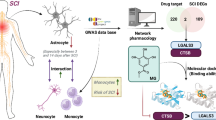
Abstract
Study design
An integrated bioinformatics data study.
Objective
This study, through bioinformatics analysis, aims to map the landscape of astrocytes, explore key signaling pathways, and uncover molecular mechanisms that support SCI recovery facilitated by MSCs and iPSCs.
Setting
Beijing Tsinghua Changgung Hospital, School of Clinical Medicine, Tsinghua University.
Methods
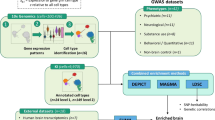
We performed a bioinformatics analysis of single-cell transcriptomes (scRNA-seq), spatial transcriptomics, and bulk RNA-seq data sourced from Gene Expression Omnibus (GEO) datasets. The data processing involved R packages like “Seurat,” “DESeq2,” and “WGCNA.” For pathway enrichment, we used Gene Set Enrichment Analysis (GSEA) and the Enrichr web server.
Results
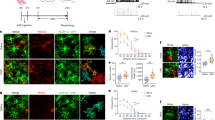
Single-cell and spatial transcriptomic analysis revealed notable changes in the astrocyte landscape after SCI, highlighting a significant disruption in astrocyte populations within the injured region. Findings suggest that BDNF regulation of GABA neurotransmission and GABA receptor signaling in astrocytes plays a key role in promoting neuronal regeneration. Additionally, hUC-MSCs were found to enhance neural repair by activating BDNF-regulated GABA signaling of astrocytes. A promising alternative involves iPS-derived MSCs, which have shown potential to boost neural regeneration through BDNF, GABA, and GABA receptor signaling pathways of astrocytes.
Conclusions
In summary, SCI disrupts astrocyte populations, impacting their ability to support neural repair. BDNF-regulated GABA signaling in astrocytes is essential for neuron regeneration. Both hUC-MSCs and iPS-derived MSCs show promise in enhancing neural recovery by activating these pathways, offering potential new therapeutic options for SCI.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data sources and handling of the publicly available datasets used in this study are described in the Materials and Methods. All source code developed for this study is proprietary and cannot be shared publicly. However, researchers interested in accessing the code for non-commercial purposes may contact the corresponding author, Yifeng Sun, at syf498054232@163.com.
References
Simpson LA, Eng JJ, Hsieh JT, Wolfe DL. the Spinal Cord Injury Rehabilitation Evidence Research Team. The health and life priorities of individuals with spinal cord injury: a systematic review. J Neurotrauma. 2012;29:1548–55.
Gaudet AD, Fonken LK. Glial cells shape pathology and repair after spinal cord injury. Neurotherapeutics. 2018;15:554–77.
Orr MB, Gensel JC. Spinal cord injury scarring and inflammation: therapies targeting glial and inflammatory responses. Neurotherapeutics. 2018;15:541–53.
Pirttimaki TM, Parri HR. Astrocyte plasticity: implications for synaptic and neuronal activity. Neuroscientist. 2013;19:604–15.
Windle WF. Regeneration of axons in the vertebrate central nervous system. Physiol Rev. 1956;36:427–40.
Chun H, An H, Lim J, Woo J, Lee J, Ryu H, et al. Astrocytic proBDNF and tonic GABA distinguish active versus reactive astrocytes in hippocampus. Exp Neurobiol. 2018;27:155.
Cao T, Chen H, Huang W, Xu S, Liu P, Zou W, et al. hUC-MSC-mediated recovery of subacute spinal cord injury through enhancing the pivotal subunits β3 and γ2 of the GABAA receptor. Theranostics. 2022;12:3057.
Nagoshi N, Okano H. iPSC-derived neural precursor cells: potential for cell transplantation therapy in spinal cord injury. Cell Mol Life Sci. 2018;75:989–1000.
Dai M, Pei X, Wang X-J. Accurate and fast cell marker gene identification with COSG. Brief Bioinform. 2022;23:bbab579.
Sun Y, Zhang C, Fang Q, Zhang W, Liu W. Abnormal signal pathways and tumor heterogeneity in osteosarcoma. J Transl Med. 2023;21:99.
Anderson MA, Burda JE, Ren Y, Ao Y, O’Shea TM, Kawaguchi R, et al. Astrocyte scar formation aids central nervous system axon regeneration. Nature. 2016;532:195–200.
Xiao S, Zhang Y, Liu Z, Li A, Tong W, Xiong X, et al. Alpinetin inhibits neuroinflammation and neuronal apoptosis via targeting the JAK2/STAT3 signaling pathway in spinal cord injury. CNS Neurosci Ther. 2023;29:1094–108.
