Abstract
Background
Recent studies have shown that from the 23rd week of gestation onward, the fetus becomes progressively more hypoxic, with oxygenation levels rising again after 33–34 weeks. The biological significance of this biphasic oxygenation pattern has remained unclear.
Methods
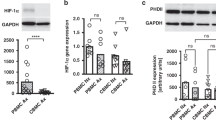
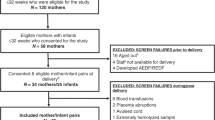
Umbilical cord blood samples from 100 preterm and 100 full-term neonates were analyzed for blood gas parameters and for HIF1A, ADRB3 (β3-adrenoceptor), and VEGFA gene expression.
Results
A progressive increase in mRNA expression of all three genes was observed with advancing gestational age, followed by a decline during the final weeks of pregnancy. This gene expression trend was inversely correlated with fetal oxygenation status.
Conclusion
This study demonstrates that β3-adrenoceptor expression progressively increases with gestational age, supporting the concept that this receptor plays a key role in fetal development and well-being. These findings strengthen the evidence from animal models showing that pharmacological activation of β3-adrenoceptors can reproduce, even after birth, some of the beneficial effects normally provided by the intrauterine environment. Collectively, this work supports the conceptual framework for developing a “pharmacological artificial placenta” aimed at mimicking intrauterine conditions to promote physiological neonatal adaptation.
Impact
-
Recent studies have shown that from the 23rd week of gestation onward, the fetus becomes progressively more hypoxic, with oxygenation levels rising again after 33–34 weeks. However, the biological significance of this biphasic oxygenation pattern had remained unclear.
-
This study demonstrates for the first time that fetal oxygen fluctuations are accompanied by a synchronous and coordinated increase in the mRNA expression of HIF1A, ADRB3 (β3-adrenoceptor), and VEGFA genes.
-
These findings support a mechanistic link between intrauterine hypoxia, β3-adrenergic signaling, and fetal maturation.
-
This work contributes to ongoing research suggesting that pharmacological β3-adrenoceptor activation may help recreate intrauterine-like conditions, potentially promoting physiological fetal development in adverse environments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Carter, A. M. Placental gas exchange and the oxygen supply to the fetus. Compr. Physiol. 5, 1381–1403 (2015).
Semenza, G. L. Hypoxia-inducible factor 1: control of oxygen homeostasis in health and disease. Pediatr. Res. 49, 614–617 (2001).
Tomita, S. et al. Defective brain development in mice lacking the Hif-1alpha gene in neural cells. Mol. Cell Biol. 23, 6739–6749 (2003).
Ateghang, B., Wartenberg, M., Gassmann, M. & Sauer, H. Regulation of cardiotrophin-1 expression in mouse embryonic stem cells by HIF-1alpha and intracellular reactive oxygen species. J. Cell Sci. 119, 1043–1052 (2006).
Weinberger, B., Laskin, D. L., Heck, D. E. & Laskin, J. D. Oxygen toxicity in premature infants. Toxicol. Appl. Pharmacol. 181, 60–67 (2002).
Filippi, L. et al. Fetal oxygenation from the 23rd to the 36th week of gestation evaluated through the umbilical cord blood gas analysis. Int. J. Mol. Sci. 24, 12487 (2023).
Filippi, L. et al. Fetal oxygenation in the last weeks of pregnancy evaluated through the umbilical cord blood gas analysis. Front. Pediatr. 11, 1140021 (2023).
Filippi, L. et al. β3-adrenoceptor agonism to mimic the biological effects of intrauterine hypoxia: taking great strides toward a pharmacological artificial placenta. Med. Res. Rev. 45, 842–866 (2025).
Filippi, L. et al. Decoupling oxygen tension from retinal vascularization as a new perspective for management of retinopathy of prematurity. New opportunities from β-adrenoceptors. Front. Pharmacol. 13, 835771 (2022).
Filippi, L. et al. Infantile hemangiomas, retinopathy of prematurity and cancer: a common pathogenetic role of the β-adrenergic system. Med. Res. Rev. 35, 619–652 (2015).
Xiang, S. et al. Insights into the mechanisms of angiogenesis in infantile hemangioma. Biomed. Pharmacother. 178, 117181 (2024).
Amato, R. et al. HIF-1-dependent induction of β3 adrenoceptor: evidence from the mouse retina. Cells 11, 1271 (2022).
Rouget, C. et al. Beta3-adrenoceptor is the predominant beta-adrenoceptor subtype in human myometrium and its expression is up-regulated in pregnancy. J. Clin. Endocrinol. Metab. 90, 1644–1650 (2005).
Cammalleri, M., Amato, R., Dal Monte, M., Filippi, L. & Bagnoli, P. The β3 adrenoceptor in proliferative retinopathies: “Cinderella” steps out of its family shadow. Pharmacol. Res. 190, 106713 (2023).
