Abstract
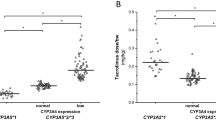
Tacrolimus, a calcineurin inhibitor with a narrow therapeutic index, requires precise dosing to optimize efficacy and minimize adverse effects in kidney transplant recipients. Although CYP3A5 genetic variants influence tacrolimus pharmacokinetics, they do not fully explain inter-individual differences. This retrospective study evaluated the combined impact of CYP3A4 [*1B (rs2740574), *1 G (rs2242480), *22 (rs35599367)] and CYP3A5 [*3 (rs776746), *6 (rs10264272), *7 (rs41303343)] genetic variants, as CYP3A phenotypes, on tacrolimus dose-adjusted trough concentrations (C0/D), in 94 Greek kidney transplant recipients at five time points during the first-year post-transplantation. Significant differences in tacrolimus C0/D ratios were observed across the groups. Group 4 (CYP3A5 expressers, carriers of CYP3A4*1B or *1 G) had consistently lower C0/D ratios compared to Groups 1 and 2 (CYP3A5 nonexpressers, carriers of CYP3A4*22 or CYP3A4 *1/*1) at multiple timepoints (p ≤ 0.022 and p ≤ 0.004, respectively). These findings suggest that CYP3A phenotypes could improve tacrolimus dosing decisions in kidney transplant recipients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Brunet M, van Gelder T, Åsberg A, Haufroid V, Hesselink DA, Langman L, et al. Therapeutic Drug Monitoring of Tacrolimus-Personalized Therapy: Second Consensus Report. Ther Drug Monit. 2019;41:261–307.
Shuker N, van Gelder T, Hesselink DA. Intra-patient variability in tacrolimus exposure: Causes, consequences for clinical management. Transplant Rev. 2015;29:78–84.
Zhang X, Lin G, Tan L, Li J. Current progress of tacrolimus dosing in solid organ transplant recipients: Pharmacogenetic considerations. Biomedicine & Pharmacotherapy. 2018;102:107–14.
Passey C, Birnbaum AK, Brundage RC, Oetting WS, Israni AK, Jacobson PA. Dosing equation for tacrolimus using genetic variants and clinical factors. Br J Clin Pharmacol. 2011;72:948–57.
Picard N, Bergan S, Marquet P, van Gelder T, Wallemacq P, Hesselink DA, et al. Pharmacogenetic Biomarkers Predictive of the Pharmacokinetics and Pharmacodynamics of Immunosuppressive Drugs. Ther Drug Monit. 2016;38:S57–69.
Brunet M, Pastor-Anglada M. Insights into the Pharmacogenetics of Tacrolimus Pharmacokinetics and Pharmacodynamics. Pharmaceutics. 2022;14:1755.
Haufroid V, Mourad M, Van Kerckhove V, Wawrzyniak J, De Meyer M, Eddour DC, et al. The effect of CYP3A5 and MDR1 (ABCB1) polymorphisms on cyclosporine and tacrolimus dose requirements and trough blood levels in stable renal transplant patients. Pharmacogenetics. 2004;14:147–54.
Kuehl P, Zhang J, Lin Y, Lamba J, Assem M, Schuetz J, et al. Sequence diversity in CYP3A promoters and characterization of the genetic basis of polymorphic CYP3A5 expression. Nat Genet. 2001;27:383–91.
Birdwell K, Decker B, Barbarino J, Peterson J, Stein C, Sadee W, et al. Clinical Pharmacogenetics Implementation Consortium (CPIC) Guidelines for CYP3A5 Genotype and Tacrolimus Dosing. Clin Pharmacol Ther. 2015;98:19–24.
Liu M, Hernandez S, Aquilante CL, Deininger KM, Lindenfeld J, Schlendorf KH, et al. Composite CYP3A (CYP3A4 and CYP3A5) phenotypes and influence on tacrolimus dose adjusted concentrations in adult heart transplant recipients. Pharmacogenomics J. 2024;24:4.
Wang D, Guo Y, Wrighton SA, Cooke GE, Sadee W. Intronic polymorphism in CYP3A4 affects hepatic expression and response to statin drugs. Pharmacogenomics J. 2011;11:274–86.
Shi WL, Tang HL, Zhai SD. Effects of the CYP3A4*1B Genetic Polymorphism on the Pharmacokinetics of Tacrolimus in Adult Renal Transplant Recipients: A Meta-Analysis. PLoS One. 2015;10:e0127995.
Furuse M, Hosomi S, Nishida Y, Itani S, Nadatani Y, Fukunaga S, et al. The impact of cytochrome P450 3A genetic polymorphisms on tacrolimus pharmacokinetics in ulcerative colitis patients. PLoS One. 2021;16:e0250597.
