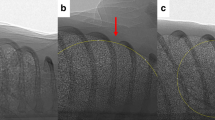
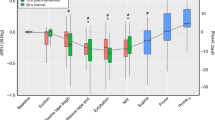
Abstract
Background
As very preterm infants have surfactant-deficient and highly incompliant lungs, slowing lung deflation during expiration might help preserve functional residual capacity(FRC) during lung aeration. In this study, we investigated the effect of expiratory resistance(Re) on lung aeration during positive pressure ventilation in preterm rabbits immediately after birth.
Methods
Preterm rabbit pups were delivered at 29 days gestation, mechanically ventilated from birth and simultaneously imaged to measure lung aeration using phase-contrast X-ray. Re was varied by altering the length (0, 60 or 1000 mm) of the expiratory circuit.
Results
Increasing Re led to a decrease in lung deflation rates and both peak expiratory flows and flow rates at mid-deflation. As a result, the rate of de-acceleration(slowing) in lung deflation when approaching FRC was markedly reduced with increasing resistance. During lung aeration, FRC was significantly different between resistance groups and was significantly higher over time in the high compared to the low resistance group. While FRC values tended to be higher with higher Re, they were not significantly different at end-ventilation (t = 7 min).
Conclusion
Increasing Re of the ventilation circuit during lung aeration in preterm rabbits immediately after birth decreased lung deflation rates and increased the accumulation of FRC over time.
Impact
-
The expiratory phase of the ventilatory cycle has been largely overlooked as an opportunity to improve ventilation in preterm infants after birth.
-
Increasing the expiratory resistance of the ventilator circuit during lung aeration in preterm rabbits immediately after birth markedly decreased lung deflation rates and increased FRC accumulation, compared to a low expiratory resistance.
-
This indicates that ventilation devices that reduce the “work of breathing” by reducing the expiratory resistance, may have the unintended effect of reducing FRC, particularly in extremely preterm infants that have surfactant deficient highly incompliant lungs.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Hooper, S. B., Siew, M. L., Kitchen, M. J. & te Pas, A. B. Establishing functional residual capacity in the non-breathing infant. Semin. Fetal Neonatal Med. 18, 336–343 (2013).
Hooper, S. B., Te Pas, A. B. & Kitchen, M. J. Respiratory transition in the newborn: a three-phase process. Arch. Dis. Child Fetal Neonatal Ed. 101, F266–F271 (2016).
te Pas, A. B. et al. Breathing Patterns in Preterm and Term Infants Immediately After Birth. Pediatr. Res. 65, 352 (2009).
Gaertner, V. D., Büchler, V. L., Waldmann, A., Bassler, D. & Rüegger, C. M. Deciphering Mechanisms of Respiratory Fetal-to-Neonatal Transition in Very Preterm Infants. Am. J. Respir. Crit. Care Med. 209, 738–747 (2024).
Kuypers, K. et al. The Effect of a Higher Bias Gas Flow on Imposed T-Piece Resistance and Breathing in Preterm Infants at Birth. Front. Pediatr. 10, 817010 (2022).
Tingay, D. G. et al. Imaging the Respiratory Transition at Birth: Unraveling the Complexities of the First Breaths of Life. Am. J. Respir. Crit. Care Med. 204, 82–91 (2021).
Davies, I. M. et al. Respiratory patterns in spontaneously breathing near-term lambs delivered by caesarean section under spinal anaesthesia. Front. Pediatr. 11, 1273136 (2023).
Davies, I. M. et al. Adverse respiratory patterns in near-term spontaneously breathing newborn lambs with elevated airway liquid volumes at birth. Front. Pediatr. 12, https://doi.org/10.3389/fped.2024.1336154 (2024).
Kuypers, K., Kashyap, A. J., Cramer, S. J. E., Hooper, S. B. & Te Pas, A. B. The effect of imposed resistance in neonatal resuscitators on pressure stability and peak flows: a bench test. Pediatr. Res. https://doi.org/10.1038/s41390-023-02715-x (2023).
Polglase, G. R. et al. Positive end-expiratory pressure differentially alters pulmonary hemodynamics and oxygenation in ventilated, very premature lambs. J. Appl Physiol. 99, 1453–1461 (2005).
