Abstract
Cognitive deficits are a hallmark of Alzheimer’s disease (AD), and effective treatments remain elusive. Transcranial alternating current stimulation (tACS), a non-invasive technique, has shown potential in improving cognitive function across various populations, but further research is needed to investigate its efficacy in AD. In a randomized, double-blind, sham-controlled pilot trial, 36 mild AD patients received active or sham theta-tACS (8 Hz, 1.6 mA, 20-min daily) during n-back task for two weeks, followed by a 10-week follow-up. Cognitive assessments and resting-state EEG were analyzed at baseline, after-treatment, and follow-up. The results showed that the active group demonstrated significant cognitive improvements after treatment (MMSE: t (15) =-3.273, p = 0.005, Cohen’s d = 0.82), particularly in short-term memory (MMSE-recall: Z = -2.11, p = 0.035, r = 0.53), with maintained benefits after 10 weeks. In contrast, the sham group exhibited long-term cognitive decline (MMSE: t (4) = 3.586, p = 0.023, Cohen’s d = -1.60). EEG analysis revealed reduced gamma power (t (23) = 2.689, p = 0.013, Cohen’s d = 1.077) and theta connectivity in active group, particularly in the frontotemporal regions (F4/F7: t (23) = 2.467, p = 0.021, Cohen’s d = 0.988; F4/T3: t (23) = 2.465, p = 0.022, Cohen’s d = 0.987), which was correlated with cognitive improvements (R = –0.57, p = 0.043). In conclusion, tACS combining cognitive training may offer cognitive benefits in mild AD by modulating neural activity, though further studies are needed to clarify its mechanisms.
Similar content being viewed by others
Introduction
Alzheimer’s disease (AD), the leading cause of dementia, is marked by progressive cognitive decline, neuropsychiatric symptoms, and impaired daily functioning [1]. With the global prevalence of dementia projected to exceed 150 million by 2050 [2], the demand for more effective interventions is urgent. Currently, there is no cure for AD, and while pharmaceutical treatments remain the primary approach for managing symptoms, their impact on cognitive function is limited and often accompanied by undesirable long-term side effects [1].
In light of these challenges, non-pharmaceutical approaches targeting cognitive function are gaining increased attention. Cognitive training (CT) has emerged as a promising method for enhancing neural processing, particularly in working memory tasks like the adaptive n-back task [3, 4]. In parallel, non-invasive brain stimulation (NIBS) has attracted great interest for its safety and precision in modulating neural circuits [5]. Specifically, transcranial alternating current stimulation (tACS) synchronizes externally applied oscillatory currents with the brain’s natural rhythms through neural entrainment [6]. This process recruits neurons into coordinated networks, leading to alterations in brain dynamics—such as amplitude [7], frequency [8], and connectivity [9]—which enhance cortical plasticity and improve cognitive function. tACS has shown cognitive benefits across various populations, including healthy individuals and those with neuropsychiatric conditions such as schizophrenia, major depressive disorder and mild cognitive impairment [10]. Notably, researchers have hypothesized that combining cognitive training (CT) with transcranial electrical stimulation (tES) could further amplify cognitive gains by pairing ‘exogenous’ neuromodulation with ‘endogenous’ neural activation [11]. However, despite promising initial findings [12,13,14], large-scale, randomized clinical trials with appropriate control groups are missing to fully confirm these effects.
The choice of stimulation frequency is key for targeting specific cognitive functions. Beta (13–29 Hz) band tACS has been linked to improvements in short-term memory [15], while gamma-tACS (30–140 Hz) is associated with fluid intelligence [16]. The frontal, parietal, and occipital cortices are critical in working memory processing [17], with brain oscillations—particularly in the theta (4–8 Hz) and gamma bands—playing distinct roles. Theta oscillations are believed to be essential for the temporal organization of memory items, while gamma oscillations aid in their maintenance [18, 19]. Growing evidence supports the idea that theta-tACS enhances working memory performance in healthy individuals [20, 21]. Specifically, our recent study has linked 8 Hz tACS over the prefrontal region to improved accuracy in verbal working memory tasks, with the strongest dependency observed between 8 Hz power in the prefrontal cortex and n-back task performance, compared to other frequency-performance pairs [22]. However, all these findings are based on healthy individuals, while evidence in AD cohorts remains limited. Additionally, repeated tACS sessions were associated with strengthened neuroplastic changes, leading to prolonged therapeutic benefits [23].
Building on these foundations, we conducted a double-blind, randomized controlled pilot trial to explore the therapeutic potential of theta (θ) band tACS combined with cognitive training (CT) in mild Alzheimer’s disease (AD) and its electrophysiological effects. Participants received repeated 8 Hz θ-tACS sessions targeting the bilateral prefrontal cortex, paired with the n-back task, with cognitive function assessed via the Mini-Mental State Examination (MMSE) to track disease progression.
Materials and methods
Study participants
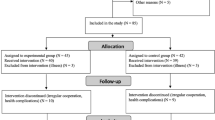
This study was approved by the Ethics Committee of Sir Run Run Shaw Hospital, Zhejiang University School of Medicine (No: 20170228-1) and has been registered on the Chinese Clinical Trial Registry (ChiCTR2400093929). All methods were performed in accordance with the relevant guidelines and regulations. A total of 51 patients with mild AD were recruited from the hospital’s inpatient and outpatient departments between 2020 and 2022. Of these, 6 did not meet the inclusion criteria, 4 declined participation, and 5 withdrew participant owing to COVID-19 pandemic-related disruptions. Ultimately, 36 participants were enrolled in this double-blind, randomized, sham-controlled pilot study and completed baseline assessments (T0). All participants approved written informed consent before enrollment. The COVID-19 pandemic introduced additional challenges, as isolation measures impacted participant retention. As shown in Fig. S1, four patients (active/sham: 2/2) withdrew during treatment and missed the post-treatment visit (T1), while 19 (active/sham: 10/9) were lost to follow-up at 10 weeks (T2).
