Abstract
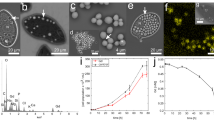
The central Baltic Sea is characterized by a pelagic redox zone exhibiting high dark CO2 fixation rates below the chemocline. These rates are mainly driven by chemolithoautotrophic and denitrifying Sulfurimonas GD17 subgroup cells which are motile and fast-reacting r-strategists. Baltic Sea redox zones are unstable and a measurable overlap of nitrate and reduced sulfur, essential for chemosynthesis, is often only available on small scales and short times due to local mixing events. This raises the question of how GD17 cells gain access to electron donors or acceptors over longer term periods and under substrate deficiency. One possible answer is that GD17 cells store high-energy-containing polyphosphate during favorable nutrient conditions to survive periods of nutrient starvation. We used scanning electron microscopy with energy-dispersive X-ray spectroscopy to investigate potential substrate enrichments in single GD17 cells collected from Baltic Sea redox zones. More specific substrate enrichment features were identified in experiments using Sulfurimonas gotlandica GD1T, a GD17 representative. Sulfurimonas cells accumulated polyphosphate both in situ and in vitro. Combined genome and culture-dependent analyses suggest that polyphosphate serves as an energy reservoir to maintain cellular integrity at unfavorable substrate conditions. This redox-independent energy supply would be a precondition for sustaining the r-strategy lifestyle of GD17 and may represent a newly identified survival strategy for chemolithoautotrophic prokaryotes occupying eutrophic redox zones.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Carstensen J, Andersen JH, Gustafsson BG, Conley DJ. Deoxygenation of the Baltic Sea during the last century. Proc Natl Acad Sci USA. 2014;111:5628–33.
Conley DJ, Björck S, Bonsdorff E, Carstensen J, Destouni G, Gustafsson BG, et al. Hypoxia-related processes in the Baltic Sea. Environ Sci Technol. 2009;43:3412–20.
Jørgensen BB, Fossing H, Wirsen CO, Jannasch HW. Sulfide oxidation in the anoxic Black Sea chemocline. Deep Sea Res Part 1 Oceanogr Res Pap. 1991;38:1083–103.
Taylor GT, Iabichella M, Ho TY, Scranton MI, Thunell RC, Muller-Karger F, et al. Chemoautotrophy in the redox transition zone of the Cariaco Basin: a significant mid-water source of organic carbon production. Limnol Oceanogr. 2001;46:148–63.
Brettar I, Rheinheimer G. Denitrification in the central Baltic: evidence for H2S-oxidation as motor of denitrification at the oxic-anoxic interface. Mar Ecol Prog Ser. 1991;77:157–69.
Brüchert V, Jørgensen BB, Neumann K, Riechmann D, Schlösser M, Schulz H. Regulation of bacterial sulfate reduction and hydrogen sulfide fluxes in the central Namibian coastal upwelling zone. Geochim Chosmochim Acta. 2003;67:4505–18.
Canfield DE, Stewart FJ, Thamdrup B, De Brabandere L, Dalsgaard T, DeLong EF, et al. A cryptic sulfur cycle in oxygen-minimum-zone waters off the Chilean Coast. Science. 2010;330:1375–8.
Lam P, Kuypers MM. Microbial nitrogen cycling processes in oxygen minimum zones. Ann Rev Mar Sci. 2011;3:317–45.
Grote J, Labrenz M, Pfeiffer B, Jost G, Jürgens K. Quantitative distributions of Epsilonproteobacteria and a Sulfurimonas subgroup in pelagic redoxclines of the central Baltic Sea. Appl Environ Microbiol. 2007;73:7155–61.
Labrenz M, Brettar I, Christen R, Flavier S, Botel J, Höfle MG. Development and application of a real-time PCR approach for quantification of uncultured bacteria in the central Baltic Sea. Appl Environ Microbiol. 2004;70:4971–9.
Brettar I, Labrenz M, Flavier S, Botel J, Kuosa H, Christen R, et al. Identification of a Thiomicrospira denitrificans-like epsilonproteobacterium as a catalyst for autotrophic denitrification in the central Baltic Sea. Appl Environ Microbiol. 2006;72:1364–72.
Grote J, Schott T, Bruckner CG, Glöckner FO, Jost G, Teeling H, et al. Genome and physiology of a model Epsilonproteobacterium responsible for sulfide detoxification in marine oxygen depletion zones. Proc Natl Acad Sci USA. 2012;109:506–10.
Rogge A, Vogts A, Voss M, Jürgens K, Günter J, Labrenz M. NanoSIMS analyses reveal different lifestyles of chemolithoautotrophic SUP05 and Sulfurimonas GD17 cells in a pelagic Baltic Sea redox zone. Env Microbiol. 2017;19:2495–506.
