Abstract
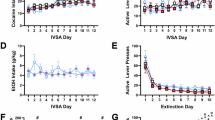
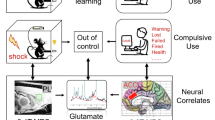
Decision-making impairments are a core symptom of several psychiatric disorders, including gambling and substance use disorders (SUD). These disorders frequently co-occur, suggesting shared neurobiological mechanisms underlying dysfunctional decision-making. We previously demonstrated that chronic cocaine exposure increases risk preference in a rat gambling task (rGT). Given that the prelimbic cortex (PrL) to the nucleus accumbens (NAc) core pathway plays a crucial role in regulating risk-based decision-making, we further explored how chemogenetic modulation of this pathway alters cocaine-induced increase in risky decision-making in the rGT. Notably, activation of Gi, but not Gq, designer receptors exclusively activated by designer drugs (DREADD) in the PrL attenuated the cocaine-induced increase of risk preference in risk-averse rats, while simultaneously reducing cocaine-induced attentional deficits measured by task omissions. Subsequent molecular analyses revealed that cocaine significantly induced changes in the expression levels of calcium channel alpha 1 C subunit (CaV1.2) and in the ratio of phosphorylation at serine 97 of total dopamine- and cAMP-regulated phosphoprotein, 32 kDa (DARPP-32) in the PrL region of these rats, which returned to basal levels with concurrent Gi-DREADD activation. No significant behavioral or molecular changes were observed in risk-seeking rats. These results suggest that modulating the PrL-NAc core pathway can selectively control risk-based decision-making behavior and attentional processes affected by cocaine exposure, offering therapeutic potential for addressing decision-making impairments in dual diagnoses of gambling and SUD.
Similar content being viewed by others
Data availability
The raw data supporting the conclusion of this article will be made available by the authors, without undue reservation. Correspondence and requests for materials should be addressed to Wha Young Kim or Jeong-Hoon Kim.
Code availability
The code used for analysis is available from the corresponding author on reasonable request.
References
Orsini CA, Moorman DE, Young JW, Setlow B, Floresco SB. Neural mechanisms regulating different forms of risk-related decision-making: Insights from animal models. Neurosci Biobehav Rev. 2015;58:147–67.
Chen S, Yang P, Chen T, Su H, Jiang H, Zhao M. Risky decision-making in individuals with substance use disorder: A meta-analysis and meta-regression review. Psychopharmacology. 2020;237:1893–908.
Potenza MN, Balodis IM, Derevensky J, Grant JE, Petry NM, Verdejo-Garcia A, et al. Gambling disorder. Nat Rev Dis Primers. 2019;5:51.
Kim SW, Grant JE, Eckert ED, Faris PL, Hartman BK. Pathological gambling and mood disorders: clinical associations and treatment implications. J Affect Disord. 2006;92:109–16.
Dani JA, Harris RA. Nicotine addiction and comorbidity with alcohol abuse and mental illness. Nat Neurosci. 2005;8:1465–70.
Hasin DS, Stinson FS, Ogburn E, Grant BF. Prevalence, correlates, disability, and comorbidity of DSM-IV alcohol abuse and dependence in the united states: results from the national epidemiologic survey on alcohol and related conditions. Arch Gen Psychiatry. 2007;64:830–42.
Volkow ND, Koob GF, McLellan AT. Neurobiologic advances from the brain disease model of addiction. N Engl J Med. 2016;374:363–71.
Ethier AR, Kim HS, Hodgins DC, McGrath DS. High rollers: correlates of problematic cocaine use among a community sample of gamblers. J Gambl Stud. 2020;36:513–25.
Bechara A, Damasio AR, Damasio H, Anderson SW. Insensitivity to future consequences following damage to human prefrontal cortex. Cognition. 1994;50:7–15.
Bechara A. Risky business: emotion, decision-making, and addiction. J Gambl Stud. 2003;19:23–51.
Rivalan M, Ahmed SH, Dellu-Hagedorn F. Risk-prone individuals prefer the wrong options on a rat version of the iowa gambling task. Biol Psychiatry. 2009;66:743–9.
Zeeb FD, Robbins TW, Winstanley CA. Serotonergic and dopaminergic modulation of gambling behavior as assessed using a novel rat gambling task. Neuropsychopharmacology. 2009;34:2329–43.
van den Bos R, Koot S, de Visser L. A rodent version of the Iowa Gambling Task: 7 years of progress. Front Psychol. 2014;5:203.
Kwak MJ, Choi SJ, Cai WT, Cho BR, Han J, Park JW, et al. Manipulation of radixin phosphorylation in the nucleus accumbens core modulates risky choice behavior. Prog Neurobiol. 2024;242:102681.
