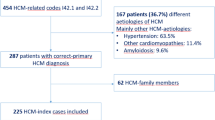
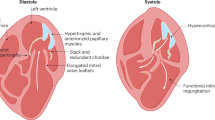
Abstract
Background
Hypertrophic cardiomyopathy (HCM) presents a wide range of clinical scenarios depending on the age of manifestation, with a less favorable prognosis in children. The genetic spectrum and clinical causes of HCM diagnosed before one year of age is rarely reported.
Methods
We analyzed the genetic causes and genotype-phenotype correlations in 68 children diagnosed with HCM during the first year of life. Genetic analysis was performed using targeted gene sequencing (39 HCM-related genes), followed by whole-exome sequencing for genotype-negative cases. The genetic data were correlated with clinical characteristics, disease progression, and prognosis.
Results
The overall genotype-positive rate was 81%, with an equal proportion of sarcomeric (29%) and RAS-related genetic cases (29%). Gestational diabetes in mothers was more frequently observed in children with variants in Z-disc-related genes. Overall, one year-survival rate from all causes was 91.2%, with the best survival outcomes associated with sarcomeric and Z-disk-related gene variants.
Conclusion
HCM manifesting in children before one year of age showed an approximately equal proportion of sarcomeric and RAS cascade-related cases. A more favorable prognosis was associated with sarcomeric mutations; whereas metabolic gene-related HCM cases were characterized by the highest one-and five-year mortality due to heart failure.
Impact
-
We analyzed the genetic causes and genotype-phenotype correlations in 68 children diagnosed with HCM during the first year of life.
-
Patients with sarcomeric mutations demonstrated a more favorable prognosis, whereas metabolic gene-related HCM cases were the highest one- and five-year mortality rates due to HF.
-
We identified several factors associated with unfavorable outcomes, including LV thickness, HF class, elevated troponin, increased NT-proBNP levels, and RV hypertrophy.
-
We proposed several new and previously unreported genes, such as ROBO4 and KMT2D, as potentially causative for infantile HCM. The true role of these genes in this disease requires confirmation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon reasonable request.
References
Nugent, A. W. et al. The epidemiology of childhood cardiomyopathy in Australia. N. Engl. J. Med. 348, 1639–1646 (2003).
Lipshultz, S. E. et al. The incidence of pediatric cardiomyopathy in two regions of the United States. N. Engl. J. Med. 348, 1647–1655 (2003).
Karim, S. et al. Re-evaluating the Incidence and Prevalence of Clinical Hypertrophic Cardiomyopathy: An Epidemiological Study of Olmsted County, Minnesota. Mayo Clin. Proc. 99, 362–374 (2024).
Amr, A. et al. Improving sudden cardiac death risk stratification in hypertrophic cardiomyopathy using established clinical variables and genetic information. Clin. Res. Cardiol. J. Ger. Card. Soc. 113, 728–736 (2024).
Norrish, G. et al. External validation of the HCM Risk-Kids model for predicting sudden cardiac death in childhood hypertrophic cardiomyopathy. Eur. J. Prevent. Cardiol. 29, 678–686 (2022).
Stegeman, R. et al. The etiology of cardiac hypertrophy in infants. Sci. Rep. 11, 10626 (2021).
Marston, N. A. et al. Clinical characteristics and outcomes in childhood-onset hypertrophic cardiomyopathy. Eur. Heart J. 42, 1988–1996 (2021).
Limongelli, G. et al. Prevalence and clinical significance of red flags in patients with hypertrophic cardiomyopathy. Int. J. Cardiol. 299, 186–191 (2020).
Norrish, G. et al. Clinical presentation and long-term outcomes of infantile hypertrophic cardiomyopathy: a European multicentre study. ESC Heart Fail. 8, 5057–5067 (2021).
Hong, Y. et al. Risk factors of sudden cardiac death in hypertrophic cardiomyopathy. Curr. Opin. Cardiol. 37, 15–21 (2022).