Inclusion and exclusion criteria
All patients were diagnosed with probable AD based on the criteria outlined in the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) at the inpatient and outpatient departments of Sir Run Run Shaw Hospital (Hangzhou, China). Inclusion criteria were as follows: (1) structural coronal MRI evidence of hippocampal atrophy, defined by a Scheltens’s scale score of ≥2; (2) a Clinical Dementia Rating (CDR) score of 0.5 to 1; (3) age between 55 and 85 years; and (4) right-handedness. Exclusion criteria included: (1) severe cardiac, pulmonary, hepatic, or renal disease; (2) a history of significant cranial trauma, neurological disorders, or psychiatric illness; (3) focal brain lesions on T1 or T2 imaging; or (4) contraindications for tACS, such as the presence of medical implants, electronic devices, or metallic elements in the body.
Study design and blinding procedure
Enrolled patients were randomly assigned to either the active transcranial Alternating Current Stimulation (tACS) group or the sham group in a 1:1 ratio. The randomization sequence was generated using a computer-based randomization algorithm by an independent biostatistician who was not involved in participant recruitment, enrollment, or data collection. To maintain rigorous allocation concealment, the sequence was securely stored in sequentially numbered, opaque, sealed envelopes prepared by a study coordinator with no involvement in participant recruitment or outcome assessment. Each envelope was opened only after the participant had completed baseline evaluations and formally met all inclusion criteria. This procedure ensured that both participants and investigators responsible for recruitment and clinical assessments remained blind to group assignment until after randomization, thereby minimizing selection bias and ensuring the integrity of the allocation process. Throughout the entire treatment and follow-up periods, both participants and outcome assessors remained blinded to group allocation; only the interventionists administering the tACS were aware of the assignment, and they were not involved in any evaluations, follow-up procedures, or data analyses.
Following randomization, all participants underwent 10 sessions of either active or sham tACS while performing the n-back task, with each session lasting 20 min. Sessions were administered five days per week over two weeks (Fig. 1A). Cognitive function, neuropsychiatric symptoms, and daily living activities were evaluated, along with neuroelectrophysiological activity, which was assessed through resting-state electroencephalography (rsEEG) at three key time points (shown in Fig. 1A and Fig. S1).
This double-blind, randomized, sham-controlled study was designed following the pipeline diagram (A). Participants in the active group underwent a daily 20-minute intervention over two weeks, which combined 8 Hz theta-tACS (B: stimulation sites indicated in orange) with an n-back working memory task (C). In the experiment, the adaptive n-back task started with 1-back and dynamically adjusted difficulty based on performance (see Methods). Panels A and C were created using BioRender.com. The figures in Panel B were adapted from SimNIBS software and a Wikimedia source (https://commons.wikimedia.org/wiki/File:21_electrodes_of_International_10-20_system_for_EEG.svg), licensed under CC BY-SA 4.0. Timepoints: baseline (T0), after-treatment (T1), and 10-week follow-up (T2).
Cognitive training
Participants were trained on an n-back task, a widely used experimental working memory training paradigm, where they needed to remember a series of stimuli, such as images or characters, presented on the screen. Their task was to determine whether the latest stimulus matched the one presented N items earlier in the sequence (Fig. 1C). The task was tailored to each participant’s performance, starting at an n-back level of 1, with a maximum level of 2. If the participant’s accuracy exceeded 95% on a given trial, the n-back level was increased to 2. If accuracy remained between 75% and 95%, the level was maintained, and if accuracy dropped below 75%, the level was decreased.
To assess the effectiveness of this protocol, we analyzed participants’ decision accuracy and average reaction times, which were recorded daily throughout the 2-week treatment period. A one-way repeated measures ANOVA, with treatment date as a repeated factor, showed no significant differences between the two groups in accuracy (F (1,18) = 2.218, p = 0.154) or average reaction times (F (1,18) = 2.443, p = 0.136). Within-group comparisons revealed a significant improvement in accuracy (F (1.841,33.14) = 11.24, p < 0.001) and a significant reduction in average reaction times (F (1.743,31.38) = 13.40, p < 0.001) over the course of the treatment (Fig. S2).
tACS protocol
For the electrical stimulation, we used a tACS Stimulator (N-ET-001, Shenzhen Zhongkehuayi Technology Co., Ltd., China; website: www.neuraplus.cn) with the control software NeuroPlus v1.0. Each patient had two 4.7 cm×9.0 cm electrode patches placed on both sides of the forehead at the FP1-AF7 and FP2-AF8 sites according to the EEG 10-20 system (Fig. 1B). The stimulation was administered using an alternating current with a frequency of 8 Hz (theta-band) and an amplitude of 1.6 mA, for a duration of 20 min.
In the sham condition, the current was ramped up and down over 20 s at the beginning and end of the 20-minute session. The current intensity was set to 1.6 mA, with a frequency of 8 Hz, but no continuous stimulation was applied during the session. This allowed participants to feel the initial tingling of tACS without affecting neuronal excitability.
Participant characteristics and clinical outcome measures
We collected demographic information, including age, gender, years of education, APOE genotype, and medication status, prior to the intervention. Then cognitive function, neuropsychiatric status, and daily functional activities were assessed at three time points respectively: before treatment (T0), immediately after 2-week treatment (T1), and at a 10-week follow-up (T2).
