Abstract
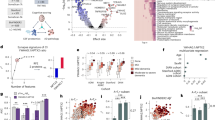
Recent advances have identified YWHAG as a promising synaptic biomarker, with evidence showing that the YWHAG:NPTX2 ratio strongly predicts cognitive decline and Alzheimer’s disease (AD) progression independent of amyloid and tau pathology. However, the links between YWHAG and astrocytic processes-key regulators of amyloid clearance, tau phosphorylation, and neuroinflammation-remain poorly understood. A total of 530 participants were included. Levels of YWHAG and a panel of biologically relevant astrocyte-derived proteins were measured using a proximity extension assay and validated immunoassay platforms. Associations with AD biomarkers and cognition were examined using multivariable regression, longitudinal mixed-effects models, and mediation analyses. Path analysis was performed to explore the potential pathways from YWHAG through astrocytic proteins to AD pathology and cognition. We evaluated whether combining YWHAG with astrocyte-related proteins improves its predictive performance, by comparing the area under the curve (AUC) of the combined model with that of YWHAG alone. YWHAG was positively associated with glial fibrillary acidic protein (GFAP, β = 0.558, p < 0.001), vimentin (β = 0.329, p < 0.001), aquaporin-4 (AQP4, β = 0.097, p = 0.044), thrombospondin (THBS) -1 (β = 0.470, p < 0.001), and THBS2 (β = 0.285, p < 0.001), while showing negative associations with gap junction alpha-1 protein (GJα1, β = -0.161, p < 0.001) and serpin family A member 3 (SERPINA3, β = -0.350, p < 0.001). Mediation analysis indicated that certain astrocyte-related proteins may be involved in the association between YWHAG and AD pathology. Additionally, path analysis suggested a potential pathway involving YWHAG, GJα1, Aβ42, and cognitive function. The combination of YWHAG with SERPINA3 and THBS1 achieved an AUC of 0.981, outperforming YWHAG alone (AUC = 0.885). YWHAG is associated with astrocyte-related proteins, and combining them enhances its predictive accuracy for AD, highlighting its potential utility in early clinical screening.
Similar content being viewed by others
Data availability
Data used in this study were obtained from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) database (https://adni.loni.usc.edu). The ADNI is a publicly available dataset, and access is granted to qualified researchers upon application through the Laboratory of Neuro Imaging (LONI) Image and Data Archive (IDA). Researchers must comply with the ADNI Data Use Agreement and data sharing policies when accessing and using these data.
Code availability
The analytical code used in this study is not publicly available at this stage. Researchers with legitimate scientific purposes may request access to the code from the corresponding author, and the code can be shared upon reasonable request and mutual agreement.
References
Habib N, McCabe C, Medina S, Varshavsky M, Kitsberg D, Dvir-Szternfeld R, et al. Disease-associated astrocytes in Alzheimer’s disease and aging. Nat Neurosci. 2020;23:701–6.
Deng Q, Wu C, Parker E, Liu TC, Duan R, Yang L. Microglia and Astrocytes in Alzheimer’s Disease: Significance and Summary of Recent Advances. Aging Dis. 2024;15:1537–64.
Escartin C, Galea E, Lakatos A, O’Callaghan JP, Petzold GC, Serrano-Pozo A, et al. Reactive astrocyte nomenclature, definitions, and future directions. Nat Neurosci. 2021;24:312–25.
Bolsewig K, van Unnik AAJM, Blujdea ER, Gonzalez MC, Ashton NJ, Aarsland D, et al. Association of plasma amyloid, P-Tau, GFAP, and NfL with CSF, clinical, and cognitive features in patients with dementia with lewy bodies. Neurology. 2024;102:e209418.
Querol-Vilaseca M, Colom-Cadena M, Pegueroles J, San Martín-Paniello C, Clarimon J, Belbin O, et al. YKL-40 (Chitinase 3-like I) is expressed in a subset of astrocytes in Alzheimer’s disease and other tauopathies. J Neuroinflammation. 2017;14:118.
Craig-Schapiro R, Perrin RJ, Roe CM, Xiong C, Carter D, Cairns NJ, et al. YKL-40: a novel prognostic fluid biomarker for preclinical Alzheimer’s disease. Biol Psychiatry. 2010;68:903–12.
Son SM, Nam DW, Cha MY, Kim KH, Byun J, Ryu H, et al. Thrombospondin-1 prevents amyloid beta-mediated synaptic pathology in Alzheimer’s disease. Neurobiol Aging. 2015;36:3214–27.