Labrenz M, Grote J, Mammitzsch K, Boschker HTS, Laue M, Jost G, et al. Sulfurimonas gotlandica sp. nov., a chemoautotrophic and psychrotolerant epsilonproteobacterium isolated from a pelagic redoxcline, and an emended description of the genus Sulfurimonas. Int J Syst Evol Micr. 2013;63:4141–8.
Axell LB. On the variability of Baltic Sea deepwater mixing. J Geophys Res Oceans. 1998;103:21667–82.
Otte S, Kuenen JG, Nielsen LP, Paerl HW, Zopfi J, Schulz HN, et al. Nitrogen, carbon, and sulfur metabolism in natural Thioploca samples. Appl Environ Microbiol. 1999;65:3148–57.
Pasteris JD, Freeman JJ, Goffredi SK, Buck KR. Raman spectroscopic and laser scanning confocal microscopic analysis of sulfur in living sulfur-precipitating marine bacteria. Chem Geol. 2001;180:3–18.
Preisler A, De Beer D, Lichtschlag A, Lavik G, Boetius A, Jørgensen BB. Biological and chemical sulfide oxidation in a Beggiatoa inhabited marine sediment. ISME J. 2007;1:341–53.
Schulz HN. The genus Thiomargarita. In: Dworkin M, Falkow S, Rosenberg E, Schleifer K-H, Stackebrandt E, editors. The prokaryotes. New York: Springer; 2006. p. S. 1156–63.
Brock J, Schulz-Vogt HN. Sulfide induces phosphate release from polyphosphate in cultures of a marine Beggiatoa strain. ISME J. 2011;5:497–506.
Harold FM. Depletion and replenishment of the inorganic polyphosphate pool in Neurospora crassa. J Bacteriol. 1962;83:1047–57.
Kornberg A. Inorganic polyphosphate: toward making a forgotten polymer unforgettable. J Bacteriol. 1995;177:491–6.
Baker AL, Schmidt RR. Induced utilization of polyphosphate during nuclear division in synchronously growing. Chlorella BBA Gen Subj. 1964;93:180–2.
Hsieh PC, Shenoy BC, Haase FC, Jentoft JE, Phillips NF. Involvement of tryptophan(s) at the active site of polyphosphate/ATP glucokinase from Mycobacterium tuberculosis. Biochemistry. 1993;32:6243–9.
Skorko R. Polyphosphate as a source of phosphoryl group in protein modification in the archaebacterium Sulfolobus acidocaldarius. Biochimie. 1989;71:1089–93.
Ault-Riché D, Fraley CD, Tzeng C-M, Kornberg A. Novel assay reveals multiple pathways regulating stress-induced accumulations of inorganic polyphosphate in Escherichia coli. J Bacteriol. 1998;180:1841–7.
Rao NN, Kornberg A. Inorganic polyphosphate supports resistance and survival of stationary-phase Escherichia coli. J Bacteriol. 1996;178:1394–400.
Sureka K, Dey S, Datta P, Singh AK, Dasgupta A, Rodrigue S, et al. Polyphosphate kinase is involved in stress‐induced mprAB‐sigE‐rel signalling in mycobacteria. Mol Microbiol. 2007;65:261–76.
Kim HY, Schlictman D, Shankar S, Xie Z, Chakrabarty AM, Kornberg A. Alginate, inorganic polyphosphate, GTP and ppGpp synthesis co‐regulated in Pseudomonas aeruginosa: implications for stationary phase survival and synthesis of RNA/DNA precursors. Mol Microbiol. 1998;27:717–25.
Pick U, Chitlaru E, Weiss M. Polyphosphate-hydrolysis-a protective mechanism against alkaline stress? FEBS Lett. 1990;274:15–8.
Blum E, Py B, Carpousis AJ, Higgins CF. Polyphosphate kinase is a component of the Escherichia coli RNA degradosome. Mol Microbiol. 1997;26:387–98.
Bonting CF, Kortstee GJ, Zehnder AJ. Properties of polyphosphate: AMP phosphotransferase of Acinetobacter strain 210A. J Bacteriol. 1991;173:6484–8.
Seufferheld MJ, Alvarez HM, Farias ME. Role of polyphosphates in microbial adaptation to extreme environments. Appl Environ Microbiol. 2008;74:5867–74.
Varela C, Mauriaca C, Paradela A, Albar JP, Jerez CA, Chávez FP. et al. New structural and functional defects in polyphosphate deficient bacteria: a cellular and proteomic study. BMC Microbiol. 2010;10:7
Rivas-Lamelo S, Benzerara K, Lefèvre CT, Monteil CL, Jézéquel D, Menguy N. et al. Magnetotactic bacteria as a new model for P sequestration in the ferruginous Lake Pavin. Geochem Persp Let. 2017;5:35–41.