Filippi, L., Pini, A., Cammalleri, M., Bagnoli, P. & Dal Monte, M. β3-Adrenoceptor, a novel player in the round-trip from neonatal diseases to cancer: Suggestive clues from embryo. Med. Res. Rev. 42, 1179–1201 (2022).
Calvani, M. et al. 3-adrenoceptor as a potential immuno-suppressor agent in melanoma. Br. J. Pharmacol. 176, 2509–2524 (2019).
Calvani, M. et al. β3-Adrenoceptors as putative regulator of immune tolerance in cancer and pregnancy. Front. Immunol. 11, 2098 (2020).
Calvani, M. et al. β3-Adrenoreceptor blockade reduces hypoxic myeloid leukemic cells survival and chemoresistance. Int. J. Mol. Sci. 21, 4210 (2020).
Calvani, M. et al. β3-Adrenoreceptors control mitochondrial dormancy in melanoma and embryonic stem cells. Oxid. Med. Cell Longev. 2018, 6816508 (2018).
Dal Monte, M. et al. Functional involvement of β3-adrenergic receptors in melanoma growth and vascularization. J. Mol. Med. 91, 1407–1419 (2013).
Dal Monte, M., Filippi, L. & Bagnoli, P. Beta3-adrenergic receptors modulate vascular endothelial growth factor release in response to hypoxia through the nitric oxide pathway in mouse retinal explants. Naunyn Schmiedebergs Arch. Pharmacol. 386, 269–278 (2013).
Scaramuzzo, R. T. et al. 3-adREnoceptor analysis in CORD blood of neonates (β3 RECORD): study protocol of a pilot clinical investigation. Life 14, 776 (2024).
Ayres-de-Campos, D., Arulkumaran, S. & FIGO Intrapartum Fetal Monitoring Expert Consensus Panel FIGO consensus guidelines on intrapartum fetal monitoring: Physiology of fetal oxygenation and the main goals of intrapartum fetal monitoring. Int. J. Gynaecol. Obstet. 131, 5–8 (2015).
Executive summary: Neonatal encephalopathy and neurologic outcome, second edition Report of the American College of Obstetricians and Gynecologists’ Task Force on Neonatal Encephalopathy. Obstet. Gynecol. 123, 896–901 (2014).
Procino, G. et al. β3 adrenergic receptor in the kidney may be a new player in sympathetic regulation of renal function. Kidney Int. 90, 555–567 (2016).
Lassus, P. et al. Vascular endothelial growth factor and angiogenin levels during fetal development and in maternal diabetes. Biol. Neonate. 84, 287–292 (2003).
Galazios, G. et al. Umbilical cord serum vascular endothelial growth factor (VEGF) levels in normal pregnancies and in pregnancies complicated by preterm delivery or pre-eclampsia. Int. J. Gynaecol. Obstet. 85, 6–11 (2004).
Caniggia, I. et al. Hypoxia-inducible factor-1 mediates the biological effects of oxygen on human trophoblast differentiation through TGFbeta(3). J. Clin. Investig. 105, 577–587 (2000).
Ohls, R. K. Erythropoietin and hypoxia inducible factor-1 expression in the mid-trimester human fetus. Acta Paediatr. Suppl. 91, 27–30 (2002).
Ginouvès, A., Ilc, K., Macías, N., Pouysségur, J. & Berra, E. PHDs overactivation during chronic hypoxia “desensitizes” HIFalpha and protects cells from necrosis. Proc. Natl. Acad. Sci. USA 105, 4745–4750 (2008).
Berra, E., Roux, D., Richard, D. E. & Pouysségur, J. Hypoxia-inducible factor-1 alpha (HIF-1 alpha) escapes O(2)-driven proteasomal degradation irrespective of its subcellular localization: nucleus or cytoplasm. EMBO Rep. 2, 615–620 (2001).
Jewell, U. R. et al. Induction of HIF-1alpha in response to hypoxia is instantaneous. FASEB J. 15, 1312–1314 (2001).
Dal Monte, M., Evans, B. A., Arioglu-Inan, E. & Michel, M. C. Upregulation of β3-adrenoceptors-a general marker of and protective mechanism against hypoxia? Naunyn Schmiedebergs. Arch. Pharmacol. 393, 141–146 (2020).
Taylor, C. T. & Scholz, C. C. The effect of HIF on metabolism and immunity. Nat. Rev. Nephrol. 18, 573–587 (2022).
Simon, M. C. & Keith, B. The role of oxygen availability in embryonic development and stem cell function. Nat. Rev. Mol. Cell Biol. 9, 285–296 (2008).
Krock, B. L., Skuli, N. & Simon, M. C. Hypoxia-induced angiogenesis: good and evil. Genes Cancer 2, 1117–1133 (2011).
McGettrick, A. F. & O’Neill, L. A. J. The role of HIF in immunity and inflammation. Cell Metab. 32, 524–536 (2020).
Shweiki, D., Itin, A., Soffer, D. & Keshet, E. Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 359, 843–845 (1992).
