h of intubation, were evaluated via 16 S ribosomal RNA sequencing. Smoke exposure was determined by creatinine corrected urine cotinine levels ≥30 µg/g.
Results
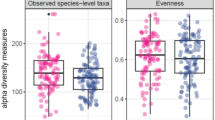
Patients had a median age of 16 (IQR 568) months. The most common admission diagnosis was lower respiratory tract infection (53%). Seventy-four (20%) patients were smoke exposed and exhibited decreased richness and Shannon diversity. Smoke exposed children had higher relative abundances of Serratia spp., Moraxella spp., Haemophilus spp., and Staphylococcus aureus. Differences were most notable in patients with bacterial and viral respiratory infections. There were no differences in development of acute respiratory distress syndrome, days of mechanical ventilation, ventilator free days at 28 days, length of stay, or mortality.
Conclusion
Among critically ill children requiring prolonged mechanical ventilation, tobacco smoke exposure is associated with decreased richness and Shannon diversity and change in microbial communities.
Impact
-
Tobacco smoke exposure is associated with changes in the lower airways microbiome but is not associated with clinical outcomes among critically ill pediatric patients requiring prolonged mechanical ventilation.
-
This study is among the first to evaluate the impact of tobacco smoke exposure on the lower airway microbiome in children.
-
This research helps elucidate the relationship between tobacco smoke exposure and the lower airway microbiome and may provide a possible mechanism by which tobacco smoke exposure increases the risk for poor outcomes in children.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
Deidentified data is publicly available at: https://hheardatacenter.mssm.edu/Search/Study. Microbiome data are available via: PRJNA533819 (data generated from methods development PRJNA436139 were also used for this study).
References
Oberg, M., Jaakkola, M. S., Woodward, A., Peruga, A. & Pruss-Ustun, A. Worldwide burden of disease from exposure to second-hand smoke: a retrospective analysis of data from 192 countries. Lancet 377, 139–146 (2011).
Liu, L. et al. Global, regional, and national causes of under-5 mortality in 2000-15: an updated systematic analysis with implications for the sustainable development goals. Lancet 388, 3027–3035 (2016).
Merianos, A. L., Jandarov, R. A., Choi, K. & Mahabee-Gittens, E. M. Tobacco smoke exposure disparities persist in U.S. children: Nhanes 1999–2014. Prev. Med. 123, 138–142 (2019).
Merianos, A. L., Jandarov, R. A. & Mahabee-Gittens, E. M. Secondhand smoke exposure and pediatric healthcare visits and hospitalizations. Am. J. Prev. Med. 53, 441–448 (2017).
Hur, K., Liang, J. & Lin, S. Y. The role of secondhand smoke in sinusitis: a systematic review. Int. Forum Allergy Rhinol. 4, 22–28 (2014).
Zhang, X., Johnson, N., Carrillo, G. & Xu, X. Decreasing trend in passive tobacco smoke exposure and association with asthma in U.S. children. Environ. Res. 166, 35–41 (2018).
Moore, B. F. et al. Interactions between diet and exposure to secondhand smoke on the prevalence of childhood obesity: results from Nhanes, 2007-2010. Environ. Health Perspect. 124, 1316–1322 (2016).
Wilson, K. M., Pier, J. C., Wesgate, S. C., Cohen, J. M. & Blumkin, A. K. Secondhand tobacco smoke exposure and severity of influenza in hospitalized children. J. Pediatr. 162, 16–21 (2013).
DiFranza, J. R., Masaquel, A., Barrett, A. M., Colosia, A. D. & Mahadevia, P. J. Systematic literature review assessing tobacco smoke exposure as a risk factor for serious respiratory syncytial virus disease among infants and young children. BMC Pediatr. 12, 81 (2012).
Calfee, C. S. et al. Cigarette smoke exposure and the acute respiratory distress syndrome. Crit. Care Med. 43, 1790–1797 (2015).
Hsieh, S. J. et al. Prevalence and impact of active and passive cigarette smoking in acute respiratory distress syndrome. Crit. Care Med. 42, 2058–2068 (2014).
Strzelak, A., Ratajczak, A., Adamiec, A. & Feleszko, W. Tobacco smoke induces and alters immune responses in the lung triggering inflammation, allergy, asthma and other lung diseases: a mechanistic review. Int. J. Environ. Res. Public Health 15, 1033 (2018).
Jette, M. E., Dill-McFarland, K. A., Hanshew, A. S., Suen, G. & Thibeault, S. L. The human laryngeal microbiome: effects of cigarette smoke and reflux. Sci. Rep. 6, 35882 (2016).
Stewart, C. J. et al. Effects of tobacco smoke and electronic cigarette vapor exposure on the oral and gut microbiota in humans: a pilot study. PeerJ 6, e4693 (2018).