Peters J, Vega T, Weinstein D, Mitchell J, Kayser A. Dopamine and risky decision-making in gambling disorder. eNeuro. 2020;7:ENEURO.0461–19.2020.
Grant JE, Odlaug BL, Chamberlain SR, Hampshire A, Schreiber LR, Kim SW. A proof of concept study of tolcapone for pathological gambling: relationships with COMT genotype and brain activation. Eur Neuropsychopharmacol. 2013;23:1587–96.
Balodis IM, Kober H, Worhunsky PD, Stevens MC, Pearlson GD, Potenza MN. Diminished frontostriatal activity during processing of monetary rewards and losses in pathological gambling. Biol Psychiatry. 2012;71:749–57.
Bolla KI, Eldreth DA, London ED, Kiehl KA, Mouratidis M, Contoreggi C, et al. Orbitofrontal cortex dysfunction in abstinent cocaine abusers performing a decision-making task. Neuroimage. 2003;19:1085–94.
Verdejo-Garcia A, Bechara A. A somatic marker theory of addiction. Neuropharmacology. 2009;56:48–62.
Dandy KL, Gatch MB. The effects of chronic cocaine exposure on impulsivity in rats. Behav Pharmacol. 2009;20:400–5.
Simon NW, Mendez IA, Setlow B. Cocaine exposure causes long-term increases in impulsive choice. Behav Neurosci. 2007;121:543–9.
Kim WY, Cho BR, Kwak MJ, Kim JH. Interaction between trait and housing condition produces differential decision-making toward risk choice in a rat gambling task. Sci Rep. 2017;7:5718.
Ferland JN, Winstanley CA. Risk-preferring rats make worse decisions and show increased incubation of craving after cocaine self-administration. Addict Biol. 2017;22:991–1001.
Cocker PJ, Rotge JY, Daniel ML, Belin-Rauscent A, Belin D. Impaired decision making following escalation of cocaine self-administration predicts vulnerability to relapse in rats. Addict Biol. 2020;25:e12738.
Dong Y, Nasif FJ, Tsui JJ, Ju WY, Cooper DC, Hu XT, et al. Cocaine-induced plasticity of intrinsic membrane properties in prefrontal cortex pyramidal neurons: adaptations in potassium currents. J Neurosci. 2005;25:936–40.
Lee BR, Dong Y. Cocaine-induced metaplasticity in the nucleus accumbens: silent synapse and beyond. Neuropharmacology. 2011;61:1060–9.
Wang X, Liu L, Adams W, Li S, Zhang Q, Li B, et al. Cocaine exposure alters dopaminergic modulation of prefronto-accumbens transmission. Physiol Behav. 2015;145:112–7.
Hynes TJ, Hrelja KM, Hathaway BA, Hounjet CD, Chernoff CS, Ebsary SA, et al. Dopamine neurons gate the intersection of cocaine use, decision making, and impulsivity. Addict Biol. 2021;26:e13022.
Beyer DKE, Horn L, Klinker N, Freund N. Risky decision-making following prefrontal D1 receptor manipulation. Transl Neurosci. 2021;12:432–43.
Zeeb FD, Baarendse PJ, Vanderschuren LJ, Winstanley CA. Inactivation of the prelimbic or infralimbic cortex impairs decision-making in the rat gambling task. Psychopharmacology. 2015;232:4481–91.
Goto Y, Grace AA. Limbic and cortical information processing in the nucleus accumbens. Trends Neurosci. 2008;31:552–8.
Stopper CM, Floresco SB. Contributions of the nucleus accumbens and its subregions to different aspects of risk-based decision making. Cogn Affect Behav Neurosci. 2011;11:97–112.
Ghods-Sharifi S, Floresco SB. Differential effects on effort discounting induced by inactivations of the nucleus accumbens core or shell. Behav Neurosci. 2010;124:179–91.
McGlinchey EM, James MH, Mahler SV, Pantazis C, Aston-Jones G. Prelimbic to accumbens core pathway is recruited in a dopamine-dependent manner to drive cued reinstatement of cocaine seeking. J Neurosci. 2016;36:8700–11.
Stefanik MT, Kupchik YM, Kalivas PW. Optogenetic inhibition of cortical afferents in the nucleus accumbens simultaneously prevents cue-induced transient synaptic potentiation and cocaine-seeking behavior. Brain Struct Funct. 2016;221:1681–9.
Siemsen BM, Barry SM, Vollmer KM, Green LM, Brock AG, Westphal AM, et al. A subset of nucleus accumbens neurons receiving dense and functional prelimbic cortical input are required for cocaine seeking. Front Cell Neurosci. 2022;16:844243.
Roth BL. DREADDs for Neuroscientists. Neuron. 2016;89:683–94.