Emorine, L. J. et al. Molecular characterization of the human beta 3-adrenergic receptor. Science 245, 1118–1121 (1989).
Michel, L. Y. M., Farah, C. & Balligand, J. L. The Beta3 adrenergic receptor in healthy and pathological cardiovascular tissues. Cells 9, 2584 (2020).
Cheng, H. J. et al. Upregulation of functional beta(3)-adrenergic receptor in the failing canine myocardium. Circ. Res. 89, 599–606 (2001).
Dessy, C. et al. Endothelial beta3-adrenoceptors mediate vasorelaxation of human coronary microarteries through nitric oxide and endothelium-dependent hyperpolarization. Circulation 110, 948–954 (2004).
Bruno, G. et al. β3-adrenoreceptor blockade reduces tumor growth and increases neuronal differentiation in neuroblastoma via SK2/S1P2 modulation. Oncogene 39, 368–384 (2020).
Calvani, M. et al. β3-Adrenoreceptor blockade induces stem cells differentiation in melanoma microenvironment. Int. J. Mol. Sci. 21, 1420 (2020).
Ma, I. T. et al. VEGF mRNA and protein concentrations in the developing human eye. Pediatr. Res. 77, 500–505 (2015).
Miao, G. et al. Relationship between the autoantibody and expression of β3-adrenoceptor in lung and heart. PLoS ONE 8, e68747 (2013).
Ågren, J., Segar, J. L., Söderström, F. & Bell, E. F. Fluid management considerations in extremely preterm infants born at 22-24 weeks of gestation. Semin. Perinatol. 46, 151541 (2022).
Stritzke, A., Thomas, S., Amin, H., Fusch, C. & Lodha, A. Renal consequences of preterm birth. Mol. Cell. Pediatr. 4, 2 (2017).
Filippi, L., Pini, A., Bagnoli, P., Dal Monte, M. & Cammalleri, M. Emerging opportunities in ROP therapy: from inhibiting pathological vessel growth to promoting physiological vascularization. Front. Med. 12, 1716449 (2025).
Filippi, L. et al. β3 adrenoceptor agonism prevents hyperoxia-induced colonic alterations. Biomolecules 13, 1755 (2023).
Nardini, P. et al. Protective effects of Beta-3 adrenoceptor agonism on mucosal integrity in hyperoxia-induced ileal alterations. Antioxidants 13, 863 (2024).
Nardini, P. et al. Beta-3 adrenoceptor agonism protects the enteric nervous tissue against hyperoxia-induced damage. Cells 14, 475 (2025).
Pini, A. et al. β3-adrenoceptor agonism exerts lung protection in a rat model of bronchopulmonary dysplasia. Br. J. Pharmacol. https://doi.org/10.1111/bph.70333 (2026).
Acknowledgements
We sincerely thank Jean-Luc Baroni and Cristina Ranzato for their exceptional support throughout this study.
Funding
Specifically, we would like to indicate that these analyses were supported by funds from the Italian Ministry of Universities and Research (Progetto di Ricerca di Rilevante Interesse Nazionale – PRIN), project entitled “OBERON – Oxygen dependence of the functional role of Beta3 adRenoceptors: nOvel iNsights from animal models of human neonatal diseases” (grant number 2022FYBMEX).
Author information
Authors and Affiliations
Contributions
R.T.S.: Interpretation of data, revision of the manuscript, and final approval. Lucr.F.: Acquisition of biological samples and interpretation of data, revision, and final approval. M.Cal.: Analysis, interpretation, and description of genetic results, revision of manuscript, and final approval. B.T.: Analysis, interpretation, and description of genetic results, revision of manuscript, and final approval. S.C.: Analysis, interpretation, and description of genetic results, revision of manuscript and final approval. M.D.R.: Analysis, interpretation, and description of genetic results, revision of manuscript and final approval. R.M.: statistical analysis, revision of manuscript and final approval. L.D.M.: Analysis, interpretation of results. M.Cam.: Interpretation of results, revision of manuscript. P.B.: Interpretation of results, revision of manuscript. M.D.M.: Interpretation of results, revision of manuscript. A.P.: Analysis of biological samples and interpretation of data, review of manuscript. Luca F.: Conception and design, acquisition of clinical and biological data, analysis and interpretation of data and results, writing primary draft of manuscript, revision, and final approval. Luca F. wrote the first draft of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki and its subsequent amendments. The research protocol was approved by the Pediatric Ethics Committee for Clinical Research of the Tuscany Region, Italy (approval number 318/2021).
Consent statement
Consent was obtained from the parents according to the research protocol approved by the Pediatric Ethics Committee for Clinical Research of the Tuscany Region, Italy (approval number 318/2021).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Scaramuzzo, R.T., Filippini, L., Calvani, M. et al. HIF1A, ADRB3, and VEGFA gene expression in human cord blood across gestation: insights toward a pharmacological artificial placenta. Pediatr Res (2026). https://doi.org/10.1038/s41390-026-04879-8
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-026-04879-8