The primary outcome was assessed using the Chinese version of the Mini Mental State Examination (MMSE) [24], a widely used tool for evaluating global cognitive function (see Supplementary Material). Secondary outcomes included: (i) the Screening Scale for Mild Cognitive Impairment (sMCI) [25] for detecting mild cognitive impairment; (ii) the 12-Word Philadelphia Verbal Learning Test (PVLT) specializing in memory domain evaluation; (iii) neurobehavioral status and dementia-related symptoms evaluated through the Clinical Dementia Rating (CDR), the Neuropsychiatric Inventory (NPI), and the Patient Health Questionnaire-9 (PHQ-9); and (iv) functional independence in daily activities measured by the Lawton-Brody Activities of Daily Living (ADL) scale.
The sMCI is specifically tailored for the Chinese population, integrating elements from well-established international assessments like the MMSE and Montreal Cognitive Assessment (MoCA). It examines key cognitive domains—including Orientation, Attention, Language, Recall, and Abstract Reasoning—while thoughtfully considering cultural and linguistic nuances unique to Chinese individuals [25]. To minimize bias, MMSE scores were categorized into four levels of severity according to the Chinese Longitudinal Healthy Longevity Survey [26]: Level 1 (0–9), Level 2 (10–17), Level 3 (18–24), and Level 4 (25–30).
EEG data collection and preprocessing
Resting-state electroencephalography (rsEEG) data were collected to assess neuroelectrophysiological activity at three time points: baseline (prior to intervention), immediately following the final tACS session, and at the 10-week follow-up. Participants were seated comfortably in a sound-attenuated, dimly lit room, minimizing external stimuli during the recordings. The EEG data were recorded in the Sir Run Run Shaw Hospital EEG room by a 16-channel Nihon Kohden device (EEG-1200C, Nihon Kohden, Japan) at a sampling frequency of 128 Hz. The EEG collection process is from closed-eye recording until the end, which takes around 5 min. To obtain resting-state EEG data and assure the data quality, we selected the EEG recording from 75 s before the end to 15 s before the end as the samples to avoid the interference caused by signals of electromyography and electrooculogram at the beginning of the subject’s EEG acquisition.
We used Automagic, an open-source MATLAB toolbox [27], to standardize preprocessing of EEG data. Automagic has proven to be an effective method to reduce a large extent of EEG by applying a pipeline of algorithms to identify artifactual channels in combination with multiple artifact rejection algorithms. All EEG data were preprocessed using the same procedure, ensuring consistency across subjects. Poor-quality recordings (two in the sham group and three in the active group) were objectively identified and excluded using predefined thresholds.
Statistical analysis
All statistical analyses were performed using R version 4.4.0 [28]. The following R packages were utilized: tidyverse [29] for data manipulation and visualization, rstatix [30] for statistical tests, and ggplot2 [31] for figure creation. Descriptive statistics summarise demographic and baseline characteristics, presented as means and standard deviations for continuous variables, and counts with percentages for categorical variables.
To compare baseline characteristics between the active and sham groups, unpaired Student’s t-tests were used for normally distributed continuous variables, and the Mann-Whitney U test for non-normally distributed data. Categorical variables were compared using the chi-square test (χ²). Within-group differences before and after treatment were assessed using paired t-tests for normally distributed data or Wilcoxon signed-rank tests for non-normal data. For the n-back task performance analysis, one-way repeated measures ANOVA were employed to evaluate training effects within each group across the intervention period. Spearman’s rank correlation coefficient was employed to examine the relationship between electrophysiological changes and cognitive improvements. All statistical tests were two-tailed, and a p-value less than 0.05 was considered statistically significant.
EEG analysis
EEG data were analyzed with the Fieldtrip MATLAB toolbox [32]. Sixteen channels in the EEG datasets are selected, including FP1, FP2, F3, F4, C3, C4, P3, P4, O1, O2, F7, F8, T3, T4, T5, and T6. The electrode distribution for all channels is set according to the international 10-20 system. The continuous EEG data were segmented into 1-second epochs with 50% overlap and power spectral density was then computed using the multi-taper method with a Hanning window, focusing on the 1-40 Hz range. Power values were subsequently log-transformed (log10) for normalization and statistical analysis. The imaginary part of coherency, which minimizes the effect of spurious connectivity arising from volume conduction [33], was used to compute the all-to-all functional connectivity between electrodes in all frequency band.
For power spectrum statistics, nonparametric cluster permutation tests [34] were performed to statistically evaluate group differences between the active and sham groups at baseline phase T0 and immediate after treatment phase T1. Two-sample independent t-tests were used as test statistics to examine differences between the active and sham groups across different time points or between the pre- and post-treatment values between the active group and the sham group, with a predefined threshold of p < 0.05. Two-sample paired t-tests were used as test statistics to examine differences in each group before and after treatment (T0 vs T1), with a predefined threshold of p < 0.05. The sum of the t values within a given cluster was defined as the cluster-level statistic. To obtain the reference cluster distribution, group labels were randomly shuffled 1000 times. For each shuffling, the cluster with maximum t values were used to create reference distribution. Observed cluster statistic values exceeding the 95th percentile of the reference cluster distribution was considered as significant cluster.
