Abstract
Study design
Retrospective study.
Objectives
To check the hypothesis that irisin could mediate systemic metabolic effects of testosterone in men with chronic spinal cord injury (SCI).
Setting
Spinal Unit of the San Raffaele Institute in Sulmona.
Methods
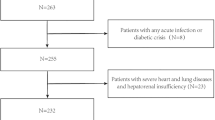
Fifteen men with chronic SCI admitted to a rehabilitation program were involved. They underwent clinical and biochemical evaluations. Irisin levels were measured with a high-sensitivity ELISA kit. Free testosterone levels were calculated (cFT) from total testosterone, sex hormone binding globulin, and albumin concentrations using the Vermeulen formula.
Results
Androgen deficiency (total testosterone <3 ng/ml and cFT <64 pg/ml) was found in 53% of participants and was associated with significantly lower irisin levels, higher body mass index (BMI), and higher triglycerides. Participants were engaged in significantly poorer leisure time physical activity (LTPA). Circulating irisin correlated with cFT (r = 0.55; p = 0.03) and both were negatively correlated with triglycerides levels, homeostatic model assessment of insulin resistance (HOMA-IR) and systemic inflammation, as assessed by erythrocyte sedimentation rate (ESR). Correlations with irisin did not reach statistical significance for either BMI (r = −0.40; p = 0.13) or LTPA (r = 0.46; p = 0.08). In bivariate linear regression models, lower irisin levels were significantly associated with higher triglycerides (β = −0.46; 95% CI: −0.75 to −0.16), HOMA-IR (β = −0.32; 95% CI: −0.63 to −0.004) and ESR (β = −0.89; 95% CI: −1.69 to −0.10) independently of cFT. Conversely, the negative associations of cFT with the same variables were lost after adjustment for irisin levels.
Conclusions
Spinal cord-injured men with androgen deficiency exhibit lower levels of irisin, which could mediate the systemic effects of testosterone.

Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Boström P, Wu J, Jedrychowski MP, Korde A, Ye L, Lo JC, et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481:463–8.
Torabi A, Reisi J, Kargarfard M, Mansourian M. Differences in the Impact of Various Types of Exercise on Irisin Levels: A Systematic Review and Meta-Analysis. Int J Prev Med. 2024;15:11.
Varela-Rodríguez BM, Pena-Bello L, Juiz-Valiña P, Vidal-Bretal B, Cordido F, Sangiao-Alvarellos S. FNDC5 expression and circulating irisin levels are modified by diet and hormonal conditions in hypothalamus, adipose tissue and muscle. Sci Rep. 2016;6:29898.
Aladag T, Mogulkoc R, Baltaci AK. Irisin and Energy Metabolism and the Role of Irisin on Metabolic Syndrome. Mini Rev Med Chem. 2023;23:1942–58.
Hu X, Wang Z, Wang W, Cui P, Kong C, Chen X, et al. Irisin as an agent for protecting against osteoporosis: A review of the current mechanisms and pathways. J Adv Res. 2024;62:175–86.
Zhang Y, Li R, Meng Y, Li S, Donelan W, Zhao Y, et al. Irisin stimulates browning of white adipocytes through mitogen-activated protein kinase p38 MAP kinase and ERK MAP kinase signaling. Diabetes. 2014;63:514–25.
Li Q, Tan Y, Chen S, Xiao X, Zhang M, Wu Q, et al. Irisin alleviates LPS-induced liver injury and inflammation through inhibition of NLRP3 inflammasome and NF-κB signaling. J Recept Signal Transduct Res. 2021;41:294–303.
Park MJ, Kim DI, Choi JH, Heo YR, Park SH. New role of irisin in hepatocytes: The protective effect of hepatic steatosis in vitro. Cell Signal. 2015;27:1831–9.
Ahmad IH, Mohamed Mostafa ER, Mohammed SA, Shipl W, Soliman AA, Said M. Correlations between serum testosterone and irisin levels in a sample of Egyptian men with metabolic syndrome; (case-control study). Arch Physiol Biochem. 2023;129:180–5.
Assyov Y, Gateva A, Karamfilova V, Gatev T, Nedeva I, Velikova T, et al. Impact of testosterone treatment on circulating irisin in men with late-onset hypogonadism and metabolic syndrome. Aging Male. 2021;23:1381–7.
Kamenov Z, Assyov Y, Angelova P, Gateva A, Tsakova A. Irisin and Testosterone in Men with Metabolic Syndrome. Horm Metab Res. 2017;49:755–9.
Barbonetti A, Castellini C, Francavilla S, Francavilla F, D’Andrea S. Metabolic syndrome in spinal cord injury: Impact on health. In: Rajendram R, Preedy VR, Martin RCR, editors. The Neuroscience of spinal cord injury: Cellular, molecular, physiological, and behavioral aspects of spinal cord injury. ELSEVIER, Acedemic press, London, England; 2022. pp 377–88.
Gorgey AS, Dolbow DR, Dolbow JD, Khalil RK, Castillo C, Gater DR. Effects of spinal cord injury on body composition and metabolic profile - Part I. J Spinal Cord Med. 2014;37:693–702.
Barbonetti A, Caterina Vassallo MR, Cotugno M, Felzani G, Francavilla S, Francavilla F. Low testosterone and non-alcoholic fatty liver disease: Evidence for their independent association in men with chronic spinal cord injury. J Spinal Cord Med. 2016;39:443–9.
Farkas GJ, Gater DR. Neurogenic obesity and systemic inflammation following spinal cord injury: A review. J Spinal Cord Med. 2018;41:378–87.