Tian W, Sawyer A, Kocaoglu FB, Kyriakides TR. Astrocyte-derived thrombospondin-2 is critical for the repair of the blood-brain barrier. Am J Pathol. 2011;179:860–8.
Shigetomi E, Suzuki H, Hirayama YJ, Sano F, Nagai Y, Yoshihara K, et al. Disease-relevant upregulation of P2Y(1) receptor in astrocytes enhances neuronal excitability via IGFBP2. Nat Commun. 2024;15:6525.
Khakh BS, Deneen B. The emerging nature of astrocyte diversity. Annu Rev Neurosci. 2019;42:187–207.
Yang Z, Wang KK. Glial fibrillary acidic protein: from intermediate filament assembly and gliosis to neurobiomarker. Trends Neurosci. 2015;38:364–74.
Heneka MT, Gauthier S, Chandekar SA, Hviid Hahn-Pedersen J, Bentsen MA, Zetterberg H. Neuroinflammatory fluid biomarkers in patients with Alzheimer’s disease: a systematic literature review. Mol Psychiatry. 2025;30:2783–98.
Christopherson KS, Ullian EM, Stokes CC, Mullowney CE, Hell JW, Agah A, et al. Thrombospondins are astrocyte-secreted proteins that promote CNS synaptogenesis. Cell. 2005;120:421–33.
Connolly K, Lehoux M, O’Rourke R, Assetta B, Erdemir GA, Elias JA, et al. Potential role of chitinase-3-like protein 1 (CHI3L1/YKL-40) in neurodegeneration and Alzheimer’s disease. Alzheimers Dement. 2023;19:9–24.
Kim J, Yoo ID, Lim J, Moon JS. Pathological phenotypes of astrocytes in Alzheimer’s disease. Exp Mol Med. 2024;56:95–99.
Oh HS-H, Urey DY, Karlsson L, Zhu Z, Shen Y, Farinas A, et al. A cerebrospinal fluid synaptic protein biomarker for prediction of cognitive resilience versus decline in Alzheimer’s disease. Nat Med. 2025;31:1592–603.
Guo Y, Chen S-D, You J, Huang S-Y, Chen Y-L, Zhang Y, et al. Multiplex cerebrospinal fluid proteomics identifies biomarkers for diagnosis and prediction of Alzheimer’s disease. Nature Human Behaviour. 2024;8:2047–66.
Ramocki MB, Bartnik M, Szafranski P, Kołodziejska KE, Xia Z, Bravo J, et al. Recurrent distal 7q11.23 deletion including HIP1 and YWHAG identified in patients with intellectual disabilities, epilepsy, and neurobehavioral problems. The American Journal of Human Genetics. 2010;87:857–65.
Roy DS, Zhang Y, Aida T, Choi S, Chen Q, Hou Y, et al. Anterior thalamic dysfunction underlies cognitive deficits in a subset of neuropsychiatric disease models. Neuron. 2021;109:2590–603. e2513.
Peng Y, Zhu L, Bai Q, Wang L, Li Q. Serum level of YWHAG as a diagnostic marker of cognitive impairment in Parkinson’s disease patients. Acta Neurol Belg. 2024;124:879–85.
Spellman DS, Wildsmith KR, Honigberg LA, Tuefferd M, Baker D, Raghavan N, et al. Development and evaluation of a multiplexed mass spectrometry based assay for measuring candidate peptide biomarkers in Alzheimer’s Disease Neuroimaging Initiative (ADNI) CSF. Proteomics Clin Appl. 2015;9:715–31.
Pekny M, Pekna M. Astrocyte reactivity and reactive astrogliosis: costs and benefits. Physiol Rev. 2014;94:1077–98.
Risher WC, Kim N, Koh S, Choi JE, Mitev P, Spence EF, et al. Thrombospondin receptor α2δ-1 promotes synaptogenesis and spinogenesis via postsynaptic Rac1. J Cell Biol. 2018;217:3747–65.
Silva I, Silva J, Ferreira R, Trigo D. Glymphatic system, AQP4, and their implications in Alzheimer’s disease. Neurol Res Pract. 2021;3:5.
Bittner T, Zetterberg H, Teunissen CE, Ostlund RE Jr., Militello M, Andreasson U, et al. Technical performance of a novel, fully automated electrochemiluminescence immunoassay for the quantitation of β-amyloid (1-42) in human cerebrospinal fluid. Alzheimers Dement. 2016;12:517–26.
Jack CR Jr., Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB, et al. NIA-AA research framework: toward a biological definition of Alzheimer’s disease. Alzheimers Dement. 2018;14:535–62.