For functional connectivity statistics, the network-based statistic (NBS) [35] was conducted to test the network statistical significance while controlling for multiple comparison corrections. When comparing the functional connectivity network strength difference between the active and sham groups across different time points or between the pre- and post-treatment values between the active group and the sham group, an independent two-sample t-test was performed on the connectivity value. When comparing the difference in each group before and after treatment (T0 vs T1), a paired two-sample t-test was performed on the connectivity value. Connections exceeding the predefined threshold (p < 0.05) constituted a set of suprathreshold links. Then, connecting graph components formed topological clusters, and the number of connections was defined as a cluster score. The maximum number of connections across clusters was used as the test statistic. By randomizing the data across groups and recalculating the test statistic 5000 times, a reference distribution of maximum cluster values was obtained to evaluate the statistic of the observed cluster scores. Observed cluster scores higher than the 97.5th percentile or lower than 2.5th percentile were considered significant at p < 0.05 level. These statistics were performed with the open source NBS toolbox (NITRC: Network-Based Statistic (NBS): Tool/Resource Info), and brain network visualization was done by using the python toolkit matplotlib [36].
Results
We assessed both groups using multi-dimensional assessments, including neuropsychological tests and resting-state EEG (rsEEG) at baseline (T0), immediately post-treatment (T1), and at the 10-week follow-up (T2), as shown in Fig. 1 and Fig. S1. A total of 36 mild AD patients completed the baseline assessment, with demographic details in Tables 1 and 2. The mean age in the active and sham groups were no statistically significant differences (68.22 vs. 69.72, p = 0.57), with similar gender distribution across both. The average years of education in the active group (5.06 ± 4.52) were also comparable to those in the sham group (6.16 ± 5.28). Clinical Dementia Rating (CDR) scores were 0.78 ± 0.26 for the active group and 0.86 ± 0.23 for the sham group, with no statistically significant differences between the two groups (p = 0.30). The two groups were also similar in APOE ε4 carrier status, medication use, and baseline cognitive assessments.
Primary and secondary outcome measures
To evaluate the effect of this combined intervention on general cognition, we compared baseline MMSE scores with post-treatment and 10-week follow-up results for each group. In the active group, significant improvements were seen after treatment (T1 vs. T0: t (15) =-3.273, p = 0.005, Cohen’s d = 0.82), while no such gains were observed in the sham group (Fig. 2 and Fig. S3). By the 10-week follow-up, the sham group showed a significant decline in MMSE scores (T2 vs. T0: t (4) = 3.586, p = 0.023, Cohen’s d = -1.60), whereas the active group maintained their cognitive performance (Fig. 2). The secondary outcome of sMCI aligned directionally with MMSE improvements. It was the active group, not the sham group, that showed significant post-treatment improvement from baseline (T1 vs. T0: t (13) =-3.296, p = 0.005, Cohen’s d = 0.82). In contrast, at the 10-week follow-up, the sham group exhibited a marginal decline in sMCI scores relative to baseline (T2 vs. T0: p = 0.077) (Fig. 2).
Participants number (Active/Sham): T0 = 18/18, T1 = 16 (PVLT: 15)/14, T2 = 6/5; Dots and lines show paired values of individual participants; all statistic differences were conducted using paired t-test (* p < 0.05, ** p < 0.01, ns means no significance). MMSE Mini-Mental State Examination, sMCI Screening Scale for Mild Cognitive Impairment, PVLT 12-Word Philadelphia Verbal Learning Test, PHQ-9 Patient Health Questionnaire-9, ADL Activities of Daily Living, CBI Caregiver Burden Inventory.
We further examined specific cognitive domains using subscales from the MMSE and sMCI, as well as the PVLT, a test for verbal learning and memory (Fig. 2, Table 2). The active group showed significant improvements in short-term memory, reflected by gains in MMSE-recall (T1 vs. T0: Z = -2.11, p = 0.035, r = 0.53) and PVLT-total learning (T1 vs. T0: t (14) = -2.475, p = 0.027, Cohen’s d = 0.64) and short free recall scores (T1 vs. T0: Z = -2.536, p = 0.011, r = 0.65). Notable improvements were also observed in working memory (MMSE-registration (T1 vs. T0: p = 0.063), PVLT-distractor trial (T2 vs. T0: t (5) =-2.712, p = 0.042, Cohen’s d = 1.107)) and long-term memory (PVLT-long cued recall (T1 vs. T0: p = 0.055)). In contrast, the sham group exhibited only minor improvements in long-term memory, with slight increases in PVLT-long free recall (T1 vs. T0: p = 0.104) and long cued recall scores (T1 vs. T0: p = 0.082). Beyond cognitive assessments, we also evaluated depression, activities of daily living, caregiver burden, and neuropsychiatric symptoms at each time point (Fig. 2 and Fig. S4). No significant changes or trends were observed in these secondary outcomes in either group.
Gamma power analysis
Resting-state EEG (rsEEG) data were collected before and after the intervention to evaluate the effects of tACS combined with the n-back task on brain oscillatory dynamics. A cluster-based permutation test identified significant group differences in gamma power, particularly within the 32–40 Hz range. As illustrated in Fig. 3A, the active group exhibited a marked reduction in gamma power post-treatment, particularly in frontal and temporal regions (F7, F4, C3, T3, P4), while the sham group showed minimal changes over the same period. This divergence is further highlighted in the T1–T0 difference maps and topographic representation (Fig. 3B), both of which underscore the more pronounced decline in gamma power observed in the active group. Furthermore, the box plot reveals a statistically significant reduction in gamma power in the active group compared to the sham group (t (23) = 2.689, p = 0.013, Cohen’s d = 1.077), underscoring the impact of tACS on gamma oscillatory activity in this cohort.
