Abstract
Background
MMA and CBS deficiency are rare autosomal recessive metabolic disorders caused by defects in cobalamin metabolism and cystathionine-beta-synthase activity, respectively. Advanced molecular genetic techniques, have become essential for diagnosing these conditions. This study aimed to analyze the genetic variations in three MMA and four CBS deficiency cases using WES and correlate the findings with clinical, biochemical, and treatment outcomes.
Methods
Clinical evaluation, biochemical testing, neuroimaging, and WES were performed. WES data were analyzed using the reference genome GRCh37, and variants were classified according to ACMG guidelines. Treatment outcomes were monitored for three months post-intervention.
Results
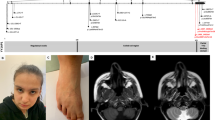
In MMA cases, a homozygous pathogenic variant (c.394 C > T, p.Arg132Ter) in MMACHC was identified in all three patients. Biochemical abnormalities included methylmalonic aciduria, elevated homocysteine, and megaloblastic anemia. Hydroxycobalamin therapy improved behavioral, cognitive, and dermatological symptoms, though residual neuropathy persisted in one case. In CBS deficiency, pathogenic variants in CBS (c.992 C > T, p.Ala331Val; c.862 G > A, p.Ala288Thr; c.700 G > A, p.Asp234Asn) were identified. Clinical features included developmental delayed milestones, lens dislocation, and vascular complications. Treatment outcomes varied based on early diagnosis and compliance.
Conclusion
This study highlights the importance of newborn screening program with WES in diagnosing and managing MMA and CBS deficiency, facilitating early intervention, improving clinical outcomes, and supporting precision medicine approaches.

Impact
-
Early newborn screening and diagnosis are critical for effective treatment and improved patient outcomes.
-
Novel clinical and pathogenic variants will expand the current understanding of these disorders.
-
WES enhances the diagnostic precision for MMA and CBS deficiency, facilitating timely intervention and superior clinical management.
-
Accurate and early detection of treatable IEMs through NBS can significantly reduce disease burden and healthcare costs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Driesen, K. & Witters, P. Understanding inborn errors of metabolism through metabolomics. Metabolites 12, 398 (2022).
Agana, M., Frueh, J., Kamboj, M., Patel, D. R. & Kanungo, S. Common metabolic disorder (inborn errors of metabolism) concerns in primary care practice. Ann. Transl. Med. 6, 469 (2018).
Vankwani, S., Wasim, M., Mirza, M. R. & Awan, F. R. Closing the gap: An urgent need for newborn screening of organic acid disorders in developing countries. J. Pak. Med. Assoc. 74, 1136–1143 (2024).
Wasim, M. et al. Aminoacidopathies: prevalence, etiology, screening, and treatment options. Biochem. Genet. 56, 7–21 (2018).
Huemer, M. et al. Newborn screening for homocystinurias and methylation disorders: systematic review and proposed guidelines. J. Inherit. Metab. Dis. 38, 1007–1019 (2015).
Morris, A. A. et al. Guidelines for the diagnosis and management of cystathionine beta-synthase deficiency. J. Inherit. Metab. Dis. 40, 49–74 (2017).
Therrell, B. L. et al. Current Status of Newborn Bloodspot Screening Worldwide 2024: A Comprehensive Review of Recent Activities (2020-2023). Int. J. Neonatal Screen. 10, 38 (2024).
Zhou, X., Cui, Y. & Han, J. Methylmalonic acidemia: Current status and research priorities. Intractable. Rare. Dis. Res. 7, 73–78 (2018).
Sloan, J. L. et al. The vitamin B12 processing enzyme, mmachc, is essential for zebrafish survival, growth and retinal morphology. Hum. Mol. Genet. 29, 2109–2123 (2020).
Beyzaei, Z., Moravej, H., Imanieh, M. H., Inaloo, S. & Geramizadeh, B. Clinical spectrum and genetic variation of six patients with methylmalonic aciduria (MMA); a report from Iran. Bmc. Pediatr. 24, 795 (2024).
Morrison, T. et al. Homocystinuria patient and caregiver survey: experiences of diagnosis and patient satisfaction. Orphanet. J. Rare. Dis. 16, 124 (2021).
Wasim, M., Khan, H. N., Ayesha, H. & Awan, F. R. Initiating newborn screening for metabolic disorders in Pakistan: A qualitative study of the early challenges and opportunities. Rare 1, 100011 (2023).
Wasim, M., H. N. Khan, H. Ayesha, and F. R. Awan. Need and Challenges in Establishing Newborn Screening Programs for Inherited Metabolic Disorders in Developing Countries. Adv. Biol (Weinh). e2200318 (2023).
Held, P. K., Singh, E. & Schwoerer, J. S. cott Screening for Methylmalonic and Propionic Acidemia: Clinical Outcomes and Follow-Up Recommendations. Int. J. Neonatal Screen. 8, 13 (2022).
Schnabel, E. et al. Combined Newborn Screening Allows Comprehensive Identification also of Attenuated Phenotypes for Methylmalonic Acidurias and Homocystinuria. Nutrients 15, 3355 (2023).
He, R. et al. Variable phenotypes and outcomes associated with the MMACHC c.609G>A homologous mutation: long term follow-up in a large cohort of cases. Orphanet. J. Rare. Dis. 15, 200 (2020).
Liu, X. et al. Prominent renal complications associated with MMACHC pathogenic variant c.80A > G in Chinese children with cobalamin C deficiency. Front. Pediatr. 10, 1057594 (2022).
