Abstract
Background
Bronchopulmonary dysplasia (BPD) and significant hemodynamic patent ductus arteriosus (hsPDA) are both common and important clinical issues in extremely preterm infants. The potential impact on prognosis when these conditions coexist is a major focus of clinical concern. This study examined the relationship between hsPDA and adverse outcomes in BPD infants.
Methods
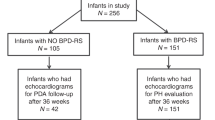
A retrospective analysis of 781 preterm infants (<32 weeks) from three hospitals (2018–2023). Based on echocardiographic assessment, infants were categorized as the non-PDA group, the non-hsPDA group, or the hsPDA group. Further subgroups were formed according to treatment and ductus arteriosus size (<1.5 mm, 1.5–3 mm, ≥3 mm). The effects of hsPDA on short-term outcomes in infants with BPD were assessed using logistic regression and linear regression.
Results
The study included 781 infants (548 non-BPD, 233 BPD). The hsPDA subgroup had lower gestational age, higher birth asphyxia rates, and required more invasive respiratory support. In BPD infants, hsPDA was linked to longer respiratory support, higher pneumonia and feeding intolerance risks, prolonged oxygen therapy, and PH. Infants with hsPDA had longer hospital stays and oxygen therapy. Intervention therapy in infants with hsPDA was associated with prolonged oxygen therapy duration, reduced feeding intolerance, and increased risk of pulmonary hypertension(PH). Meanwhile, ductus arteriosus diameter >3 mm was linked to elevated risks of feeding intolerance, pulmonary hypertension, and extrauterine growth restriction. After adjusting for gestational age and birth weight, results from multivariate logistic regression and multiple linear regression analyses indicated that hsPDA was independently associated with increased risk of neonatal PH (aOR = 7.502, 95% CI: 4.046–13.911, P < 0.001) and significantly prolonged invasive respiratory support duration (β = 6.530 days, 95% CI: 1.691–11.368, P = 0.008).
Conclusion
In BPD infants, hsPDA is associated with the occurrence of PH and longer duration of invasive respiratory support.
Impact
-
This study highlights the significant correlation between hemodynamically significant patent ductus arteriosus (hsPDA) and adverse outcomes in infants diagnosed with bronchopulmonary dysplasia (BPD), providing valuable clinical evidence for better management strategies.
-
This study adds to the literature by showing that in very preterm infants with BPD, the presence of hsPDA was independently correlated with both an increased risk of pulmonary hypertension and a longer duration of invasive respiratory support.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
The datasets generated during or analyzed during the current study are not publicly available due to patient privacy, but are available from the corresponding author on reasonable request.
References
Cao, Y. et al.Chinese Neonatal Network Assessment of neonatal intensive care unit practices, morbidity, and mortality among very preterm infants in China. JAMA Netw. Open. 4, e2118904 (2021).
Katz, T. A. et al. Comparison of new bronchopulmonary dysplasia definitions on long-term outcomes in preterm infants. J. Pediatr. 253, 86–93.e4 (2023).
Lian, W. et al. Chinese Multicenter EUGR Collaborative Group. Risk factors of extrauterine growth restriction in very preterm infants with bronchopulmonary dysplasia: a multi-center study in China. BMC Pediatr. 22, 363 (2022).
Qian, A. M. et al.Chinese Neonatal Network [Treatment of patent ductus arteriosus in very preterm infants in China]. Chin. J. Contemp. Pediatr. 61, 896–901 (2023).
Al-Turkait, A., Szatkowski, L., Choonara, I. & Ojha, S. Management of patent ductus arteriosus in very preterm infants in England and Wales: a retrospective cohort study. BMJ Paediatr. Open 6, e001424 (2022).
Willis, K. A. & Weems, M. F. Hemodynamically significant patent ductus arteriosus and the development of bronchopulmonary dysplasia. Congenit. Heart Dis. 14, 27–32 (2019).
Higgins, D. R. et al. Bronchopulmonary dysplasia: executive summary of a workshop. J. Pediatr. 197, 300–308 (2018).
Souvik, M. et al. Management of the patent ductus arteriosus in preterm infants. Pediatr. Child Health 27, 63–63 (2022).
Dudley, S., Sen, S., Hanson, A., El Khuffash, A. & Levy, P. T. The role of furosemide and fluid management for a hemodynamically significant patent ductus arteriosus in premature infants. J. Perinatol. 42, 1703–1707 (2022).
Mitra, S., de Boode, W. P., Weisz, D. E. & Shah, P. S. Interventions for patent ductus arteriosus (PDA) in preterm infants: an overview of Cochrane Systematic Reviews. Cochrane Database Syst. Rev. 4, CD013588 (2023).
Mcnamara, P. J. & Sehgal, A. Towards rational management of the patent ductus arteriosus: the need for disease staging. Arch. Dis. Child. Fetal Neonatal Ed. 92, F424–F427 (2007).
Ambalavanan, N. et al. Patent ductus arteriosus in preterm infants. Pediatrics 155, e2025071425 (2025).
The Subspecialty Group of Neonatology, the Society of Pediatrics, Chinese Medical Association Expert consensus on diagnosis and management of neonatal bacteria sepsis (2024). Chin. J. Pediatr. 62, 931–940 (2024).
Perlman, J. M. Periventricular- intraventricular hemorrhage in the premature infant—a historical perspective. Semin. Perinatol. 46, 151591 (2022).
Evidence-Based Medicine Group Clinical guidelines for the diagnosis and treatment of neonatal necrotizing enterocolitis (2020). Chin. J. Contemp. Pediatr. 23, 1–11 (2021).