Jost G, Zubkov MV, Yakushev E, Labrenz M, Jürgens K. High abundance and dark CO2 fixation of chemolithoautotrophic prokaryotes in anoxic waters of the Baltic Sea. Limnol Oceanogr. 2008;53:14–22.
Laas P, Šatova E, Lips I, Lips U, Simm J, Kisand V, et al. Near-bottom hypoxia impacts dynamics of bacterioplankton assemblage throughout water column of the Gulf of Finland (Baltic Sea). PloS ONE. 2016;11:e0156147.
Bruns A, Cypionka H, Overmann J. Cyclic AMP and acyl homoserine lactones increase the cultivation efficiency of heterotrophic bacteria from the central Baltic Sea. Appl Environ Microbiol. 2002;68:3978–87.
Martin P, Van Mooy BA. Fluorometric quantification of polyphosphate in environmental plankton samples: extraction protocols, matrix effects, and nucleic acid interference. Appl Environ Microbiol. 2013;79:273–81.
Kulakova AN, Hobbs D, Smithen M, Pavlov E, Gilbert JA, Quinn JP, et al. Direct quantification of inorganic polyphosphate in microbial cells using 4′-6-diamidino-2-phenylindole (DAPI). Environ Sci Technol. 2011;45:7799–803.
Fischer JP, Cypionka H. Analysis of aerotactic band formation by Desulfovibrio desulfuricans in a stopped-flow diffusion chamber. FEMS Microbiol Ecol. 2006;55:186–94.
Adler J, Dahl MM. A method for measuring the motility of bacteria and for comparing random and non-random motility. J Gen Microbiol. 1967;46:161–73.
Grote J, Jost G, Labrenz M, Herndl GJ, Jürgens K. Epsilonproteobacteria represent the major portion of chemoautotrophic bacteria in sulfidic waters of pelagic redoxclines of the Baltic and Black Seas. Appl Environ Microbiol. 2008;74:7546–51.
Friedberg I, Avigad G. Structures containing polyphosphate in Micrococcus lysodeikticus. J Bacteriol. 1968;96:544–53.
Morrissey JH, Choi SH, Smith SA. Polyphosphate: an ancient molecule that links platelets, coagulation, and inflammation. Blood. 2012;119:5972–9.
Peverly JH, Adamec J, Parthasarathy MV. Association of potassium and some other monovalent cations with occurrence of polyphosphate bodies in Chlorella pyrenoidosa. Plant Physiol. 1978;62:120–6.
Gomes F, Ramos I, Wendt C, Girard-Dias W, De Souza W, Machado EA, et al. New insights into the in situ microscopic visualization and quantification of inorganic polyphosphate stores by 4’, 6-diamidino-2-phenylindole (DAPI)-staining. Eur J Histochem. 2013;57:227–5.
Liu WT, Nielsen AT, Wu JH, Tsai CS, Matsuo Y, Molin S. In situ identification of polyphosphate‐and polyhydroxyalkanoate‐accumulating traits for microbial populations in a biological phosphorus removal process. Environ Microbiol. 2001;3:110–22.
Kulaev IS, Vagabov VM, Kulakovskaya TV. Localization of polyphosphates in cells of prokaryotes and eukaryotes. In: Kulaev IS, Vagabov VM, Kulakovskaya TV, editors. The biochemistry of inorganic polyphosphates. 2nd ed. Chichester: John Wiley & Sons; 2004. p. 53–4.
Tijssen JPF, Beekes HW, Van Steveninck J. Localization of polyphosphates in Saccharomyces fragilis, as revealed by 4′, 6-diamidino-2-phenylindole fluorescence. BBA-Mol Cell Res. 1982;721:394–8.
Kolozsvari B, Parisi F, Saiardi A. Inositol phosphates induce DAPI fluorescence shift. Biochem J. 2014;460:377–85.
Streichan M, Golecki JR, Schön G. Polyphosphate‐accumulating bacteria from sewage plants with different processes for biological phosphorus removal. FEMS Microbiol Ecol. 1990;6:113–24.
Chávez FP, Lünsdorf H, Jerez CA. Growth of polychlorinated-biphenyl-degrading bacteria in the presence of biphenyl and chlorobiphenyls generates oxidative stress and massive accumulation of inorganic polyphosphate. Appl Environ Microbiol. 2004;70:3064–72.
Clark JE, Beegen H, Wood HG. Isolation of intact chains of polyphosphate from” Propionibacterium shermanii” grown on glucose or lactate. J Bacteriol. 1986;168:1212–9.
Mammitzsch K, Jost G, Jürgens K. Impact of dissolved inorganic carbon concentrations and pH on growth of the chemolithoautotrophic epsilonproteobacterium Sulfurimonas gotlandica GD1T. MicrobiologyOpen. 2014;3:80–88.