Norrish, G. et al. Clinical Features and Natural History of Preadolescent Nonsyndromic Hypertrophic Cardiomyopathy. J. Am. Coll. Cardiol. 79, 1986–1997 (2022).
Chan, W. et al. Clinical characteristics and survival of children with hypertrophic cardiomyopathy in China: A multicentre retrospective cohort study. EClinicalMedicine 49, 101466 (2022).
Maron, B. J. et al. Contemporary definitions and classification of the cardiomyopathies: an American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation 113, 1807–1816 (2006).
Roberts, AE. Noonan Syndrome (GeneReviews, 1993–2024).
Tariq, M. & Ware, S. M. Importance of genetic evaluation and testing in pediatric cardiomyopathy. World J. Cardiol. 6, 1156–1165 (2014).
Bai, R. et al. Yield of genetic screening in inherited cardiac channelopathies: how to prioritize access to genetic testing. Circ. Arrhythmia Electrophysiol. 2, 6–15 (2009).
van Spaendonck-Zwarts, K. Y. et al. Genetic analysis in 418 index patients with idiopathic dilated cardiomyopathy: overview of 10 years’ experience. Eur. J. Heart Fail. 15, 628–636 (2013).
Garcia, M. et al. Sarek: A portable workflow for whole-genome sequencing analysis of germline and somatic variants. F1000Research 9, 63 (2020).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 17, 405–424 (2015).
Marston, S. Obscurin variants and inherited cardiomyopathies. Biophys. Rev. 9, 239–243 (2017).
Ramos-Kuri, M. et al. Molecules linked to Ras signaling as therapeutic targets in cardiac pathologies. Biol. Res. 54, 23 (2021).
Nagalingam, R. S. et al. A cardiac-enriched microRNA, miR-378, blocks cardiac hypertrophy by targeting Ras signaling. J. Biol. Chem. 292, 5123 (2017).
Dhandapany, P. S. et al. Cyclosporine attenuates cardiomyocyte hypertrophy induced by RAF1 mutants in Noonan and LEOPARD syndromes. J. Mol. Cell. Cardiol. 51, 4–15 (2011).
Colella, P. Advances in Pompe Disease Treatment: From Enzyme Replacement to Gene Therapy. Mol. Diagnosis Ther. 28, 703–719 (2024).
Yadin, D. et al. Effect of pharmacological heart failure drugs and gene therapy on Danon’s cardiomyopathy. Biochem. Pharmacol. 215, 115735 (2023).
Kaski, J. P. et al. Cardiomyopathies in children and adolescents: aetiology, management, and outcomes in the European Society of Cardiology EURObservational Research Programme Cardiomyopathy and Myocarditis Registry. Eur. Heart J. 45, 1443–1454 (2024).
Wanert, C. et al. Genetic profile and genotype-phenotype correlations in childhood cardiomyopathy. Arch. Cardiovasc. Dis. 116, 309–315 (2023).
Ahamed, H. et al. Phenotypic expression, genotypic profiling and clinical outcomes of infantile hypertrophic cardiomyopathy: a retrospective study. Arch. Dis. Child. 109, 913–917 (2024).
Monda, E. et al. Hypertrophic Cardiomyopathy in Children: Pathophysiology, Diagnosis, and Treatment of Non-sarcomeric Causes. Front. Pediatrics 9, 632293 (2021).
Lesurf, R. et al. Whole genome sequencing delineates regulatory, copy number, and cryptic splice variants in early onset cardiomyopathy. NPJ Genom. Med. 7, 18 (2022).
Sedaghat-Hamedani, F. et al. Clinical outcomes associated with sarcomere mutations in hypertrophic cardiomyopathy: a meta-analysis on 7675 individuals. Clin. Res. Cardiol. 107, 30–41 (2018).
Velicki, L. et al. Genetic determinants of clinical phenotype in hypertrophic cardiomyopathy. BMC Cardiovasc. Disord. 20, 516 (2020).