Charlson, E. S. et al. Disordered microbial communities in the upper respiratory tract of cigarette smokers. PLoS One 5, e15216 (2010).
Panzer, A. R. et al. Lung microbiota is related to smoking status and to development of acute respiratory distress syndrome in critically Ill trauma patients. Am. J. Respir. Crit. Care Med. 197, 621–631 (2018).
Mourani, P. M. et al. Temporal airway microbiome changes related to ventilator-associated pneumonia in children. Eur. Respir. J. 57, 2001829 (2021).
Zemanick, E. T. et al. Airway microbiota across age and disease spectrum in cystic fibrosis. Eur. Respir. J. 50, 1700832 (2017).
Hara, N. et al. Prevention of virus-induced type 1 diabetes with antibiotic therapy. J. Immunol. 189, 3805–3814 (2012).
Markle, J. G. et al. Sex differences in the gut microbiome drive hormone-dependent regulation of autoimmunity. Science 339, 1084–1088 (2013).
Balshaw, D. M., Collman, G. W., Gray, K. A. & Thompson, C. L. The children’s health exposure analysis resource: enabling research into the environmental influences on children’s health outcomes. Curr. Opin. Pediatr. 29, 385–389 (2017).
Murphy, S. E. et al. Nicotine N-Glucuronidation relative to N-Oxidation and C-Oxidation and Ugt2b10 genotype in five ethnic/racial groups. Carcinogenesis 35, 2526–2533 (2014).
Benowitz, N. L. et al. Urine nicotine metabolite concentrations in relation to plasma cotinine during low-level nicotine exposure. Nicotine Tob. Res. 11, 954–960 (2009).
Benowitz, N. L. et al. Urine 4-(Methylnitrosamino)-1-(3) Pyridyl-1-Butanol and cotinine in alaska native postpartum women and neonates comparing smokers and smokeless tobacco users. Int J. Circumpolar Health 77, 1528125 (2018).
Benowitz, N. L. et al. Urine cotinine screening detects nearly ubiquitous tobacco smoke exposure in urban adolescents. Nicotine Tob. Res. 19, 1048–1054 (2017).
Torres, S., Merino, C., Paton, B., Correig, X. & Ramirez, N. Biomarkers of exposure to secondhand and thirdhand tobacco smoke: recent advances and future perspectives. Int. J. Environ. Res. Public Health 15, 2693 (2018).
Fried, P. A., Perkins, S. L., Watkinson, B. & McCartney, J. S. Association between creatinine-adjusted and unadjusted urine cotinine values in children and the mother’s report of exposure to environmental tobacco smoke. Clin. Biochem. 28, 415–420 (1995).
Henderson, F. W. et al. Home air nicotine levels and urinary cotinine excretion in preschool children. Am. Rev. Respir. Dis. 140, 197–201 (1989).
Zhang, C. et al. Unintentional tobacco smoke exposure in children. Int. J. Environ. Res. Public Health 19, 7076 (2022).
Langelier, C. et al. Integrating host response and unbiased microbe detection for lower respiratory tract infection diagnosis in critically Ill adults. Proc. Natl Acad. Sci. USA 115, E12353–E12362 (2018).
Force, A. D. T. et al. Acute respiratory distress syndrome: the berlin definition. JAMA 307, 2526–2533 (2012).
Pollack, M. M., Patel, K. M. & Ruttimann, U. E. Prism Iii: an updated pediatric risk of mortality score. Crit. Care Med. 24, 743–752 (1996).
Williamson, K. M. et al. Modified Pcr protocol to increase sensitivity for determination of bacterial community composition. Microbiome 9, 90 (2021).
Keskinoglu, P., Cimrin, D. & Aksakoglu, G. Which cut-off level of urine cotinine:creatinine ratio (Ccr) should be used to determine passive smoking prevalence in children in community based studies? Tob. Control 16, 358–359 (2007).
Dickson, R. P. The lung microbiome and ards. it is time to broaden the model. Am. J. Respir. Crit. Care Med. 197, 549–551 (2018).
Fritz, C. Q. et al. Prevalence, risk factors, and outcomes of bacteremic pneumonia in children. Pediatrics 144, e20183090 (2019).
Srinivasan, R., Asselin, J., Gildengorin, G., Wiener-Kronish, J. & Flori, H. R. A prospective study of ventilator-associated pneumonia in children. Pediatrics 123, 1108–1115 (2009).
Avila-Tang, E. et al. Assessing secondhand smoke exposure with reported measures. Tob. Control 22, 156–163 (2013).
