Abstract
Background
This study examined the effects of growth hormone (GH) treatment on growth and metabolic parameters, gross motor function, and musculoskeletal complications in children with cerebral palsy.
Methods
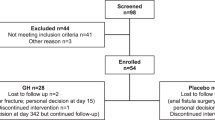
This retrospective single-center study included children who received GH treatment for over 1 year. Data on growth-related variables, metabolic indicators, and disease-specific factors were collected. Statistical analyses examined associations between GH treatment duration and outcomes, with pre- and post-treatment comparisons.
Results
Twenty-two patients were enrolled in this study. GH treatment significantly improved height and weight Z-scores, along with insulin-like growth factor-1 Z-scores, regardless of GH deficiency or ambulatory status. Metabolic changes significantly included an increased bone age-to-chronological age ratio, decreased aspartate aminotransferase levels, and elevated uric acid levels. The gross motor function did not decline, and there was no significant worsening of hip subluxation or scoliosis.
Conclusion
GH treatment led to significant improvements in growth and metabolic-related parameters without adverse effects on gross motor function or musculoskeletal complications. These findings suggest that GH treatment may be a safe and effective treatment option for children with cerebral palsy, providing potential benefits without increasing the risk of orthopedic complications.
Impact
-
This study is the first to assess the long-term effects of GH treatment in children with CP.
-
GH treatment improved height, weight, and insulin-like growth factor-1 Z-scores.
-
Gross motor function did not decline during treatment, and no significant worsening of hip subluxation or scoliosis detected.
-
There is a lack of longitudinal studies on the long-term effects of growth hormone administration in children with CP.
-
It is anticipated that this study will lay the groundwork for large-scale multicenter prospective research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Kuperminc, M. N. & Stevenson, R. D. Growth and nutrition disorders in children with cerebral palsy. Dev. Disabil. Res. Rev. 14, 137–146 (2008).
Fung, E. B. et al. Feeding dysfunction is associated with poor growth and health status in children with cerebral palsy. J. Am. Diet. Assoc. 102, 361–373 (2002).
Devesa, J., Casteleiro, N., Rodicio, C., López, N. & Reimunde, P. Growth hormone deficiency and cerebral palsy. Ther. Clin. Risk Manag 6, 413–418 (2010).
Devesa, J., Lima, L. & Tresguerres, J. A. Neuroendocrine control of growth hormone secretion in humans. Trends Endocrinol. Metab. 3, 175–183 (1992).
Ali, O., Shim, M., Fowler, E., Cohen, P. & Oppenheim, W. Spinal bone mineral density, IGF-1 and IGFBP-3 in children with cerebral palsy. Horm. Res 68, 316–320 (2007).
Shim, M. L., Moshang, T. Jr., Oppenheim, W. L. & Cohen, P. Is treatment with growth hormone effective in children with cerebral palsy?. Dev. Med. Child Neurol. 46, 569–571 (2004).
Reimunde, P. et al. Effects of recombinant growth hormone replacement and physical rehabilitation in recovery of gross motor function in children with cerebral palsy. Ther. Clin. Risk Manag 6, 585–592 (2010).
Bache, C. E., Selber, P. & Graham, H. K. (ii) The management of spastic diplegia. Curr. Orthop. 17, 88–104 (2003).
TANNER, J. M. Growth and endocrinology of the adolescent. Endocr. Genet. Dis. Childhood Adolesc. 26, 117–130 (1975).
Kim, J. H. et al. The 2017 Korean National Growth Charts for children and adolescents: development, improvement, and prospects. Korean J. Pediatr. 61, 135–149 (2018).
Hyun, S. E. et al. Reference values for serum levels of insulin-like growth factor-I and insulin-like growth factor binding protein-3 in Korean children and adolescents. Clin. Biochem. 45, 16–21 (2012).
Surveillance of cerebral palsy in Europe: a collaboration of cerebral palsy surveys and registers Surveillance of Cerebral Palsy in Europe (SCPE). Dev. Med. Child Neurol. 42, 816–824 (2000).
Himmelmann, K. et al. MRI classification system (MRICS) for children with cerebral palsy: development, reliability, and recommendations. Dev. Med. Child Neurol. 59, 57–64 (2017).
Cobb, J. Outline for the study of scoliosis. Instruct. Course Lect. 5, 261–275 (1948).
Miller, F., Slomczykowski, M., Cope, R. & Lipton, G. E. Computer modeling of the pathomechanics of spastic hip dislocation in children. J. Pediatr. Orthop. 19, 486–492 (1999).
Horng, M. H., Kuok, C. P., Fu, M. J., Lin, C. J. & Sun, Y. N. Cobb angle measurement of spine from X-ray images using convolutional neural network. Comput Math. Methods Med 2019, 6357171 (2019).
Wu, S., Yang, W. & De Luca, F. Insulin-like growth factor-independent effects of growth hormone on growth plate Chondrogenesis and longitudinal bone growth. Endocrinology 156, 2541–2551 (2015).
Mosier, H. D. Jr. & Jansons, R. A. Growth hormone during catch-up growth and failure of catch-up growth in rats. Endocrinology 98, 214–219 (1976).
Hermanussen, M. et al. Differential catch-up in body weight and bone growth after short-term starvation in rats. Growth Regul. 6, 230–237 (1996).