Fundus Disease Group of Ophthalmological Society of Chinese Medical Association, Fundus Disease Group of Ophthalmologist Branch of Chinese Medical Doctor Association Expert consensus on the classification and treatment of retinopathy of prematurity in China (2023 edition). Chin. J. Ocul. Fundus Dis. 39, 720–727 (2023).
Kim, Y. J., Shin, S. H., Park, H. W., Kim, E. K. & Kim, H. S. Risk factors of early pulmonary hypertension and its clinical outcomes in preterm infants: a systematic review and meta-analysis. Sci. Rep. 12, 14186 (2022).
Chesi, E. et al. Patent ductus arteriosus (also non-hemodynamically significant) correlates with poor outcomes in very low birth weight infants. A multicenter cohort study. PLoS ONE 19, e0306769 (2024).
Martini, S. et al. Impact of patent ductus arteriosus on non-invasive assessments of lung fluids in very preterm infants during the transitional period. Eur. J. Pediatr. 182, 4247–4251 (2023).
Diez, S. et al. Clinical characteristics of necrotizing enterocolitis in preterm patients with and without persistent ductus arteriosus and in patients with congenital heart disease. Front. Pediatr. 8, 257 (2020).
Giesinger, R. E., Hobson, A. A., Bischoff, A. R., Klein, J. M. & McNamara, P. J. Impact of early screening echocardiography and targeted PDA treatment on neonatal outcomes in “22-23” week and “24-26” infants. Semin. Perinatol. 47, 151721 (2023).
Sankar, M. N., Bhombal, S. & Benitz, W. E. PDA: to treat or not to treat. Congenit. Heart Dis. 14, 46–51 (2019).
Clyman, R. I. et al.PDA-TOLERATE (PDA: TO LEave it alone or Respond And Treat Early) Trial Investigators PDA-TOLERATE Trial: an exploratory randomized controlled trial of treatment of moderate-to-large patent ductus arteriosus at 1 week of age. J. Pediatr. 205, 41–48.e6 (2019).
Mitra, S., Scrivens, A., von Kursell, A. M. & Disher, T. Early treatment versus expectant management of hemodynamically significant patent ductus arteriosus for preterm infants. Cochrane Database Syst. Rev. 12, CD013278 (2020).
Mitra, S., Bischoff, A. R., Sathanandam, S., Lakshminrusimha, S. & McNamara, P. J. Procedural closure of the patent ductus arteriosus in preterm infants: a clinical practice guideline. J. Perinatol. 44, 1402–1408 (2024).
Huang, Z. et al. Diameter of ductus arteriosus on postnatal Day 7 is associated with late pulmonary hypertension in extremely preterm infants. Pediatr. Pulmonol. 58, 3523–3529 (2023).
Wu, Z. Q. et al. Risk factors for the failure of ibuprofen treatment in preterm infants with hemodynamically significant patent ductus arteriosus. Chin. J. Contemp. Pediatr. 26, 343–349 (2024).
Nawaytou, H., Hills, N. K. & Clyman, R. I. Patent ductus arteriosus and the risk of bronchopulmonary dysplasia-associated pulmonary hypertension. Pediatr. Res 94, 547–554 (2023).
Bjorkman, K. R. et al. Patent ductus arteriosus and lung magnetic resonance imaging phenotype in moderate and severe bronchopulmonary dysplasia-pulmonary hypertension. Am. J. Respir. Crit. Care Med. 210, 318–328 (2024).
Kim, C. et al. Long-term impact of late pulmonary hypertension requiring medication in extremely preterm infants with severe bronchopulmonary dysplasia. Sci. Rep. 14, 8705 (2024).
Acknowledgements
The authors acknowledge the financial support from the special project of the Action Plan of “Science and Technology Rejuvenating Mongolia” of Shanghai Jiao Tong University in 2023 (2023XYJG0001-01-09) and the 2023 hospital-level Clinical Research Training Project of Shanghai Children’s Hospital (2023YLY02). The authors acknowledge Feng Tienan, statistician at the Clinical Research Institute of Shanghai Jiao Tong University School of Medicine, Qu Tiantian, project manager, and Chen Zhaoxia and Huang Xuequn, database developers, for their valuable contributions to this research. The authors also thank all participants for their efforts in performing this study.
Funding
This study was financially supported by the Special Project of the Action Plan of “Science and Technology Rejuvenating Mongolia” of Shanghai Jiao Tong University in 2023 (grant number 2023XYJG0001-01-09) and the 2023 Hospital-Level Clinical Research Training Project of Shanghai Children’s Hospital (grant number 2023YLY02).
Author information
Authors and Affiliations
Contributions
Manuscript writing, data collection, data organization, statistical analysis: Qianhan Ouyang. Research supervision: Fei Bei. Data collection, data organization: Chongbing Yan, Bowen Weng, Yuanyang Zhang, You You. Research supervision, manuscript revision: Hongping Xia. Research supervision, manuscript revision, funding support: Cheng Cai.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study was approved by the Medical Ethics Committee of Shanghai Children’s Hospital (Approval number: 2024RY069-E01) and certifies that the study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki. The study was retrospective, did not involve a diagnostic intervention for children, and informed consent was waived.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ouyang, Q., Bei, F., Yan, C. et al. Hemodynamically significant PDA impacts adverse outcomes in infants with BPD: a multicenter study. Pediatr Res (2026). https://doi.org/10.1038/s41390-026-04772-4
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-026-04772-4