(A) Spectral topographies of baseline (T0), after-treatment (T1), and the difference before and after treatment (T1–T0) in the active (N = 13) and sham (N = 12) groups. (B) Gamma power differences (Post–Pre intervention) between the active and sham groups. Left panel: significant channels and frequency bins identified by cluster permutation t-test between two groups. The significant channels and frequency bins were highlighted. Middle panel: Spectral topography of group differences in gamma power with t values assessed by independent t-test between active and sham groups. Marked sensors belong to significant clusters identified by the cluster permutation t-test (p < 0.05). Right panel: Mean gamma power differences over the marked sensors compared by independent t-test between two groups. * p < 0.05. Timepoints: baseline (T0) and after-treatment (T1).
Cognitive impact of theta connectivity patterns
Although we explored functional connectivity in all frequency bands, only a significant theta-band network was identified using network-based statistics (NBS) (Fig. 4A), primarily localized in the frontal, temporal, and occipital regions, corresponding to the previously noted gamma power cluster. We further examined changes in connectivity strength between specific brain regions and their relationship to cognitive outcomes, as measured by changes in MMSE scores. In the active group, there were significant reductions in connectivity strength (Fig. 4B). The most notable decreases were observed in the bilateral frontal regions (F4/Fp1: t (23) = 2.884, p = 0.008, Cohen’s d = 1.154; F4/F7: t (23) = 2.467, p = 0.021, Cohen’s d = 0.988) and between the right frontal and left temporal regions (F4/T3: t (23) = 2.465, p = 0.022, Cohen’s d = 0.987; F8/T3: t (23) = 3.070, p = 0.005, Cohen’s d = 1.229) (Fig. 4B and Fig. S5). To link these treatment effects with clinical outcomes, we explored the relationship between changes in cognitive performance and functional connectivity. In the active group, a significant negative correlation was found between F4/F7 connectivity strength and MMSE improvement (R = –0.57, p = 0.043), with similar negative trends observed in other connections, though not statistically significant (Fig. 4C). This suggests that reduced connectivity in these regions may be associated with cognitive improvement. In contrast, few similar correlations were observed in the sham group.
Electrode positions (A) showing significant post-pre connectivity differences between the active and sham groups were indicated with dotted lines, as determined by the Network-Based Statistic (NBS). (B) Changes in connectivity strengths between two groups were compared by independent t-test. All electrode position pairs in the above plot are non-directional. (C): Correlations between changes in MMSE levels and connectivity strengths were analyzed using Spearman’s rank test (Orange: Active group; Blue: Sham group). All Δ above represents differences between after-treatment and baseline (Δ = T1 – T0). * p < 0.05, ** p < 0.01. Timepoints: baseline (T0) and after-treatment (T1).
Discussion
This study demonstrates that combining conventional prefrontal theta-band tACS with cognitive training (n-back task) significantly modulates neural connectivity (in the theta and gamma band) and improves cognitive deficits in individuals with mild Alzheimer’s disease (AD). Our primary and secondary outcomes both reveal notable improvements in short-term and working memory, which were sustained over a 10-week follow-up. These results suggest that this approach may serve as a promising non-invasive intervention to slow cognitive decline in AD.
Multiple cognitive assessment tools were employed to evaluate the impact of tACS combined with cognitive training in mild AD patients. In addition to the widely used MMSE [24], we also applied the Screening Scale for Mild Cognitive Impairment (sMCI) [25], which has proven more accurate than the MoCA in elderly Chinese populations. Although sMCI has not been extensively tested in AD, our results suggest that it is effective in monitoring therapeutic responses in elderly Chinese patients with mild AD and may offer a valuable tool for capturing nuanced cognitive changes during interventions.
Our findings, based on assessments using the MMSE, sMCI, and memory-specific PVLT, provide compelling evidence that combined intervention primarily enhanced general cognition and short-term memory. Furthermore, while short-term memory exhibited the most significant improvement, we also observed positive trends in working memory, long-term memory, and language abilities. Critically, these cognitive gains occurred alongside preserved functional independence (stable ADL scores), a key determinant of quality of life in early AD. These results align with prior researches, which has consistently identified memory enhancement as one of the most frequent outcomes of multi-session tACS interventions [37, 38]. Though improvements in general cognition were noted, the literature presents mixed findings regarding tACS’s broader impact on cognitive functions [37, 39, 40]. Some studies have reported significant gains in global cognition, while others have found no measurable effect [38]. These discrepancies likely stem from variations in study design, stimulation protocols, and the specific cognitive tasks employed.
While trends of improvement were observed across several cognitive domains in our study, attention and executive functions did not show significant gains. Previous research has shown that single-session tACS, particularly when applied to the frontal cortex (F3/F4), can improve working memory, visual attention, and processing speed, especially in non-AD populations [41]. The absence of such changes in our study could be due to the cumulative nature of multi-session tACS or the specific cognitive profile of AD patients, where memory deficits are more prominent.
As another pivotal neurophysiological marker in AD clinical trials, we observed distinct changes in gamma-band power and theta-band connectivity in resting-state EEG measures, both consistently localized in the frontal, temporal, and occipital regions. This finding reflects an important aspect of neural dynamics: while different frequency bands represent distinct neural processes, they can overlap in the brain regions involved. Gamma oscillations are typically associated with local neural processing and higher-order cognitive functions, such as attention and the integration of long-term memory [18, 19, 42]. In contrast, theta oscillations are linked with long-range communication and coordination between brain regions, particularly for tasks involving working memory and cognitive control [18, 19, 42]. The frontal, temporal, and occipital cortices serve as hubs for both local and network-level neural processes [43]. The observed reduction in both gamma power and theta connectivity within these regions post-treatment suggests they are central to cognitive processing, where local computations (gamma) and broader network integration (theta) occur simultaneously. The combined effect of tACS and CT likely helps recalibrate both local gamma oscillations and long-range theta connectivity, potentially improving cognitive functions such as working memory and memory consolidation, with possible benefits for long-term memory. The interaction between local processing and global network coordination suggests that gamma oscillations may be nested within theta rhythms during complex tasks, with theta modulating gamma activity across wider networks. This corresponds with earlier findings that hippocampal gamma power is modulated by the theta phase during working memory retention [44], which may explain the observed changes in both frequency bands within the same regions. tACS could be influencing these interactions. Further evidence is needed to confirm their role in cognitive improvements.