Huemer, M. et al. Guidelines for diagnosis and management of the cobalamin-related remethylation disorders cblC, cblD, cblE, cblF, cblG, cblJ and MTHFR deficiency. J. Inherit. Metab. Dis. 40, 21–48 (2017).
Chen, T. et al. Methylmalonic acidemia: Neurodevelopment and neuroimaging. Front. Neurosci. 17, 1110942 (2023).
Sun, M. & Dai, Y. Late-onset cobalamin C deficiency type in adult with cognitive and behavioral disturbances and significant cortical atrophy and cerebellar damage in the MRI: a case report. Front. Neurol. 14, 1308289 (2023).
Carrillo-Carrasco, N., Chandler, R. J. & Venditti, C. P. Combined methylmalonic acidemia and homocystinuria, cblC type. I. Clinical presentations, diagnosis and management. J. Inherit. Metab. Dis. 35, 91–102 (2012).
Khalil, A., Khan, H. N., Wasim, M., Ayesha, H. & Awan, F. R. Development of low-cost in-house tetra-ARMS-PCR assay for the screening of five CBS mutations found in Pakistani homocystinuria patients. Nucleosides Nucleotides Nucleic Acids 43, 517–529 (2024).
Wasim, M. et al. Identification of three novel pathogenic mutations in cystathionine beta-synthase gene of Pakistani intellectually disabled patients. J. Pediatr. Endocrinol. Metab. 35, 325–332 (2021).
Voskoboeva, E., Semyachkina, A., Yablonskaya, M. & Nikolaeva, E. Homocystinuria due to cystathionine beta-synthase (CBS) deficiency in Russia: Molecular and clinical characterization. Mol. Genet. Metab. Rep. 14, 47–54 (2018).
Kaur, R. et al. Seven novel genetic variants in a North Indian cohort with classical homocystinuria. Sci. Rep. 10, 17299 (2020).
Kluijtmans, L. A. et al. Molecular genetic analysis in mild hyperhomocysteinemia: a common mutation in the methylenetetrahydrofolate reductase gene is a genetic risk factor for cardiovascular disease. Am. J. Hum. Genet. 58, 35–41 (1996).
Yap, S., Rushe, H., Howard, P. M. & Naughten, E. R. The intellectual abilities of early-treated individuals with pyridoxine-nonresponsive homocystinuria due to cystathionine β-synthase deficiency. J. Inherit. Metab. Dis. 24, 437–447 (2001).
Vankwani, S. et al. Label free quantitative proteomic profiling of serum samples of intellectually disabled young patients revealed dysregulation of complement coagulation and cholesterol cascade systems. Metab. Brain. Dis. 39, 855–869 (2024).
Wasim, M. et al. High-performance liquid chromatography-based assay optimization for the detection of plasma amino acids for applications in metabolic disorders in developing countries. Sep. Sci. n/a, 2300119 (2023).
Modi, B. P. et al. Adult GAMT deficiency: A literature review and report of two siblings. Mol. Genet. Metab. Rep. 27, 100761 (2021).
Wasim, M. et al. High levels of blood glutamic acid and ornithine in children with intellectual disability. Int. J. Dev. Disabil. 68, 609–614 (2020).
Wasim, M., Khan, H. N., Ayesha, H. & Awan, F. R. Biochemical screening of intellectually disabled and healthy children in Punjab, Pakistan: differences in liver function test and lipid profiles. Int. J. Dev. Disabil. 66, 190–195 (2020).
Wasim, M. et al. Biochemical screening of intellectually disabled patients: a stepping stone to initiate a newborn screening program in Pakistan. Front Neurol. 10, 762 (2019).
Khan, H. N., Wasim, M., Ayesha, H. & Awan, F. R. Molecular genetic diagnosis of Wilson disease by ARMS-PCR in a Pakistani family. Mol. Biol. Rep. 45, 2585–2591 (2018).
Acknowledgements
We express gratitude to the 3biollion company for generously providing sequencing services for the patients at no cost.
Funding
This study supported by a postdoctoral start-up fund from the Maternal and Children’s Health Research Institute, Shunde Women and Children’s Hospital, Guangdong Medical University, China (2024BSHQD002).
Author information
Authors and Affiliations
Contributions
“The study conception and design were a collaborative effort involving all authors. IJ, HNK, YW, GM, and MW were responsible for supervision, material preparation, data collection, and analysis. The initial data and draft of the manuscript was written by MW, IJ, AI, NM, and TS. HNK, YW, GM, and MW provides genetic analyses and provided feedback on earlier versions / final version of the manuscript. The final manuscript was thoroughly reviewed and approved by all authors, including IJ, HNK, and MW.”
Corresponding authors
Ethics declarations
Competing interests
Nothing to declare. The research was conducted in the absence of any financial and commercial relationship.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the institute “Institutional Ethical Review Committee of Children Hospital & Institute of Child Health, Faisalabad”.
Consent to participate
Informed written consent was obtained for all the studied participants from their parents/guardian/head of special education centers.
Consent for publication
Consent has also been taken from the participants to publish this data for the welfare of humankind.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wasim, M., Javed, I., Iqbal, A. et al. Unveiling clinical and genetic landscapes of MMA and CBS: insights from whole exome sequencing in a tertiary care setting. Pediatr Res (2026). https://doi.org/10.1038/s41390-026-04834-7
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-026-04834-7