Abstract
Study design
Randomised, controlled, crossover study.
Objectives
Paired corticospinal–motoneuronal stimulation (PCMS) involves repeatedly pairing stimuli to corticospinal neurones and motoneurones to induce changes in corticospinal transmission. Here, we examined whether PCMS could enhance maximal voluntary elbow flexion in people with cervical spinal cord injury.
Setting
Neuroscience Research Australia, Sydney, Australia.
Methods
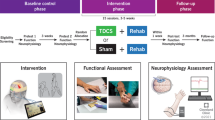
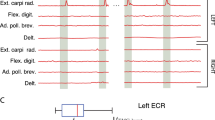
PCMS comprised 100 pairs of transcranial magnetic and electrical peripheral nerve stimulation (0.1 Hz), timed so corticospinal potentials arrived at corticospinal–motoneuronal synapses 1.5 ms before antidromic motoneuronal potentials. On two separate days, sets of five maximal elbow flexions were performed by 11 individuals with weak elbow flexors post C4 or C5 spinal cord injury before and after PCMS or control (100 peripheral nerve stimuli) conditioning. During contractions, supramaximal biceps brachii stimulation elicited superimposed twitches, which were expressed as a proportion of resting twitches to give maximal voluntary activation. Maximal torque and electromyographic activity were also assessed.
Results
Baseline median (range) maximal torque was 11 Nm (6–41 Nm) and voluntary activation was 92% (62–99%). Linear mixed modelling revealed no significant differences between PCMS and control protocols after conditioning (maximal torque: p = 0.87, superimposed twitch: p = 0.87, resting twitch: p = 0.44, voluntary activation: p = 0.36, biceps EMG: p = 0.25, brachioradialis EMG: 0.67).
Conclusions
Possible explanations for the lack of effect include a potential ceiling effect for voluntary activation, or that PCMS may be less effective for elbow flexors than distal muscles. Despite results, previous studies suggest that PCMS is worthy of further investigation.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Oudega M, Perez MA. Corticospinal reorganization after spinal cord injury. J Physiol. 2012;590:3647–63.
de Noordhout AM, Rapisarda G, Bogacz D, Gérard P, De Pasqua V, Pennisi G, et al. Corticomotoneuronal synaptic connections in normal man: an electrophysiological study. Brain. 1999;122:1327–40.
Palmer E, Ashby P. Corticospinal projections to upper limb motoneurones in humans. J Physiol. 1992;448:397–412.
Rathelot J-A, Strick PL. Subdivisions of primary motor cortex based on cortico-motoneuronal cells. Proc Natl Acad Sci. 2009;106:918–23.
Feldman Daniel E. The spike-timing dependence of plasticity. Neuron. 2012;75:556–71.
Bunday KL, Perez MA. Motor recovery after spinal cord injury enhanced by strengthening corticospinal synaptic transmission. Curr Biol. 2012;22:2355–61.
Dongés SC, D’Amico JM, Butler JE, Taylor JL. The involvement of N-methyl-D-aspartate receptors in plasticity induced by paired corticospinal–motoneuronal stimulation in humans. J Neurophysiol. 2018;119:652–61.
Fitzpatrick SC, Luu BL, Butler JE, Taylor JL. More conditioning stimuli enhance synaptic plasticity in the human spinal cord. Clin Neurophysiol. 2016;127:724–31.
Taylor JL, Martin PG. Voluntary motor output is altered by spike-timing-dependent changes in the human corticospinal pathway. J Neurosci. 2009;29:11708–16.
Urbin MA, Ozdemir RA, Tazoe T, Perez MA. Spike-timing-dependent plasticity in lower-limb motoneurons after human spinal cord injury. J Neurophysiol. 2017;118:2171–80.
D’Amico JM, Donges SC, Taylor JL. Paired corticospinal–motoneuronal stimulation increases maximal voluntary activation of human adductor pollicis. J Neurophysiol. 2017;119:369–76.
Shield A, Zhou S. Assessing voluntary muscle activation with the twitch interpolation technique. Sports Med. 2004;34:253–67.
Bellemare F, Bigland-Ritchie B. Assessment of human diaphragm strength and activation using phrenic nerve stimulation. Respir Physiol. 1984;58:263–77.
Merton PA. Voluntary strength and fatigue. J Physiol. 1954;123:553–64.
Allen GM, Gandevia SC, McKenzie DK. Reliability of measurements of muscle strength and voluntary activation using twitch interpolation. Muscle Nerve. 1995;18:593–600.
Behm DG, Whittle J, Button D, Power K. Intermuscle differences in activation. Muscle Nerve. 2002;25:236–43.
Gandevia SC, Allen GM, Butler JE, Taylor JL. Supraspinal factors in human muscle fatigue: evidence for suboptimal output from the motor cortex. J Physiol. 1996;490:529–36.
McKenzie DK, Bigland-Ritchie B, Gorman RB, Gandevia SC. Central and peripheral fatigue of human diaphragm and limb muscles assessed by twitch interpolation. J Physiol. 1992;454:643–56.
Jayaraman A, Gregory CM, Bowden M, Stevens JE, Shah P, Behrman AL, et al. Lower extremity skeletal muscle function in persons with incomplete spinal cord injury. Spinal Cord. 2006;44:680.
Kim HE, Thompson CK, Hornby TG. Muscle activation varies with contraction mode in human spinal cord injury. Muscle Nerve. 2015;51:235–45.