Pando, R., Shtaif, B., Phillip, M. & Gat-Yablonski, G. A serum component mediates food restriction-induced growth attenuation. Endocrinology 155, 932–940 (2014).
Eliakim, A. & Nemet, D. Exercise provocation test for growth hormone secretion: methodologic considerations. Pediatr. Exerc Sci. 20, 370–378 (2008).
Gat-Yablonski, G. & De Luca, F. Effect of Nutrition on Statural Growth. Horm. Res. Paediatr. 88, 46–62 (2017).
Al Shaikh, A. et al. Effect of growth hormone treatment on children with idiopathic short stature (ISS), idiopathic growth hormone deficiency (IGHD), small for gestational age (SGA) and Turner syndrome (TS) in a tertiary care center. Acta Biomed. 91, 29–40 (2020).
Xue, J., Liang, S., Ma, J. & Xiao, Y. Effect of growth hormone therapy on liver enzyme and other cardiometabolic risk factors in boys with obesity and nonalcoholic fatty liver disease. BMC Endocr. Disord. 22, 49 (2022).
Doycheva, I., Erickson, D. & Watt, K. D. Growth hormone deficiency and NAFLD: An overlooked and underrecognized link. Hepatol. Commun. 6, 2227–2237 (2022).
Takahashi, Y. Nonalcoholic fatty liver disease and adult growth hormone deficiency: An under-recognized association?. Best. Pr. Res. Clin. Endocrinol. Metab. 37, 101816 (2023).
Yoon, J. S. et al. Association between uric acid and height during growth hormone therapy in children with idiopathic short stature. Front. Endocrinol. 13, 1025005 (2022).
Ekbote, V. H. et al. Relationship of insulin-like growth factor 1 and bone parameters in 7-15 years old apparently, healthy Indian children. Indian J. Endocrinol. Metab. 19, 770–774 (2015).
Alvim, R. O. et al. Influence of muscle mass on the serum uric acid levels in children and adolescents. Nutr. Metab. Cardiovasc Dis. 30, 300–305 (2020).
Ji, Y. T., Li, L. L., Cai, S. Z. & Shi, X. Y. Body composition in preschool children with short stature: a case-control study. BMC Pediatr. 22, 98 (2022).
Boekhoff, S., Bogusz, A., Sterkenburg, A. S., Eveslage, M. & Müller, H. L. Long-term effects of growth hormone replacement therapy in childhood-onset Craniopharyngioma: Results of the German Craniopharyngioma Registry (HIT-Endo). Eur. J. Endocrinol. 179, 331–341 (2018).
Samson-Fang, L. et al. Relationship of nutritional status to health and societal participation in children with cerebral palsy. J. Pediatr. 141, 637–643 (2002).
Ali, O. et al. Growth hormone therapy improves bone mineral density in children with cerebral palsy: a preliminary pilot study. J. Clin. Endocrinol. Metab. 92, 932–937 (2007).
Terjesen, T. & Horn, J. Risk factors for hip displacement in cerebral palsy: A population-based study of 121 nonambulatory children. J. Child Orthop. 16, 306–312 (2022).
Aroojis, A., Mantri, N. & Johari, A. N. Hip displacement in cerebral palsy: the role of surveillance. Indian J. Orthop. 55, 5–19 (2021).
Park, M. et al. The association between idiopathic scoliosis and growth hormone treatment in short children. Ann. Pediatr. Endocrinol. Metab. 27, 207–213 (2022).
Willoughby, K. L. et al. Epidemiology of scoliosis in cerebral palsy: A population-based study at skeletal maturity. J. Paediatr. Child Health 58, 295–301 (2022).
Haidar, R. K., Nasrallah, M. P., Der-Boghossian, A. H. & Ghanem, I. B. Orthopedic complications related to growth hormone therapy in a pediatric population. J. Pediatr. Orthop. B. 20, 57–61 (2011).
Shore, B. J. et al. Proximal Femoral Varus Derotation Osteotomy in children with cerebral palsy: the effect of age, gross motor function classification system level, and surgeon volume on surgical success. J. Bone Jt. Surg. Am. 97, 2024–2031 (2015).
Acknowledgements
We would like to thank Editage (www.editage.co.kr) for English language editing.
Funding
This study is supported by a 2024 research grant from the Research Institute of Rehabilitation Medicine, Yonsei University College of Medicine. The funding source had no role in the study design; in the collection, analysis, and interpretation of the data; in the writing of the report; and in the decision to submit the paper for publication. The authors declare no interests that might be perceived as posing a conflict or bias.
Author information
Authors and Affiliations
Contributions
H.J.T: Conceptualization, Methodology, Visualization, Writing – original draft preparation, Software. K.S: Data curation, Methodology, Writing – original draft preparation. S.J.H: Investigation, Validation, Writing – reviewing & editing. D.W.R: Investigation, Validation, Writing – reviewing & editing. All authors reviewed and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent statement
The institutional review board of Yonsei University College of Medicine waived the requirement for informed consent, given the practical impossibility of obtaining informed consent from participants during the research process and the minimal risk posed to participants.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hong, J., Kim, S., Suh, J. et al. Growth hormone treatment in cerebral palsy: long-term impact on growth, outcomes, and complications. Pediatr Res (2025). https://doi.org/10.1038/s41390-025-04440-z
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-025-04440-z