Fuhrman, B. P., Everitt, J. & Lock, J. E. Cardiopulmonary effects of unilateral airway pressure changes in intact infant lambs. J. Appl. Physiol. 56, 1439–1448 (1984).
Australian Code for Responsible Conduct of Research (Commonwealth of Australia, National Health and Medical Research Council, Australian Research Council and Universities Australia, 2018.
Percie du Sert, N. et al. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. BMJ Open Sci. 4, e100115 (2020).
Kitchen, M. J. et al. A new design for high stability pressure-controlled ventilation for small animal lung imaging. J. Instrum. 5, T02002–T02002 (2010).
Kitchen, M. J. et al. Dynamic measures of regional lung air volume using phase contrast x-ray imaging. Phys. Med. Biol. 53, 6065–6077 (2008).
Leong, A. F., Paganin, D. M., Hooper, S. B., Siew, M. L. & Kitchen, M. J. Measurement of absolute regional lung air volumes from near-field x-ray speckles. Opt. Express 21, 27905–27923 (2013).
Ghasemi, M. & Dehpour, A. R. Ethical considerations in animal studies. J. Med. Ethics Hist. Med. 2, 12 (2009).
McGillick, E. V. et al. Increased end-expiratory pressures improve lung function in near-term newborn rabbits with elevated airway liquid volume at birth. J. Appl Physiol. 131, 997–1008 (2021).
Moomjian, A. S. et al. The effect of external expiratory resistance on lung volume and pulmonary function in the neonate. J. Pediatrics 96, 908–911 (1980).
Moomjian, A. S. et al. Use of external expiratory resistance in intubated neonates to increase lung volume. Arch. Dis. Child. 56, 869–873 (1981).
Siew, M. L. et al. Positive end-expiratory pressure enhances development of a functional residual capacity in preterm rabbits ventilated from birth. J. Appl Physiol. 106, 1487–1493 (2009).
te Pas, A. B. et al. Establishing Functional Residual Capacity at Birth: The Effect of Sustained Inflation and Positive End-Expiratory Pressure in a preterm rabbit model. Pediatr. Res. 65, 537–541 (2009).
Bach, K. P. et al. High bias gas flows increase lung injury in the ventilated preterm lamb. PLoS One 7, e47044 (2012).
Hooper, S. B. et al. Expired CO2 levels indicate degree of lung aeration at birth. PLoS One 8, e70895 (2013).
Funding
This research was supported by a National Health and Medical Research Council (NHMRC) Ideas Grant (2012443) and Program Grant (APP113902) as well as the Victorian Government’s Operational Infrastructure Support Program. IMD was supported by Australian Government Research Training Program Scholarships. SBH was supported by an NHMRC Principal Research Fellowship (APP1058537). K.L.A.M. Kuypers was the recipient of an unrestricted research grant from Fisher & Paykel Healthcare Limited.
Author information
Authors and Affiliations
Contributions
K.K.: Conceptualisation, Methodology, Formal analysis and interpretation of data, Writing – original draft, Writing – review & editing. J.D.: Conceptualisation, Methodology, Supervision, Interpretation of data, Writing – review & editing. K.C., M.W.: Investigation, Interpretation of data, Writing – review & editing. S.C., I.D.: Interpretation of data, Writing – review & editing. D.J.: formal analysis and interpretation of data, Writing – review & editing. M.K.: Investigation, formal analysis and interpretation of data, Writing – review & editing. A.tP.: Conceptualisation, Investigation, Interpretation of data, Writing – review & editing. S.H.: Conceptualisation, Methodology, Funding acquisition, Investigation, Supervision, Writing – original draft, Writing – review & editing.
Corresponding author
Ethics declarations
Competing interests
K.L.A.M. Kuypers is the recipient of an unrestricted research grant from Fisher & Paykel Healthcare Limited; they had no role in study design nor in the collection, analysis, and interpretation of data, writing of the report and decision to submit the paper for publication.
Ethics approval
All animal procedures were approved by the SPring-8 Animal Care and Monash University’s Animal Ethics Committees.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kuypers, K.L.A.M., Dekker, J., Crossley, K.J. et al. Slowing lung deflation by increasing the expiratory resistance enhances FRC in preterm rabbits. Pediatr Res 97, 723–728 (2025). https://doi.org/10.1038/s41390-024-03388-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-024-03388-w