Thomas CK, Tucker ME, Bigland-Ritchie B. Voluntary muscle weakness and co-activation after chronic cervical spinal cord injury. J Neurotrauma. 1998;15:149–61.
Needham-Shropshire MB, Klose KJ, Tucker M, Thomas C. Manual muscle test score and force comparisons after cervical spinal cord injury. J Spinal Cord Med. 1997;20:324–30.
Cremoux S, Amarantini D, Tallet J, Dal Maso F, Berton E. Increased antagonist muscle activity in cervical SCI patients suggests altered reciprocal inhibition during elbow contractions. Clin Neurophysiol. 2016;127:629–34.
Lin K-H, Chen Y-C, Luh J-J, Wang C-H, Chang Y-J. H-reflex, muscle voluntary activation level, and fatigue index of flexor carpi radialis in individuals with incomplete cervical cord injury. Neurorehabil Neural Repair. 2012;26:68–75.
Bunday KL, Oudega M, Perez MA. Aberrant crossed corticospinal facilitation in muscles distant from a spinal cord injury. PLOS ONE. 2013;8:e76747.
Kendall FP, McCreary EK, Provance PG, Rodgers MM, Romani WA. Muscles: Testing and Function, with Posture and Pain. 5th edn. Philadelphia: Lippincott Williams and Wilkins; 2005.
Faul F, Erdfelder E, Lang A-G, Buchner A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods. 2007;39:175–91.
Mukaka MM. A guide to appropriate use of correlation coefficient in medical research. Malawi Med J. 2012;24:69–71.
Herbert RD, Gandevia SC. Muscle activation in unilateral and bilateral efforts assessed by motor nerve and cortical stimulation. J Appl Physiol. 1996;80:1351–6.
Allen GM, Gandevia AS, Middleton J. Quantitative assessments of elbow flexor muscle performance using twitch interpolation in post-polio patients: no evidence for deterioration. Brain. 1997;120:663–72.
Allen GM, Gandevia SC, Neering IR, Hickie I, Jones R, Middleton J. Muscle performance, voluntary activation and perceived effort in normal subjects and patients with prior poliomyelitis. Brain. 1994;117:661–70.
Kennedy DS, McNeil CJ, Gandevia SC, Taylor JL. Fatigue-related firing of distal muscle nociceptors reduces voluntary activation of proximal muscles of the same limb. J Appl Physiol. 2014;116:385–94.
Grumbles RM, Thomas CK. Motoneuron death after human spinal cord injury. J Neurotrauma. 2017;34:581–90.
Ridding MC, Ziemann U. Determinants of the induction of cortical plasticity by non‐invasive brain stimulation in healthy subjects. J Physiol. 2010;588:2291–304.
Di Lazzaro V, Oliviero A, Pilato F, Mazzone P, Insola A, Ranieri F, et al. Corticospinal volleys evoked by transcranial stimulation of the brain in conscious humans. Neurol Res. 2003;25:143–50.
Biering-Sørensen B, Kristensen IB, Kjær M, Biering‐Sørensen F. Muscle after spinal cord injury. Muscle Nerve. 2009;40:499–519.
Bowden JL, Taylor JL, McNulty PA. Voluntary activation is reduced in both the more- and less-affected upper limbs after unilateral stroke. Front Neurol. 2014;5:239.
Lampropoulou SI, Nowicky AV, Marston L. Magnetic versus electrical stimulation in the interpolation twitch technique of elbow flexors. J Sports Sci Med. 2012;11:709–18.
Thomas CK. Contractile properties of human thenar muscles paralyzed by spinal cord injury. Muscle Nerve. 1997;20:788–99.
Acknowledgements
We acknowledge Dr James Nuzzo, Zoë Djajadikarta, Harrison Finn and Dr Euan McCaughey for their assistance during experiments, and acknowledge participants for their contributions to the study.
Funding
This study was supported by a Program Grant (1055084) from the National Health and Medical Research Council of Australia (NHMRC).
Author information
Authors and Affiliations
Contributions
SCD and JLT conceived the work, and SCD, CLBR, JEB and JLT designed the work. SCD and CLBR acquired data and SCD, CLBR, JEB and JLT played an important role in interpreting the results. SCD drafted the manuscript and SCD, CLBR, JEB and JLT revised the manuscript. SCD, CLBR, JEB and JLT approved the final version and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Statement of ethics
We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research. Procedures were approved by the Human Research Ethics Committee of the University of New South Wales (approval number HC12556). The study was conducted according to the Declaration of Helsinki but was not registered in a publicly accessible database prior to participant recruitment.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Dongés, S.C., Boswell-Ruys, C.L., Butler, J.E. et al. The effect of paired corticospinal–motoneuronal stimulation on maximal voluntary elbow flexion in cervical spinal cord injury: an experimental study. Spinal Cord 57, 796–804 (2019). https://doi.org/10.1038/s41393-019-0291-3
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41393-019-0291-3
This article is cited by
-
Novel spino-modulating strategies for neurological-/functional-recovery in spinal cord injury
Health and Technology (2026)
-
Hebbian priming of human motor learning
Nature Communications (2024)
-
The Potential of Corticospinal-Motoneuronal Plasticity for Recovery after Spinal Cord Injury
Current Physical Medicine and Rehabilitation Reports (2020)