The increase in both gamma power and theta connectivity observed in the sham group likely reflects the brain’s attempt to compensate for cognitive decline. In early-stage AD, heightened neural activity may help preserve function despite synaptic loss, but this overactivation is often inefficient and can contribute to further decline as the disease progresses [45, 46]. This is consistent with previous studies, one of which has found that patients with mild AD show greater theta synchrony than those with MCI [47], suggesting that this increase may serve as a transient compensatory mechanism—acting as a load-shifting process in response to more severe cognitive deficits [48]. Additionally, similar “overload” connectivity has been observed not only in Alzheimer’s disease but also in psychiatric disorders such as schizophrenia, obsessive-compulsive disorder (OCD), and bipolar disorder [49,50,51]. Take together, these findings suggest that pathological hyperconnectivity may represent one of the general mechanisms for coping with neurological dysfunctions.
In contrast, the active group exhibited a significant reduction in maladaptive processes following combined tACS and cognitive training. Additionally, the negative correlation we found between MMSE scores and theta connectivity further suggests that reduced connectivity is linked to better cognitive outcomes, supporting the potential role of tACS in modulating brain activity. We attribute these effects to two synergistic mechanisms: (i) frequency-specific entrainment of endogenous oscillations, with maximal efficacy when stimulation aligns with the intrinsic rhythms of target networks, and (ii) spike-timing-dependent plasticity (STDP), facilitating lasting reorganization of neural circuits [6]. Together, these processes enhance phase-locked neural communication and promote more efficient network dynamics, leading to cognitive improvement.
Recent findings may further contextualize the sustained effects of our 8 Hz protocol. Specifically, stimulation over the medial prefrontal cortex (Fp1/Fp2) at 5 Hz selectively modulates hippocampal oscillations during stimulation, while 10 Hz induces post-stimulation power changes [52]. This frequency-dependent spatiotemporal specificity suggests that 8 Hz may strike a balance—engaging both immediate entrainment and longer-term plasticity. Nonetheless, parametric studies with finely tuned stimulation parameters are needed to validate this hypothesis. Last but not least, the observed negative correlation between MMSE scores and theta-band connectivity supports the role of tACS in modulating pathological network activity, with reduced connectivity linked to better cognitive outcomes.
While our findings of this pilot study are encouraging, several limitations should be considered. First, all analyses were conducted using complete cases. Comparative analyses showed that age, gender, and disease severity did not differ significantly between completers and dropouts, indicating that although some attrition bias may exist, its impact was limited. However, future studies with larger cohorts and extended follow-up periods are needed to better understand the long-term effects of this intervention. Second, the precise mechanisms underlying the observed reductions in connectivity strength remain incompletely understood. Incorporating advanced neuroimaging techniques, such as multimodal MRI, in future research could provide further insights into the structural and functional changes associated with tACS.
Overall, this study offers valuable insights into the potential of theta-tACS combined with cognitive training to enhance cognitive performance and modulate neural connectivity in individuals with mild AD. By targeting frontal-temporal networks, tACS presents a novel, non-invasive approach to mitigating cognitive decline via normalization of pathological hyperconnectivity. These findings underscore the need for further research to assess the long-term efficacy, optimize stimulation parameters, and fully understand the neural mechanisms behind these benefits, potentially advancing treatment strategies for neurodegenerative diseases.
Data availability
The data that support the findings of this study are not publicly available due to privacy restrictions but are available from the corresponding author upon reasonable request.
References
Scheltens P, Blennow K, Breteler MM, de Strooper B, Frisoni GB, Salloway S, et al. Alzheimer’s disease. Lancet. 2016;388:505–17.
Collaborators GBDDF. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health.. 2022;7:e105–25.
Soveri A, Antfolk J, Karlsson L, Salo B, Laine M. Working memory training revisited: A multi-level meta-analysis of n-back training studies. Psychon Bull Rev. 2017;24:1077–96.
Sitzer DI, Twamley EW, Jeste DV. Cognitive training in Alzheimer’s disease: a meta-analysis of the literature. Acta Psychiatr Scand. 2006;114:75–90.
Gu L, Xu H, Qian F. Effects of non-invasive brain stimulation on alzheimer’s disease. The journal of prevention of Alzheimer’s disease. 2022;9:410–24.
Vogeti S, Boetzel C, Herrmann CS. Entrainment and spike-timing dependent plasticity - a review of proposed mechanisms of transcranial alternating current stimulation. Front Syst Neurosci. 2022;16:827353 https://doi.org/10.3389/fnsys.2022.827353.
Kasten FH, Herrmann CS. Transcranial alternating current stimulation (tACS) enhances mental rotation performance during and after stimulation. Front Hum Neurosci. 2017;11:2 https://doi.org/10.3389/fnhum.2017.00002.
Ahn S, Mellin JM, Alagapan S, Alexander ML, Gilmore JH, Jarskog LF, et al. Targeting reduced neural oscillations in patients with schizophrenia by transcranial alternating current stimulation. Neuroimage. 2019;186:126–36.