Roberts, A. E. et al. Noonan syndrome. Lancet 381, 333–342 (2013).
Pierpont, M. E. & Digilio, M. C. Cardiovascular disease in Noonan syndrome. Curr. Opin. Pediatrics 30, 601–608 (2018).
Gazzin, A. et al. Defining the variant-phenotype correlation in patients affected by Noonan syndrome with the RAF1:c.770C>T p.(Ser257Leu) variant. Eur. J. Hum. Genet. 32, 8 (2024).
Thompson, D. et al. RAF1 variants causing biventricular hypertrophic cardiomyopathy in two preterm infants: further phenotypic delineation and review of literature. Clin. Dysmorphol. 26, 195–199 (2017).
Darouich, S. et al. A severe clinicopathologic phenotype of RAF1 Ser257Leu neomutation in a preterm infant without cardiac anomaly. Am. J. Med. Genet. Part A. 191, 630–633 (2023).
Ratola, A. et al. A Novel Noonan Syndrome RAF1 Mutation: Lethal Course in a Preterm Infant. Pediatr. Rep. 7, 5955 (2015).
Gazzin, A. et al. Exploring New Drug Repurposing Opportunities for MEK Inhibitors in RASopathies: A Comprehensive Review of Safety, Efficacy, and Future Perspectives of Trametinib and Selumetinib. Life 14, 731 (2024).
Kaltenecker, E. et al. Long-term outcomes of childhood onset Noonan compared to sarcomere hypertrophic cardiomyopathy. Cardiovasc. Diagnosis Ther. 9, S299–S309 (2019).
Bos, J. M. & Ackerman, M. J. Z-disc genes in hypertrophic cardiomyopathy: stretching the cardiomyopathies? J. Am. Coll. Cardiol. 55, 1136–1138 (2010).
Wadmore, K. et al. The Role of Z-disc Proteins in Myopathy and Cardiomyopathy. Int. J. Mol. Sci. 22, 3058 (2021).
Harper, A. R. et al. Common genetic variants and modifiable risk factors underpin hypertrophic cardiomyopathy susceptibility and expressivity. Nat. Genet. 53, 135–142 (2021).
Marian, A. J. & Braunwald, E. Hypertrophic Cardiomyopathy: Genetics, Pathogenesis, Clinical Manifestations, Diagnosis, and Therapy. Circ. Res. 121, 749–770 (2017).
Maron, B. J. et al. Double or compound sarcomere mutations in hypertrophic cardiomyopathy: a potential link to sudden death in the absence of conventional risk factors. Heart Rhythm. 9, 57–63 (2012).
Lopes, L. R. et al. Genetic complexity in hypertrophic cardiomyopathy revealed by high-throughput sequencing. J. Med. Genet. 50, 228–239 (2013).
Girolami, F. et al. Clinical features and outcome of hypertrophic cardiomyopathy associated with triple sarcomere protein gene mutations. J. Am. Coll. Cardiol. 55, 1444–1453 (2010).
Ingles, J. et al. Compound and double mutations in patients with hypertrophic cardiomyopathy: implications for genetic testing and counselling. J. Med. Genet. 42, e59 (2005).
Komurcu-Bayrak, E. et al. Identification of the pathogenic effects of missense variants causing PRKAG2 cardiomyopathy. Arch. Biochem. Biophys. 727, 109340 (2022).
Xu, Y. et al. A novel, de novo mutation in the PRKAG2 gene: infantile-onset phenotype and the signaling pathway involved. Am. J. Physiol. Heart Circ. Physiol. 313, H283–H292 (2017).
Kelly, B. P. et al. Severe hypertrophic cardiomyopathy in an infant with a novel PRKAG2 gene mutation: potential differences between infantile and adult onset presentation. Pediatr. Cardiol. 30, 1176–1179 (2009).
