Abstract
Background
Epinephrine is currently the only vasopressor recommended for use during neonatal resuscitation. Epinephrine can be administered via intravenous, intraosseous, or endotracheal tube (ETT) route during cardiopulmonary resuscitation (CPR). Supraglottic airway (SGA) may be a novel route of epinephrine administration. This study aimed to compare the pharmacokinetics and pharmacodynamics of 0.1 mg/kg epinephrine administered via ETT, SGA top end, and SGA bottom end.
Design/methods
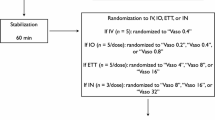
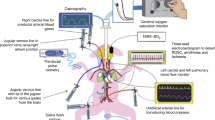
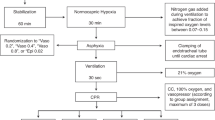
Newborn piglets (n = 5/group) were anesthetized, randomized to SGA or tracheostomy, then surgically instrumented. Piglets randomized to SGA underwent another round of randomization following stabilization to receive epinephrine at the top or bottom of the SGA. Heart rate (HR), arterial blood pressure, carotid blood flow, and cardiac function (e.g., stroke volume and ejection fraction) were continuously recorded throughout the experiment. Blood was collected prior to drug administration and throughout the observation period for pharmacodynamics and pharmacokinetic analysis.
Results
Significant changes in hemodynamic parameters of HR, carotid blood flow, and cardiac function were only observed following ETT administration of epinephrine, while pharmacokinetic parameters were not different between ETT, SGA top, or SGA bottom.
Conclusion
There were no differences in pharmacokinetic parameters between ETT, SGA top, or SGA bottom routes of epinephrine administration in neonatal piglets.
Impact
-
Endotracheal tube (ETT) epinephrine results in significant hemodynamic parameters changes, whereas supraglottic airway (SGA) epinephrine did not produce the same hemodynamic effects, despite similar pharmacokinetic profiles.
-
Systematic comparison of pharmacokinetics and pharmacodynamics of epinephrine via SGA versus ETT identifying potential limitations of SGA for epinephrine administration.
-
The study raises important questions about the effectiveness of SGA for epinephrine administration during neonatal resuscitation.
-
This research could influence future resuscitation guidelines and drive further studies to explore alternative dosing strategies or methods to improve the efficacy of SGA epinephrine.
-
Further experiments examining SGA epinephrine during neonatal cardiopulmonary resuscitation are warranted.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article. Data used to generate the results reported in this study will be made available following publication from the corresponding author upon reasonable request.
References
Wyckoff, M. H. et al. Neonatal life support: 2020 international consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations. Circulation 142, S185–S221 (2020).
Pollack, C. V. The laryngeal mask airway: a comprehensive review for the emergency physician. J. Emerg. Med. 20, 53–66 (2001).
Davies, P. R., Tighe, S. Q., Greenslade, G. L. & Evans, G. H. Laryngeal mask airway and tracheal tube insertion by unskilled personnel. Lancet 336, 977–979 (1990).
Abedini, N., Parish, M., Farzin, H., Pourfathi, H. & Akhsham, M. The determination of an appropriate time for placement of the classic laryngeal mask airway in patients undergoing general anesthesia. Anesth. Pain. Med. 8, e64427 (2018).
Wanous, A. A., Brown, R., Rudser, K. D. & Roberts, K. D. Comparison of laryngeal mask airway and endotracheal tube placement in neonates. J. Perinatol. 44, 239–243 (2024).
Barbosa, R. F., Simões e Silva, A. C. & Silva, Y. P. A randomized controlled trial of the laryngeal mask airway for surfactant administration in neonates. J. Pediatr. 93, 343–350 (2017).
Amini, E. et al. Surfactant administration in preterm neonates using laryngeal mask airway: a randomized clinical trial. Acta Med. Iran. 57, 348–354 (2019).
Roberts, K. D. et al. Laryngeal mask airway for surfactant administration in neonates: a randomized, controlled trial. J. Pediatr. 193, 40–46.e1 (2018).
Trevisanuto, D. et al. Laryngeal mask airway used as a delivery conduit for the administration of surfactant to preterm infants with respiratory distress syndrome. Neonatology 87, 217–220 (2005).
Trevisanuto, D. et al. Supreme laryngeal mask airway versus face mask during neonatal resuscitation: a randomized controlled trial. J. Pediatr. 167, 286–291.e1 (2015).