Zuo, Ng Xcong, Barrett CM, Luo JS, jing A, Zhang Bkui, et al. Effects of CYP3A4 and CYP3A5 polymorphisms on tacrolimus pharmacokinetics in Chinese adult renal transplant recipients. Pharmacogenet Genomics. 2013;23:251–61.
Mendrinou E, Mashaly ME, Al Okily AM, Mohamed ME, Refaie AF, Elsawy EM, et al. CYP3A5 Gene-Guided Tacrolimus Treatment of Living-Donor Egyptian Kidney Transplanted Patients. Front Pharmacol. 2020;11:1–7.
Brazeau DA, Attwood K, Meaney CJ, Wilding GE, Consiglio JD, Chang SS, et al. Beyond Single Nucleotide Polymorphisms: CYP3A5∗3∗6∗7 Composite and ABCB1 Haplotype Associations to Tacrolimus Pharmacokinetics in Black and White Renal Transplant Recipients. Front Genet. 2020;11:11.
Machiela MJ, Chanock SJ. LDlink: a web-based application for exploring population-specific haplotype structure and linking correlated alleles of possible functional variants. Bioinformatics. 2015;31:3555–7.
Oetting WS, Schladt DP, Guan W, Miller MB, Remmel RP, Dorr C, et al. Genomewide Association Study of Tacrolimus Concentrations in African American Kidney Transplant Recipients Identifies Multiple CYP3A5 Alleles. American Journal of Transplantation. 2016;16:574–82.
Suarez-Kurtz G, Struchiner CJ. Pharmacogenomic implications of the differential distribution of CYP3A5 metabolic phenotypes among Latin American populations. Pharmacogenomics. 2024;25:187–95.
Pallet N, Jannot AS, El Bahri M, Etienne I, Buchler M, de Ligny BH, et al. Kidney Transplant Recipients Carrying the CYP3A4*22 Allelic Variant Have Reduced Tacrolimus Clearance and Often Reach Supratherapeutic Tacrolimus Concentrations. American Journal of Transplantation. 2015;15:800–5.
Kim JS, Shim S, Yee J, Choi KH, Gwak HS Effects of CYP3A4*22 polymorphism on trough concentration of tacrolimus in kidney transplantation: a systematic review and meta-analysis. Front Pharmacol. 2023;14.
Liu M, Shaver CM, Birdwell KA, Heeney SA, Shaffer CM, Van Driest SL. Composite CYP3A phenotypes influence tacrolimus dose-adjusted concentration in lung transplant recipients. Pharmacogenet Genomics. 2022;32:209–17.
Concha J, Sangüesa E, Ribate MP, García CB. CYP3A4*1B but Not CYP3A5*3 as Determinant of Long-Term Tacrolimus Dose Requirements in Spanish Solid Organ Transplant Patients. Int J Mol Sci. 2024;25:11327.
Tsironi A, Lazaros K, Mendrinou E, Papasotiriou M, Siamoglou S, Kydonopoulou K, et al. Impact of CYP3A4 and ABCB1 genetic variants on tacrolimus dosing in Greek kidney transplant recipients. Front Pharmacol. 2025;16.
Aouam K, Kolsi A, Kerkeni E, Ben Fredj N, Chaabane A, Monastiri K, et al. Influence of Combined CYP3A4 and CYP3A5 Single-Nucleotide Polymorphisms on Tacrolimus Exposure in Kidney Transplant Recipients: A Study According to the Post-Transplant Phase. Pharmacogenomics. 2015;16:2045–54.
Acknowledgements
We would like to thank all the patients who consented to be involved in the study.
Author information
Authors and Affiliations
Contributions
Conceptualization: A.T., G.P.P.; Methodology: A.T., E.M., M.P., G.P.P.; Formal Analysis: A.T., E.M., S.S., A.J., S.G, K.K., A.G.; Resources: B.R.A., M.P., G.P.P.; Writing—Original Draft Preparation: A.T.; Writing—Review and Editing: all authors; Supervision: M.P., G.P.P.; Study Coordination, G.P.P.; All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Ethics statement
The studies involving human participants were reviewed and approved by the Ethics Committee of the University of Patras (17/03/2017). The patients/participants provided their written informed consent to participate in this study. Αll methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tsironi, A., Mendrinou, E., Siamoglou, S. et al. Combined Role of CYP3A4 and CYP3A5 genetic variants in tacrolimus dose-adjusted trough levels: a clinical retrospective study in kidney transplant patients. Pharmacogenomics J 26, 1 (2026). https://doi.org/10.1038/s41397-025-00395-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41397-025-00395-w