Goldsmith JA, Lai RE, Garten RS, Chen Q, Lesnefsky EJ, Perera RA, et al. Visceral Adiposity, Inflammation, and Testosterone Predict Skeletal Muscle Mitochondrial Mass and Activity in Chronic Spinal Cord Injury. Front Physiol. 2022;13:809845.
Bauman WA, La Fountaine MF, Spungen AM. Age-related prevalence of low testosterone in men with spinal cord injury. J Spinal Cord Med. 2014;37:32–39.
Barbonetti A, Vassallo MR, Felzani G, Francavilla S, Francavilla F. Association between 25(OH)-vitamin D and testosterone levels: Evidence from men with chronic spinal cord injury. J Spinal Cord Med. 2016;39:246–52.
Barbonetti A, Vassallo MR, Pacca F, Cavallo F, Costanzo M, Felzani G, et al. Correlates of low testosterone in men with chronic spinal cord injury. Andrology. 2014;2:721–8.
Abilmona SM, Sumrell RM, Gill RS, Adler RA, Gorgey AS. Serum testosterone levels may influence body composition and cardiometabolic health in men with spinal cord injury. Spinal Cord. 2019;57:229–39.
Harman SM, Metter EJ, Tobin JD, Pearson J, Blackman MR. Longitudinal effects of aging on serum total and free testosterone levels in healthy men. Baltim Longitud Study Aging J Clin Endocrinol Metab. 2001;86:724–31.
Maynard FM Jr, Bracken MB, Creasey G, Ditunno JF Jr, Donovan WH, Ducker TB, et al. International Standards for Neurological and Functional Classification of Spinal Cord Injury. American Spinal Injury Association. Spinal Cord. 1997;35:266–74.
Ginis KAM, Phang SH, Latimer AE, Arbour-Nicitopoulos KP. Reliability and validity tests of the leisure time physical activity questionnaire for people with spinal cord injury. Arch Phys Med Rehabil. 2012;93:677–82.
Barbonetti A, Sperandio A, Micillo A, D’Andrea S, Pacca F, Felzani G, et al. Independent Association of Vitamin D with Physical Function in People with Chronic Spinal Cord Injury. Arch Phys Med Rehabil. 2016;97:726–32.
Wu FC, Tajar A, Beynon JM, Pye SR, Silman AJ, Finn JD, et al. EMAS Group. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med. 2010;363:123–35.
Jedrychowski MP, Wrann CD, Paulo JA, Gerber KK, Szpyt J, Robinson MM, et al. Detection and Quantitation of Circulating Human Irisin by Tandem Mass Spectrometry. Cell Metab. 2015;22:734–40.
Yang S, Xiao F, Pan L, Zhang H, Ma Z, Liu S, et al. Association of serum irisin and body composition with chronic kidney disease in obese Chinese adults: a cross-sectional study. BMC Nephrol. 2015;16:16.
Moreno-Navarrete JM, Ortega F, Serrano M, Guerra E, Pardo G, Tinahones F, et al. Irisin is expressed and produced by human muscle and adipose tissue in association with obesity and insulin resistance. J Clin Endocrinol Metab. 2013;98:E769–78.
Isidori AM, Caprio M, Strollo F, Moretti C, Frajese G, Isidori A, et al. Leptin and androgens in male obesity: evidence for leptin contribution to reduced androgen levels. J Clin Endocrinol Metab. 1999;84:3673–80.
Saez JM. Leydig cells: endocrine, paracrine, and autocrine regulation. Endocr Rev. 1994;15:574–626.
Kalra PS, Edwards TG, Xu B, Jain M, Kalra SP. The anti-gonadotropic effects of cytokines: the role of neuropeptides. Domest Anim Endocrinol. 1998;15:321–32.
Assyov Y, Gateva A, Karamfilova V, Gatev T, Nedeva I, Velikova T, et al. Impact of testosterone treatment on circulating irisin in men with late-onset hypogonadism and metabolic syndrome. Aging Male. 2020;23:1381–7.
Huhtaniemi IT, Tajar A, Lee DM, O’Neill TW, Finn JD, Bartfai G, et al. EMAS Group. Comparison of serum testosterone and estradiol measurements in 3174 European men using platform immunoassay and mass spectrometry; relevance for the diagnostics in aging men. Eur J Endocrinol. 2012;166:983–91.
Jones LM, Legge M, Goulding A. Healthy body mass index values often underestimate body fat in men with spinal cord injury. Arch Phys Med Rehabil. 2003;84:1068–71.
Ravensbergen HR, Lear SA, Claydon VE. Waist circumference is the best index for obesity-related cardiovascular disease risk in individuals with spinal cord injury. J Neurotrauma. 2014;31:292–300.
Acknowledgements
The authors are grateful to Laura Marra for her valuable support in language editing.
Author information
Authors and Affiliations
Contributions
AB, CV and VG conceived the study and performed literature search; VC performed irisin assays; DT and AB performed statistical analysis; CT and LS prepared table and figure; CV, VG and GF drafted the manuscript; AB, MM and MGB supervised the manuscript writing and critically revised the manuscript for intellectual content. All authors read and approved the final manuscript. The corresponding author confirms that he has had full access to the data in the study and bears responsibility for submitting the paper for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study was approved by the Ethics Committee of L’Aquila and Teramo provinces, Italy (Approval Code: 11/CE/15 of May 7, 2015).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Venditti, C., Carnicelli, V., Gizzi, V. et al. Does irisin mediate metabolic effects of androgen deficiency? A cross-sectional study in men with chronic spinal cord injury. Spinal Cord 63, 80–85 (2025). https://doi.org/10.1038/s41393-024-01056-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41393-024-01056-8