Lee JY, Park CS, Seo KJ, Kim IY, Han S, Youn I, et al. IL-6/JAK2/STAT3 axis mediates neuropathic pain by regulating astrocyte and microglia activation after spinal cord injury. Exp Neurol. 2023;370:114576.
Clifford T, Finkel Z, Rodriguez B, Joseph A, Cai L. Current advancements in spinal cord injury research-glial scar formation and neural regeneration. Cells. 2023;12:853.
Kamiya D, Takenaka-Ninagawa N, Motoike S, Kajiya M, Akaboshi T, Zhao C, et al. Induction of functional xeno-free MSCs from human iPSCs via a neural crest cell lineage. NPJ Regen Med. 2022;7:47.
Kamata Y, Isoda M, Sanosaka T, Shibata R, Ito S, Okubo T, et al. A robust culture system to generate neural progenitors with gliogenic competence from clinically relevant induced pluripotent stem cells for treatment of spinal cord injury. Stem Cells Transl Med. 2021;10:398–413.
Wang R, Zhou R, Chen Z, Gao S, Zhou F. The glial cells respond to spinal cord injury. Front Neurol. 2022;13:844497.
Tomoda T, Sumitomo A, Shukla R, Hirota-Tsuyada Y, Miyachi H, Oh H, et al. BDNF controls GABAAR trafficking and related cognitive processes via autophagic regulation of p62. Neuropsychopharmacology. 2022;47:553–63.
Vaz SH, Jørgensen TN, Cristóvão-Ferreira S, Duflot S, Ribeiro JA, Gether U, et al. Brain-derived neurotrophic factor (BDNF) enhances GABA transport by modulating the trafficking of GABA transporter-1 (GAT-1) from the plasma membrane of rat cortical astrocytes. J Biol Chem. 2011;286:40464–76.
Liu J, Feng X, Wang Y, Xia X, Zheng JC: Astrocytes: GABAceptive and GABAergic Cells in the Brain. Front Cell Neurosci.2022;16:892497.
Boddum K, Jensen TP, Magloire V, Kristiansen U, Rusakov DA, Pavlov I, et al. Astrocytic GABA transporter activity modulates excitatory neurotransmission. Nat Commun. 2016;7:13572.
Bolteus AJ, Bordey A. GABA release and uptake regulate neuronal precursor migration in the postnatal subventricular zone. J Neurosci. 2004;24:7623–31.
Ishibashi M, Egawa K, Fukuda A. Diverse actions of astrocytes in GABAergic signaling. Int J Mol Sci. 2019;20:2964.
Kang J, Guo Y. Human umbilical cord mesenchymal stem cells derived exosomes promote neurological function recovery in a rat spinal cord injury model. Neurochem Res. 2022;47:1532–40.
Nishida F, Zappa Villar MF, Zanuzzi CN, Sisti MS, Camiña AE, Reggiani PC, et al. Intracerebroventricular delivery of human umbilical cord mesenchymal stem cells as a promising therapy for repairing the spinal cord injury induced by kainic acid. Stem Cell Rev Rep. 2020;16:167–80.
Khazaei M, Siddiqui AM, Fehlings MG. The potential for iPS-derived stem cells as a therapeutic strategy for spinal cord injury: opportunities and challenges. J Clin Med. 2014;4:37–65.
Li Y, Shen P-P, Wang B. Induced pluripotent stem cell technology for spinal cord injury: a promising alternative therapy. Neural Regen Res. 2021;16:1500.
Watt FM, Driskell RR. The therapeutic potential of stem cells. Philos Trans R Soc Lond B Biol Sci. 2010;365:155–63.
Yang Y, Pang M, Chen YY, Zhang LM, Liu H, Tan J, et al. Human umbilical cord mesenchymal stem cells to treat spinal cord injury in the early chronic phase: study protocol for a prospective, multicenter, randomized, placebo-controlled, single-blinded clinical trial. Neural Regen Res. 2020;15:1532–8.
Kawai M, Nagoshi N, Okano H, Nakamura M. A review of regenerative therapy for spinal cord injury using human iPS cells. N Am Spine Soc J. 2023;13:100184.
Funding
This work was supported by National Natural Science Foundation of China (81702667), and China Postdoctoral Science Foundation (8206300728).
Author information
Authors and Affiliations
Contributions
YS conceived the study and generated figures and wrote the manuscript, QZ collected and analysed the data, QF performed a part of data analysis. CZ and WL help to generated figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This is a bioinformatic analysis; therefore, ethics approval and consent to participate are not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, Q., Fang, Q., Zhang, C. et al. BDNF-GABA signaling in astrocytes: enhancing neural repair after SCI through MSC therapies. Spinal Cord 63, 263–269 (2025). https://doi.org/10.1038/s41393-025-01077-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41393-025-01077-x