Akman, H. O. et al. Fatal infantile cardiac glycogenosis with phosphorylase kinase deficiency and a mutation in the gamma2-subunit of AMP-activated protein kinase. Pediatr. Res. 62, 499–504 (2007).
Burwinkel, B. et al. Fatal congenital heart glycogenosis caused by a recurrent activating R531Q mutation in the gamma 2-subunit of AMP-activated protein kinase (PRKAG2), not by phosphorylase kinase deficiency. Am. J. Hum. Genet. 76, 1034–1049 (2005).
Ahamed, H. et al. Phenotypic expression and clinical outcomes in a South Asian PRKAG2 cardiomyopathy cohort. Sci. Rep. 10, 20610 (2020).
Verdonschot, J. A. J. et al. A mutation update for the FLNC gene in myopathies and cardiomyopathies. Hum. Mutat. 41, 1091–1111 (2020).
Muravyev, A. et al. Rare clinical phenotype of filaminopathy presenting as restrictive cardiomyopathy and myopathy in childhood. Orphanet J. Rare Dis. 17, 358 (2022).
Krishnaswamy, S. M. et al. Investigation of mutation spectrum amongst patients with familial primary cardiomyopathy using targeted NGS in Indian population. J. Appl. Genet., 65, 809–822 (2024).
Stanczyk, P. J. et al. Association of cardiac myosin-binding protein-C with the ryanodine receptor channel - putative retrograde regulation? J. Cell Sci. 131, jcs210443 (2018).
Landstrom, A. P. & Ackerman, M. J. Beyond the cardiac myofilament: hypertrophic cardiomyopathy- associated mutations in genes that encode calcium-handling proteins. Curr. Mol. Med. 12, 507–518 (2012).
Alvarado, F. J. et al. Cardiac hypertrophy and arrhythmia in mice induced by a mutation in ryanodine receptor 2. JCI insight 5, e126544 (2019).
Yang, Q. et al. Determining the Likelihood of Disease Pathogenicity Among Incidentally Identified Genetic Variants in Rare Dilated Cardiomyopathy-Associated Genes. J. Am. Heart Assoc. 11, e025257 (2022).
Lippi, M. et al. Spectrum of Rare and Common Genetic Variants in Arrhythmogenic Cardiomyopathy Patients. Biomolecules 12, 1043 (2022).
Shen, C. et al. Genetic variants in Chinese patients with sporadic dilated cardiomyopathy: a cross-sectional study. Ann. Transl. Med. 10, 129 (2022).
Fernlund, E. et al. Hereditary Hypertrophic Cardiomyopathy in Children and Young Adults-The Value of Reevaluating and Expanding Gene Panel Analyses. Genes 11, 1472 (2020).
Carnevale, A. et al. Genomic study of dilated cardiomyopathy in a group of Mexican patients using site-directed next generation sequencing. Mol. Genet. Genom. Med. 8, e1504 (2020).
Neagoe, O. et al. A rare case of familial restrictive cardiomyopathy, with mutations in MYH7 and ABCC9 genes. Discoveries 7, e99 (2019).
Waldmüller, S. et al. Targeted 46-gene and clinical exome sequencing for mutations causing cardiomyopathies. Mol. Cell. Probes 29, 308–314 (2015).
van Bon, B. W. M. et al. Cantú syndrome is caused by mutations in ABCC9. Am. J. Hum. Genet. 90, 1094–1101 (2012).
Hathaway, J. et al. Diagnostic yield of genetic testing in a heterogeneous cohort of 1376 HCM patients. BMC Cardiovasc. Disord. 21, 126 (2021).
Zhao, J. & Mommersteeg, M. T. M. Slit-Robo signalling in heart development. Cardiovasc. Res. 114, 794–804 (2018).
Mommersteeg, M. T. M. et al. Slit-roundabout signaling regulates the development of the cardiac systemic venous return and pericardium. Circ. Res. 112, 465–475 (2013).