Pejovic, N. J. et al. Neonatal resuscitation using a laryngeal mask airway: a randomised trial in Uganda. Arch. Dis. Child. 103, 255–260 (2018).
Pejovic, N. J. et al. A randomized trial of laryngeal mask airway in neonatal resuscitation. N. Engl. J. Med. 383, 2138–2147 (2020).
Wyckoff, M. H. et al. International consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations: summary from the basic life support; advanced life support; pediatric life support; neonatal life support; education, implementation, and teams; and first aid task forces. Circulation 146, e483–e557 (2022).
Chen, K. T., Lin, H. J., Guo, H. R., Lin, M. T. & Lin, C. C. Feasibility study of epinephrine administration via laryngeal mask airway using a porcine model. Resuscitation 69, 503–507 (2006).
Liao, C. K. et al. Epinephrine administration via a laryngeal mask airway: what is the optimal dose? (2010). https://www.signavitae.com/articles/10.22514/SV52.112010.4
Percie du Sert, N. et al. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. BMC Vet. Res. 16, 242 (2020).
Schmölzer, G. M. et al. Cardiopulmonary resuscitation with chest compressions during sustained inflations: a new technique of neonatal resuscitation that improves recovery and survival in a neonatal porcine model. Circulation 128, 2495–2503 (2013).
Schmölzer, G. M. et al. 3:1 compression to ventilation ratio versus continuous chest compression with asynchronous ventilation in a porcine model of neonatal resuscitation. Resuscitation 85, 270–275 (2014).
Cheung, P. Y., Gill, R. S. & Bigam, D. L. A swine model of neonatal asphyxia. J. Vis. Exp. 56, 3166 (2011).
O’Reilly, M. et al. Intramuscular versus intravenous epinephrine administration in a pediatric porcine model of cardiopulmonary resuscitation. Resusc. Plus 20, 100769 (2024).
Wagner, M. et al. Effects of epinephrine on hemodynamic changes during cardiopulmonary resuscitation in a neonatal piglet model. Pediatr. Res. 83, 897–903 (2018).
Shen, W., Xu, X., Lee, T. F., Schmölzer, G. & Cheung, P. Y. The relationship between heart rate and left ventricular isovolumic relaxation during normoxia and hypoxia-asphyxia in newborn piglets. Front. Physiol. 10, 525 (2019).
Chen, K. T., Lin, H. J., Jeng, H. W., Lin, C. C. & Guo, H. R. The pharmacological effect of epinephrine administration via laryngeal mask airway in a porcine model of asphyxial cardiac arrest. Emerg. Med. J. 25, 722–724 (2008).
Polglase, G. R. et al. Endotracheal epinephrine at standard versus high dose for resuscitation of asystolic newborn lambs. Resuscitation 198, 110191 (2024).
Mani, S. et al. Laryngeal mask ventilation with chest compression during neonatal resuscitation: randomized, non-inferiority trial in lambs. Pediatr. Res. 92, 671–677 (2022).
Funding
We would like to thank our funding agencies: the study was supported by a project grant from the Canadian Institutes of Health Research. MR is a recipient of the Canadian Institutes of Health Research Canada Graduate Scholarships-Master’s program, Walter H Johns Graduate Fellowship, Alberta Graduate Excellence Scholarship, and Medical Sciences Graduate Program Scholarship.
Author information
Authors and Affiliations
Contributions
Conception and design: G.M.S., P.Y.C., M.O.R., M.R., and T.F.L. Collection and assembly of data: G.M.S., P.Y.C., M.O.R., M.R., and T.F.L. Analysis and interpretation of the data: G.M.S., P.Y.C., M.O.R., M.R., T.F.L. Drafting of the first draft: MR. Critical revision of the article for important intellectual content: G.M.S., P.Y.C., M.O.R., M.R., and T.F.L. Final approval of the article: G.M.S., P.Y.C., M.O.R., M.R., and T.F.L.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
Patient consent was not required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ramsie, M., Cheung, PY., Lee, TF. et al. Pharmacokinetics and pharmacodynamics of endotracheal versus supraglottic airway epinephrine in a healthy neonatal piglet model. Pediatr Res 98, 1539–1544 (2025). https://doi.org/10.1038/s41390-025-03960-y
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-025-03960-y
This article is cited by
-
Supraglottic airway use during neonatal resuscitation: better suited to aeration than medication?
Pediatric Research (2025)