Abstract
Background
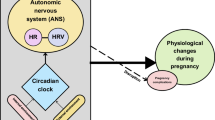
Autonomic nervous system (ANS) dysregulation is common during critical illness and is often measured using heart rate variability (HRV). It is unknown if other forms of ANS function, such as pupillary light reflex and thermoregulation, are altered in critically ill children. We aimed to determine whether automated pupillometry and delta (central-to-peripheral) skin temperatures were associated with HRV.
Methods
In this prospective observational pilot, inclusion criteria were admission to the pediatric intensive care unit (ICU) and mechanical ventilation. HRV was calculated using age-adjusted integer HRV (HRVi). Automated pupillometry and skin temperatures were recorded during the first 72 h of admission. The primary outcomes were: (1) correlation between HRVi and Neurological Pupil index (NPi), and (2) correlation between HRVi and delta skin temperature.
Results
Of 29 patients enrolled, 18 had pupillometer data and 20 had temperature data. There were significant, small correlations between left and right NPi values and HRVi (r = 0.13, r = 0.12; p < 0.001), and delta skin temperatures and HRVi (r = 0.15, p < 0.001), which persisted after adjusting for confounders.
Conclusions
Abnormal pupillary light response and decreased delta skin temperatures are associated with lower HRVi. If validated, pupillometry and skin temperature could be considered physiologic biomarkers of ANS dysregulation in critically ill children.
Impact
-
While heart rate variability has strong associations with outcomes in critically ill children, there are limited data on other bedside tools of autonomic function in critically ill children.
-
In this study, we found that pupillometry and skin temperature sensors correlate with heart rate variability in critically ill children.
-
These physiological biomarkers may have a role in early detection of autonomic nervous system dysregulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Badke, C. M., Marsillio, L. E., Weese-Mayer, D. E. & Sanchez-Pinto, L. N. Autonomic Nervous System Dysfunction in Pediatric Sepsis. Front. Pediatr. 6, 280 (2018).
Dunser, M. W. & Hasibeder, W. R. Sympathetic overstimulation during critical illness: adverse effects of adrenergic stress. J. Intensive Care Med. 24, 293–316 (2009).
Moorman, J. R. et al. Mortality reduction by heart rate characteristic monitoring in very low birth weight neonates: a randomized trial. J. Pediatr. 159, 900–906.e901 (2011).
Goldstein, B. et al. Decomplexification in critical illness and injury: relationship between heart rate variability, severity of illness, and outcome. Crit. Care Med. 26, 352–357 (1998).
Badke, C. M., Marsillio, L. E., Carroll, M. S., Weese-Mayer, D. E. & Sanchez-Pinto, L. N. Development of a Heart Rate Variability Risk Score to Predict Organ Dysfunction and Death in Critically Ill Children. Pediatr. Crit. Care Med. 22, e437–e447 (2021).
Olson, D. M. et al. Interrater Reliability of Pupillary Assessments. Neurocrit Care 24, 251–257 (2016).
Nyholm, B. et al. Superior reproducibility and repeatability in automated quantitative pupillometry compared to standard manual assessment, and quantitative pupillary response parameters present high reliability in critically ill cardiac patients. PLoS One 17, e0272303 (2022).
Nyholm, B. et al. Specific thresholds of quantitative pupillometry parameters predict unfavorable outcome in comatose survivors early after cardiac arrest. Resusc. Plus. 14, 100399 (2023).
Patwari, P. P. et al. Pupillometry in congenital central hypoventilation syndrome (CCHS): quantitative evidence of autonomic nervous system dysregulation. Pediatr. Res. 71, 280–285 (2012).
Yanovich, R., Ketko, I. & Charkoudian, N. Sex Differences in Human Thermoregulation: Relevance for 2020 and Beyond. Physiol. 35, 177–184 (2020).
Sessler, D. I. Thermoregulatory defense mechanisms. Crit. Care Med. 37, S203–S210 (2009).
Saiyed, R. et al. Congenital central hypoventilation syndrome (CCHS): Circadian temperature variation. Pediatr. Pulmonol. 51, 300–307 (2016).
Olson, D. M. et al. Establishing Normative Data for Pupillometer Assessment in Neuroscience Intensive Care: The “END-PANIC” Registry. J. Neurosci. Nurs. 49, 251–254 (2017).
Hasselberg, M. J., McMahon, J. & Parker, K. The validity, reliability, and utility of the iButton(R) for measurement of body temperature circadian rhythms in sleep/wake research. Sleep. Med. 14, 5–11 (2013).
Pollack, M. M. et al. The Pediatric Risk of Mortality Score: Update 2015. Pediatr. Crit. Care Med. 17, 2–9 (2016).
Muppidi, S. et al. Dynamic pupillometry as an autonomic testing tool. Clin. Auton. Res. 23, 297–303 (2013).
Larson, M. D. & Singh, V. Portable infrared pupillometry in critical care. Crit. Care 20, 161 (2016).
Privitera, C. M. et al. A differential of the left eye and right eye neurological pupil index is associated with discharge modified Rankin scores in neurologically injured patients. BMC Neurol. 22, 273 (2022).
Winston, M. et al. Pupillometry measures of autonomic nervous system regulation with advancing age in a healthy pediatric cohort. Clin. Auton. Res. 30, 43–51 (2020).
Khaytin, I. et al. Evolution of physiologic and autonomic phenotype in rapid-onset obesity with hypothalamic dysfunction, hypoventilation, and autonomic dysregulation over a decade from age at diagnosis. J. Clin. Sleep. Med. 18, 937–944 (2022).
Carroll, M. S., Ramirez, J. M. & Weese-Mayer, D. E. Diurnal variation in autonomic regulation among patients with genotyped Rett syndrome. J. Med. Genet. 57, 786–793 (2020).
Frank, S. M., El-Rahmany, H. K., Tran, K. M., Vu, B. & Raja, S. N. Comparison of lower extremity cutaneous temperature changes in patients receiving lumbar sympathetic ganglion blocks versus epidural anesthesia. J. Clin. Anesth. 12, 525–530 (2000).
Larson, M. D. & Behrends, M. Portable infrared pupillometry: a review. Anesth. Analg. 120, 1242–1253 (2015).
Acknowledgements
We would like to acknowledge the PICU nurses at Lurie Children’s Hospital for facilitating patient enrollment and participation in this study.
Funding
Dr. Badke received an internal grant from Stanley Manne Children’s Research Institute to support this work. The other authors have no relevant funding to report.
Author information
Authors and Affiliations
Contributions
Substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data: C.B., T.P., D.W.P., N.S.P. Drafting the article or revising it critically for important intellectual content: C.B., M.C., T.P., D.W.M., N.S.P. Final approval of the version to be published: C.B., M.C., T.P., D.W.M., N.S.P.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
Parents or legal guardians gave consent for their children to be enrolled in this study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Badke, C.M., Carroll, M.S., Pendergrast, T.R. et al. Assessing bedside measures of autonomic nervous system dysregulation in the PICU. Pediatr Res 98, 983–988 (2025). https://doi.org/10.1038/s41390-024-03778-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-024-03778-0