Rafipay, A. et al. Knockdown of slit signaling during limb development leads to a reduction in humerus length. Dev. Dyn. Publ. Am. Assoc. Anatomists 250, 1340–1357 (2021).
Münch, J. et al. Biallelic pathogenic variants in roundabout guidance receptor 1 associate with syndromic congenital anomalies of the kidney and urinary tract. Kidney Int. 101, 1039–1053 (2022).
Jiang, L. et al. Role of Slit/Robo Signaling pathway in Bone Metabolism. Int. J. Biol. Sci. 18, 1303–1312 (2022).
Mommersteeg, M. T. M. et al. Disrupted Slit-Robo signalling results in membranous ventricular septum defects and bicuspid aortic valves. Cardiovasc. Res. 106, 55–66 (2015).
Kruszka, P. et al. Loss of function in ROBO1 is associated with tetralogy of Fallot and septal defects. J. Med. Genet. 54, 825–829 (2017).
Gould, R. A. et al. ROBO4 variants predispose individuals to bicuspid aortic valve and thoracic aortic aneurysm. Nat. Genet. 51, 42–50 (2019).
Jaouadi, H. et al. Expanding the phenome and variome of the ROBO-SLIT pathway in congenital heart defects: toward improving the genetic testing yield of CHD. J. Transl. Med. 21, 160 (2023).
Musfee, F. I. et al. Rare deleterious variants of NOTCH1, GATA4, SMAD6, and ROBO4 are enriched in BAV with early onset complications but not in BAV with heritable thoracic aortic disease. Mol. Genet. Genom. Med. 8, e1406 (2020).
Liu, X. et al. Stromal Cell-SLIT3/Cardiomyocyte-ROBO1 Axis Regulates Pressure Overload-Induced Cardiac Hypertrophy. Circ. Res. 134, 913–930 (2024).
Neumann D, Karnik R. Kabuki Syndrome (StatPearls Publishing, 2024).
Lin, Y. et al. Overlap phenotypes of the left ventricular noncompaction and hypertrophic cardiomyopathy with complex arrhythmias and heart failure induced by the novel truncated DSC2 mutation. Orphanet J. Rare Dis. 16, 496 (2021).
Wu, G. et al. Deleterious Rare Desmosomal Variants Contribute to Hypertrophic Cardiomyopathy and Are Associated With Distinctive Clinical Features. Can. J. Cardiol. 38, 41–48 (2022).
Castellana, S et al. Sudden death in mild hypertrophic cardiomyopathy with compound DSG2/DSC2/MYH6 mutations: Revisiting phenotype after genetic assessment in a master runner athlete. J. Electrocardiol. 53, 95–99 (2019).
Acknowledgements
This work was financially supported by the Ministry of Science and Higher Education of the Russian Federation (Agreement No. 075-15-2022-301).
Author information
Authors and Affiliations
Contributions
F.S.G., M.O.V. made substantial contributions to the conception and design of the study, acquisition, analysis, and interpretation of the data and drafting of the initial manuscript. K.A.A., V.E.S. made substantial contributions to the study design and data analysis, and interpretation of the data and reviewed and edited the manuscript. V.T.L., K.O.A., K.A.A., F.Yu.V., S.P.S., Z.S.V., P.T.M. participated in data analyses and interpretation and critically revised and approved the final draft. F.S.G. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The study was approved by the Ethics Committee of the Almazov National Medical Research Centre (Protocol №01-23 dated 23.01.23) according to the Helsinki Declaration. Written consent was retrieved from the participating children’s guardians.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fetisova, S., Melnik, O., Vasichkina, E. et al. The clinical and genetic spectrum of pediatric hypertrophic cardiomyopathy manifesting before one year of age. Pediatr Res 98, 1301–1312 (2025). https://doi.org/10.1038/s41390-025-03989-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-025-03989-z
This article is cited by
-
Infantile Hypertrophic cardiomyopathy: steps towards an evidence-based approach to genetic testing
Pediatric Research (2025)