McCarville, M., Sohn, M. W., Oh, E., Weiss, K. & Gupta, R. Environmental tobacco smoke and asthma exacerbations and severity: the difference between measured and reported exposure. Arch. Dis. Child 98, 510–514 (2013).
Larsson Wexell, C. et al. Antimicrobial effect of a single dose of amoxicillin on the oral microbiota. Clin. Implant Dent. Relat. Res. 18, 699–706 (2016).
Buffie, C. G. et al. Profound alterations of intestinal microbiota following a single dose of clindamycin results in sustained susceptibility to clostridium difficile-induced colitis. Infect. Immun. 80, 62–73 (2012).
Acknowledgements
This study was supported by NICHD Collaborative Pediatric Critical Care Research Network.
Funding
Supported in part, by the following cooperative agreements from the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), and Heart Lung Blood Institute (NHLBI), National Institutes of Health (NIH): UG1HD083171 (P.M.M.), 1R01HL124103 (P.M.M. and M.K.S.), UG1HD049983 (J.C.), UG01HD049934 (R. Reeder, C. Locandro), UG1HD050096 (K.L.M.), UG1HD083166 (P.S.M.), UG1HD049981 (M.M.P.), and K23HL138461-01A1 (C.L.). NIH/NIEHS funded Human Heath Exposure Analysis Resource (HHEAR) under grant numbers U2CES026555, U2CES026560, and U2CES026553.
Author information
Authors and Affiliations
Contributions
M.K.L., K.M.W., P.C.C., B.D.W., L.A., M.B., S.A.B., S.E.M., L.A.P., K.R.V., C.J.S., E.A.T., J.K.H., A.B.M., C.M.O. and P.M.M. contributed to the design of the study, data analysis and interpretation of the data. K.M.W., M.K.S., B.D.W., J.K.H., R.W.R., C.L., T.C.C., E.A.F.S., C.E.R., C.L., J.A.C., K.L.M., M.M.P., P.S.M., and P.M.M. contributed to data and sample acquisition and conduct of primary study. M.K.L. was responsible for primary authorship of the manuscript. All authors contributed to revision of the content and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
M.K.L. has nothing to disclose. K.M.W. has nothing to disclose. P.C.C. reports grants from NIH during the conduct of the study. M.K.S. reports grants from NIH NHLDI during the conduct of the study. L.A. reports grant funding from Pfizier Inc. outside the submitted work. M.B. has nothing to disclose. S.A.B. has nothing to disclose. S.E.M. has nothing to disclose. L.A.P. has nothing to disclose. J.K.H. has nothing to disclose. R.W.R. has nothing to disclose. C.L. has nothing to disclose. T.C.C. reports grants from NIH NHLBI during the conduct of the study. A.B.M. reports a grant from Parker B. Francis Foundation and NIH NICHD, outside the submitted work. E.A.F.S. reports grants from Astra Zeneca Inc, Merck & Co, Regeneron Inc, Pfizer Inc, Roche Inc, Johnson and Johnson, and Novavax Inc; consulting fees from Merck & Co, Pfizer Inc, Alere Inc, Cidara Therapeutics, and Sanofi Pasteur; support from travel from Merck & Co and Pfizer Inc; participation on Data Safety and Monitoring Boards with Abbvie, Glasco Smith Klein and the Bill and Melinda Gates Foundation all outside of the submitted work. C.M.O. has nothing to disclose. C.E.R. has nothing to disclose. C.L. has nothing to disclose. J.A.C. has nothing to disclose. K.L.M. has nothing to disclose. M.M.P. reports grants from the NIH during the conduct of the study. P.S.M. has nothing to disclose. P.M.M. reports grants from NIH NIHLBI and NIH NICHD during the conduct of the study.
Consent to publish/participate
The study was approved by the University of Utah central Institutional Review Board and consent was obtained from all patients.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Leroue, M.K., Williamson, K.M., Curtin, P.C. et al. Tobacco smoke exposure, the lower airways microbiome and outcomes of ventilated children. Pediatr Res 94, 660–667 (2023). https://doi.org/10.1038/s41390-023-02502-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-023-02502-8
This article is cited by
-
The global burden of lower respiratory infections attributable to smoking and secondhand smoke, 1990–2021, with projections to 2050: a systematic analysis of the GBD 2021 study
Journal of Health, Population and Nutrition (2026)
-
Heavy Metal Transfer from Cigarette Smoke to Lung Tissue: Inducing on Pulmonary Function and Bronchoalveolar Microbiome Dysregulation in Rats
Biological Trace Element Research (2025)
-
Oxidative stress and inflammation: elucidating mechanisms of smoking-attributable pathology for therapeutic targeting
Bulletin of the National Research Centre (2024)
-
Effect of Nicotine on Pulmonary Pathogenic Bacteria
Current Microbiology (2024)