Bonaventura J, Eldridge MAG, Hu F, Gomez JL, Sanchez-Soto M, Abramyan AM, et al. High-potency ligands for DREADD imaging and activation in rodents and monkeys. Nat Commun. 2019;10:4627.
Jenni NL, Larkin JD, Floresco SB. Prefrontal dopamine D(1) and D(2) receptors regulate dissociable aspects of decision making via distinct ventral striatal and amygdalar circuits. J Neurosci. 2017;37:6200–13.
Stipanovich A, Valjent E, Matamales M, Nishi A, Ahn JH, Maroteaux M, et al. A phosphatase cascade by which rewarding stimuli control nucleosomal response. Nature. 2008;453:879–84.
Nishi A, Matamales M, Musante V, Valjent E, Kuroiwa M, Kitahara Y, et al. Glutamate Counteracts Dopamine/PKA Signaling via Dephosphorylation of DARPP-32 Ser-97 and Alteration of Its Cytonuclear Distribution. J Biol Chem. 2017;292:1462–76.
Beaulieu JM, Gainetdinov RR, Caron MG. The Akt-GSK-3 signaling cascade in the actions of dopamine. Trends Pharmacol Sci. 2007;28:166–72.
Nasif FJ, Hu XT, White FJ. Repeated cocaine administration increases voltage-sensitive calcium currents in response to membrane depolarization in medial prefrontal cortex pyramidal neurons. J Neurosci. 2005;25:3674–9.
Ford KA, Wolf ME, Hu XT. Plasticity of L-type Ca2+ channels after cocaine withdrawal. Synapse. 2009;63:690–7.
Rajadhyaksha AM, Kosofsky BE. Psychostimulants, L-type calcium channels, kinases, and phosphatases. Neuroscientist. 2005;11:494–502.
Kwak MJ, Kim WY, Jung SH, Chung YJ, Kim JH. Differential transcriptome profile underlying risky choice in a rat gambling task. J Behav Addict. 2022;11:845–57.
Ku MJ, Kim CY, Park JW, Lee S, Jeong EY, Jeong JW, et al. Wireless optogenetic stimulation on the prelimbic to the nucleus accumbens core circuit attenuates cocaine-induced behavioral sensitization. Neurobiol Dis. 2024;203:106733.
Dalley JW, Laane K, Pena Y, Theobald DE, Everitt BJ, Robbins TW. Attentional and motivational deficits in rats withdrawn from intravenous self-administration of cocaine or heroin. Psychopharmacology. 2005;182:579–87.
Arrondeau C, Uruena-Mendez G, Marchessaux F, Goutaudier R, Ginovart N. Activation of the mPFC-NAc pathway reduces motor impulsivity but does not affect risk-related decision-making in innately high-impulsive male rats. J Neurosci Res. 2024;102:e25387.
McLellan AT, Lewis DC, O’Brien CP, Kleber HD. Drug dependence, a chronic medical illness: implications for treatment, insurance, and outcomes evaluation. JAMA. 2000;284:1689–95.
MacKillop J, Weafer J, Gray JC, Oshri A, Palmer A, de Wit H. The latent structure of impulsivity: impulsive choice, impulsive action, and impulsive personality traits. Psychopharmacology. 2016;233:3361–70.
Verdejo-Garcia A, Lawrence AJ, Clark L. Impulsivity as a vulnerability marker for substance-use disorders: review of findings from high-risk research, problem gamblers and genetic association studies. Neurosci Biobehav Rev. 2008;32:777–810.
van den Bos R, Jolles J, van der Knaap L, Baars A, de Visser L. Male and female Wistar rats differ in decision-making performance in a rodent version of the Iowa Gambling Task. Behav Brain Res. 2012;234:375–9.
Becker JB, McClellan ML, Reed BG. Sex differences, gender and addiction. J Neurosci Res. 2017;95:136–47.
Acknowledgements
This work was supported by the National Research Foundation of Korea funded by the Ministry of Science and ICT (2022R1A4A5033852, RS-2025-00563430, RS-2025-24873007).
Author information
Authors and Affiliations
Contributions
Joonyeup Han: Writing – original draft, Validation, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Myung Ji Kwak: Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Wha Young Kim: Writing – original draft, Validation, Supervision, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization, Jeong-Hoon Kim: Writing – original draft, Validation, Supervision, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All animal experiments and methods were performed in accordance with the relevant guidelines and regulations. The experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of Yonsei University College of Medicine (Approval No. A-2023-0176). As this study involved only animal subjects, requirements for informed consent from human participants and consent for publication of identifiable human images are not applicable.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Han, J., Kwak, M.J., Kim, W.Y. et al. Chemogenetic modulation of the prelimbic cortex to the nucleus accumbens core pathway reduces cocaine-induced increase of risk preference. Transl Psychiatry (2026). https://doi.org/10.1038/s41398-026-04015-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-026-04015-4