Jones KT, Johnson EL, Gazzaley A, Zanto TP. Structural and functional network mechanisms of rescuing cognitive control in aging. Neuroimage. 2022;262:119547 https://doi.org/10.1016/j.neuroimage.2022.119547.
Grover S, Fayzullina R, Bullard BM, Levina V, Reinhart RMG. A meta-analysis suggests that tACS improves cognition in healthy, aging, and psychiatric populations. Sci Transl Med. 2023;15:eabo2044 https://doi.org/10.1126/scitranslmed.abo2044.
Elmasry J, Loo C, Martin D. A systematic review of transcranial electrical stimulation combined with cognitive training. Restor Neurol Neurosci. 2015;33:263–78.
Moussavi Z, Kimura K, Kehler L, de Oliveira Francisco C, Lithgow B. A novel program to improve cognitive function in individuals with dementia using transcranial alternating current stimulation (tACS) and tutored cognitive exercises. Front Aging. 2021;2:632545 https://doi.org/10.3389/fragi.2021.632545.
Varastegan S, Kazemi R, Rostami R, Khomami S, Zandbagleh A, Hadipour AL. Remember NIBS? tACS improves memory performance in elders with subjective memory complaints. GeroScience. 2023;45:851–69.
Leite J, Goncalves OF, Carvalho S. Speed of processing (SoP) training plus alpha-tACS in people with mild cognitive impairment: a double blind, parallel, placebo controlled trial study protocol. Front Aging Neurosci. 2022;14:880510.
Feurra M, Galli G, Pavone EF, Rossi A, Rossi S. Frequency-specific insight into short-term memory capacity. J Neurophysiol. 2016;116:153–8.
Santarnecchi E, Polizzotto NR, Godone M, Giovannelli F, Feurra M, Matzen L, et al. Frequency-dependent enhancement of fluid intelligence induced by transcranial oscillatory potentials. Current biology : CB. 2013;23:1449–53.
Violante IR, Li LM, Carmichael DW, Lorenz R, Leech R, Hampshire A, et al. Externally induced frontoparietal synchronization modulates network dynamics and enhances working memory performance. eLife. 2017;2017:6 https://doi.org/10.7554/eLife.22001.
Bahramisharif A, Jensen O, Jacobs J, Lisman J. Serial representation of items during working memory maintenance at letter-selective cortical sites. PLoS Biol. 2018;16:e2003805 https://doi.org/10.1371/journal.pbio.2003805.
Ursino M, Cesaretti N, Pirazzini G. A model of working memory for encoding multiple items and ordered sequences exploiting the theta-gamma code. Cogn Neurodyn. 2023;17:489–521.
Vosskuhl J, Huster RJ, Herrmann CS. Increase in short-term memory capacity induced by down-regulating individual theta frequency via transcranial alternating current stimulation. Front Hum Neurosci. 2015;9:257 https://doi.org/10.3389/fnhum.2015.00257.
Reinhart RMG, Nguyen JA. Working memory revived in older adults by synchronizing rhythmic brain circuits. Nat Neurosci. 2019;22:820–7.
Zeng L, Guo M, Wu R, Luo Y, Wei P. The effects of electroencephalogram feature-based transcranial alternating current stimulation on working memory and electrophysiology. Front Aging Neurosci. 2022;14:828377 https://doi.org/10.3389/fnagi.2022.828377.
Antal A, Paulus W. Transcranial alternating current stimulation (tACS). Front Hum Neurosci. 2013;7:317.
O’Connor DW, Pollitt PA, Hyde JB, Fellows JL, Miller ND, Brook CP, et al. The reliability and validity of the Mini-Mental State in a British community survey. J Psychiatr Res. 1989;23:87–96.
XU LY, WEI LL, ZHANG YC, ZHU ZW, LI YF, et al. Application of screening scale for mild cognitive impairment in screening mild cognitive impairment of the elderly in rural communities in Hangzhou, Zhejiang. Chinese Journal of Contemporary Neurology & Neurosurgery. 2021;21:1057–63.
An R, Liu GG. Cognitive impairment and mortality among the oldest-old Chinese. Int J Geriatr Psychiatry. 2016;31:1345–53.
Pedroni A, Bahreini A, Langer N. Automagic: standardized preprocessing of big EEG data. Neuroimage. 2019;200:460–73.
The R Core Team. R: A Language and Environment for Statistical Computing. 2024. https://www.R-project.org/.
Wickham H, Averick M, Bryan J, Chang W, McGowan L, François R. et al. Welcome to the Tidyverse. J Open Source Softw. 2019;4:1686.
Kassambara A. rstatix: Pipe-Friendly Framework for Basic Statistical Tests. 2023. https://rpkgs.datanovia.com/rstatix/.
Wickham H ggplot2: Elegant Graphics for Data Analysis. 2016. https://ggplot2.tidyverse.org.
Oostenveld R, Fries P, Maris E, Schoffelen JM. FieldTrip: Open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput Intell Neurosci. 2011;2011:156869 https://doi.org/10.1155/2011/156869.
Nolte G, Bai O, Wheaton L, Mari Z, Vorbach S, Hallett M. Identifying true brain interaction from EEG data using the imaginary part of coherency. Clin Neurophysiol. 2004;115:2292–307.
Maris E, Oostenveld R. Nonparametric statistical testing of EEG- and MEG-data. J Neurosci Methods. 2007;164:177–90.
Zalesky A, Fornito A, Bullmore ET. Network-based statistic: identifying differences in brain networks. Neuroimage. 2010;53:1197–207.
S Tosi. Matplotlib for Python developers. Packt Publishing Ltd; 2009.
Kraft JD, Hampstead BM. A systematic review of tACS effects on cognitive functioning in older adults across the healthy to dementia spectrum. Neuropsychol Rev. 2023;34:1165–90.