Tebbe FN, Wasserman E, Peet WG, Vatvars A, Hayman AC. Composition of elemental sulfur in solution: equilibrium of S6, S7 and S8 at ambient temperatures. J Am Chem Soc. 1982;104:4971–2.
Kamyshny JrA. Solubility of cyclooctasulfur in pure water and sea water at different temperatures. Geochim Cosmochim Acta. 2009;73:6022–8.
Ahn K, Kornberg A. Polyphosphate kinase from Escherichia coli. Purification and demonstration of a phosphoenzyme intermediate. J Biol Chem. 1990;265:11734–9.
Motomura K, Hirota R, Okada M, Ikeda T, Ishida T, Kuroda A. A new subfamily of polyphosphate kinase 2 (class III PPK2) catalyzes both nucleoside monophosphate phosphorylation and nucleoside diphosphate phosphorylation. Appl Environ Microbiol. 2014;80:2602–8.
Sureka K, Sanyal S, Basu J, Kundu M. Polyphosphate kinase 2: a modulator of nucleoside diphosphate kinase activity in mycobacteria. Mol Microbiol. 2009;74:1187–97.
Kawakoshi A, Nakazawa H, Fukada J, Sasagawa M, Katano Y, Nakamura S, et al. Deciphering the genome of polyphosphate accumulating actinobacterium Microlunatus phosphovorus. DNA Res. 2012;19:383–94.
Saunders AM, Mabbett AN, McEwan AG, Blackall LL. Proton motive force generation from stored polymers for the uptake of acetate under anaerobic conditions. FEMS Microbiol Lett. 2007;274:245–51.
Jones DS, Flood BE, Bailey JV. Metatranscriptomic insights into polyphosphate metabolism in marine sediments. ISME J. 2016;10:1015–9.
Pirt SJ. Maintenance energy of bacteria in growing cultures. Proc R Soc Lond B Biol Sci. 1965;163:224–31.
Larsen MH, Blackburn N, Larsen JL, Olsen JE. Influences of temperature, salinity and starvation on the motility and chemotactic response of Vibrio anguillarum. Microbiology. 2004;150:1283–90.
Mukhopadhyay B, Johnson EF, Wolfe RS. A novel pH2 control on the expression of flagella in the hyperthermophilic strictly hydrogenotrophic methanarchaeaon Methanococcus jannaschii. Proc Natl Acad Sci USA. 2000;97:11522–7.
Stocker R, Seymour JR, Samadani A, Hunt DE, Polz MF. Rapid chemotactic response enables marine bacteria to exploit ephemeral microscale nutrient patches. Proc Natl Acad Sci USA. 2008;105:4209–14.
Li G, Tang JX. Low flagellar motor torque and high swimming efficiency of Caulobacter crescentus swarmer cells. Biophys J. 2006;91:2726–34.
Thureborn P, Lundin D, Plathan J, Poole AM, Sjöberg B-M, Sjöling S. A metagenomics transect into the deepest point of the Baltic Sea reveals clear stratification of microbial functional capacities. PLOS ONE. 2013;8:e74983.
Long RA, Azam F. Antagonistic interactions among marine pelagic bacteria. Appl Environ Microbiol. 2001;67:4975–83.
Stocker R. Marine microbes see a sea of gradients. Science. 2012;338:628–33.
Acknowledgements
We are grateful to the captains and crews of the R/V Salme and R/V Poseidon. The excellent technical assistance of Ronny Baaske, Heike Brockmöller, Sascha Plewe, and Fabiola Henning is greatly appreciated. Special thanks goes to Heribert Cypionka for his essential advice concerning the freeware program “Trace”; and HC as well as Heide Schulz-Vogt for helpful discussions. We thank the Deutsche Forschungsgemeinschaft (DFG) for grants LA 1466/4-1, 4-2, the Baltisch-Deutsches Hochschulkontor, and the Leibniz Institute for Baltic Sea Research Warnemünde (IOW) for funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Möller, L., Laas, P., Rogge, A. et al. Sulfurimonas subgroup GD17 cells accumulate polyphosphate under fluctuating redox conditions in the Baltic Sea: possible implications for their ecology. ISME J 13, 482–493 (2019). https://doi.org/10.1038/s41396-018-0267-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41396-018-0267-x
This article is cited by
-
Biogenic polyphosphate as relevant regulator of seasonal phosphate storage in surface sediments of stratified eutrophic lakes
Biogeochemistry (2025)
-
Bradymonabacteria, a novel bacterial predator group with versatile survival strategies in saline environments
Microbiome (2020)
-
Effect of large magnetotactic bacteria with polyphosphate inclusions on the phosphate profile of the suboxic zone in the Black Sea
The ISME Journal (2019)
-
Picoplankton accumulate and recycle polyphosphate to support high primary productivity in coastal Lake Ontario
Scientific Reports (2019)