Manippa V, Palmisano A, Nitsche MA, Filardi M, Vilella D, Logroscino G, et al. Cognitive and Neuropathophysiological Outcomes of Gamma-tACS in Dementia: A Systematic Review. Neuropsychol Rev. 2024;34:338–61.
Sprugnoli G, Munsch F, Cappon D, Paciorek R, Macone J, Connor A. et al. Impact of multisession 40Hz tACS on hippocampal perfusion in patients with Alzheimer’s disease. Alzheimers Res Ther. 2021;13:203.
Klink K, Passmann S, Kasten FH, Peter J. The modulation of cognitive performance with transcranial alternating current stimulation: a systematic review of frequency-specific effects. Brain Sci. 2020;10:932 https://doi.org/10.3390/brainsci10120932.
Kim J, Kim H, Jeong H, Roh D, Kim DH. tACS as a promising therapeutic option for improving cognitive function in mild cognitive impairment: A direct comparison between tACS and tDCS. J Psychiatr Res. 2021;141:248–56.
Al Qasem W, Abubaker M, Kvasnak E. Working memory and transcranial-alternating current stimulation-state of the art: findings, missing, and challenges. Front Psychol. 2022;13:822545 https://doi.org/10.3389/fpsyg.2022.822545.
Oldham S, Fornito A. The development of brain network hubs. Developmental cognitive neuroscience. 2019;36:100607 https://doi.org/10.1016/j.dcn.2018.12.005.
Axmacher N, Henseler MM, Jensen O, Weinreich I, Elger CE, Fell J. Cross-frequency coupling supports multi-item working memory in the human hippocampus. Proc Natl Acad Sci USA. 2010;107:3228–33.
Jones DT, Knopman DS, Gunter JL, Graff-Radford J, Vemuri P, Boeve BF, et al. Cascading network failure across the Alzheimer’s disease spectrum. Brain. 2016;139:547–62.
Gaubert S, Raimondo F, Houot M, Corsi MC, Naccache L, Diego Sitt J, et al. EEG evidence of compensatory mechanisms in preclinical Alzheimer’s disease. Brain. 2019;142:2096–112.
Meghdadi AH, Stevanovic Karic M, McConnell M, Rupp G, Richard C, Hamilton J. et al. Resting state EEG biomarkers of cognitive decline associated with Alzheimer's disease and mild cognitive impairment. PLoS ONE. 2021;16:e0244180.
Gallego-Jutgla E, Sole-Casals J, Vialatte FB, Dauwels J, Cichocki A. A theta-band EEG based index for early diagnosis of Alzheimer’s disease. J Alzheimers Dis. 2015;43:1175–84.
Anticevic A, Hu X, Xiao Y, Hu J, Li F, Bi F, et al. Early-course unmedicated schizophrenia patients exhibit elevated prefrontal connectivity associated with longitudinal change. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2015;35:267–86.
Naze S, Hearne LJ, Roberts JA, Sanz-Leon P, Burgher B, Hall C, et al. Mechanisms of imbalanced frontostriatal functional connectivity in obsessive-compulsive disorder. Brain. 2023;146:1322–7.
Syan SK, Smith M, Frey BN, Remtulla R, Kapczinski F, Hall GBC, et al. Resting-state functional connectivity in individuals with bipolar disorder during clinical remission: a systematic review. J Psychiatry Neurosci. 2018;43:298–316.
Luo H, Ye X, Cai HT, Wang M, Wang Y, Liu Q, et al. Frequency-specific and state-dependent neural responses to brain stimulation. Mol Psychiatry. 2025;30:2880–90. https://doi.org/10.1038/s41380-025-02892-7.
Acknowledgements
We acknowledge the patients and their families, clinical and administrative staff for their important contributions. We also appreciate Shenzhen Zhongkehuayi Technology Company for technical support.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 82101581, 82071181, 82371453), the National Key R&D Program Strategic Science and Technology Innovation Cooperation key special project (SQ2023YFE0201430), STI2030-Major Projects (2022ZD0212400), Key R&D Program of Zhejiang (2024SSYS0017, 2024C03006, 2024C04024, 2025C01119), Fundamental Research Funds for the Central Universities (2025ZFJH01-01). This work was also funded by the Psych-STRATA project (European Union’s Horizon Europe programme, grant No. 101057454).
Author information
Authors and Affiliations
Contributions
Wei C. and Qian G. contributed equally to the conception and design of the study. Kehua Y., Dandan C., Xinhui L., Zhongmei Y., and Dandan L. were responsible for clinical patient management and administering the intervention (equal contribution). Qian G., Daxi F., Lili W., Chanchan S., Yingchun Z., Luoyi X., and Wenjuan C. collected patient data (equal contribution). Shuying R. and Haiteng J. processed and analyzed the EEG data. Xuemin F., Benno P., and Bertram M.-M. provided statistical support and data visualization (equal contribution). Qian G. and Xuemin F. interpreted the data and drafted the manuscript (equal contribution). Daxi F., Benno P., Bertram M.-M. and Pengfei Wei reviewed and made grammatical revisions to the manuscript (equal contribution). Wei C. and Haiteng J. critically revised and gave final approval of the manuscript (equal contribution). All authors read and approved of the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gong, Q., Fu, X., Feng, D. et al. Randomized, double-blind, sham-controlled pilot trial of theta-band transcranial alternating current stimulation during cognitive training in mild Alzheimer’s disease. Transl Psychiatry 16, 57 (2026). https://doi.org/10.1038/s41398-026-03822-z
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-026-03822-z