Jagust WJ, Bandy D, Chen K, Foster NL, Landau SM, Mathis CA, et al. The Alzheimer’s disease neuroimaging initiative positron emission tomography core. Alzheimers Dement. 2010;6:221–9.
Zhang Z, Chen X, Sheng Z. Association of triglyceride glucose-body mass index with Alzheimer’s disease pathology, cognition and brain structure in non-demented people. Sci Rep. 2024;14:16097.
Crane PK, Carle A, Gibbons LE, Insel P, Mackin RS, Gross A, et al. Development and assessment of a composite score for memory in the Alzheimer’s Disease Neuroimaging Initiative (ADNI). Brain Imaging Behav. 2012;6:502–16.
Zhang Z, Chen X, Sheng Z, Jiang N, Zou WQ. Cerebrospinal fluid neuronal pentraxin levels are associated with tau pathology via microglia-astrocyte signaling in alzheimer’s disease. J Neuroinflammation. 2025;22:221.
Jack CR Jr., Andrews SJ, Beach TG, Buracchio T, Dunn B, Graf A, et al. Revised criteria for the diagnosis and staging of Alzheimer’s disease. Nat Med. 2024;30:2121–4.
Oh SJ, Woo J, Lee YS, Cho M, Kim E, Cho NC, et al. Direct interaction with 14-3-3γ promotes surface expression of Best1 channel in astrocyte. Mol Brain. 2017;10:51.
Kawamoto Y, Akiguchi I, Tomimoto H, Shirakashi Y, Honjo Y, Budka H. Upregulated expression of 14-3-3 proteins in astrocytes from human cerebrovascular ischemic lesions. Stroke. 2006;37:830–5.
Papanikolopoulou K, Grammenoudi S, Samiotaki M, Skoulakis EMC. Differential effects of 14-3-3 dimers on Tau phosphorylation, stability and toxicity in vivo. Hum Mol Genet. 2018;27:2244–61.
Woo MS, Nilsson J, Therriault J, Rahmouni N, Brinkmalm A, Benedet AL, et al. 14-3-3 ζ/δ-reported early synaptic injury in Alzheimer’s disease is independently mediated by sTREM2. J Neuroinflammation. 2023;20:278.
Foote M, Zhou Y. 14-3-3 proteins in neurological disorders. Int J Biochem Mol Biol. 2012;3:152–64.
Lee DH, Steinacker P, Seubert S, Turnescu T, Melms A, Manzel A, et al. Role of glial 14-3-3 gamma protein in autoimmune demyelination. J Neuroinflammation. 2015;12:187.
Tzivion G, Shen YH, Zhu J. 14-3-3 proteins; bringing new definitions to scaffolding. Oncogene. 2001;20:6331–8.
Ridge KM, Eriksson JE, Pekny M, Goldman RD. Roles of vimentin in health and disease. Genes Dev. 2022;36:391–407.
Perez-Nievas BG, Serrano-Pozo A. Deciphering the astrocyte reaction in Alzheimer’s Disease. Front Aging Neurosci. 2018;10:114.
Tesseur I, Zou K, Esposito L, Bard F, Berber E, Can JV, et al. Deficiency in neuronal TGF-beta signaling promotes neurodegeneration and Alzheimer’s pathology. J Clin Invest. 2006;116:3060–9.
Kang S, Byun J, Son SM, Mook-Jung I. Thrombospondin-1 protects against Aβ-induced mitochondrial fragmentation and dysfunction in hippocampal cells. Cell Death Discov. 2018;4:31.
Sidoryk-Wegrzynowicz M, Gerber YN, Ries M, Sastre M, Tolkovsky AM, Spillantini MG. Astrocytes in mouse models of tauopathies acquire early deficits and lose neurosupportive functions. Acta Neuropathol Commun. 2017;5:89.
Norton ES, Da Mesquita S, Guerrero-Cazares H. SERPINA3 in glioblastoma and Alzheimer’s disease. Aging. 2021;13:21812–3.
Akbor MM, Kurosawa N, Nakayama H, Nakatani A, Tomobe K, Chiba Y, et al. Polymorphic SERPINA3 prolongs oligomeric state of amyloid beta. PLoS ONE. 2021;16:e0248027.
Sofroniew MV, Vinters HV. Astrocytes: biology and pathology. Acta Neuropathol. 2010;119:7–35.
Heneka MT, van der Flier WM, Jessen F, Hoozemanns J, Thal DR, Boche D, et al. Neuroinflammation in Alzheimer disease. Nat Rev Immunol. 2025;25:321–52.
Ferrer I, García MA, González IL, Lucena DD, Villalonga AR, Tech MC, et al. Aging-related tau astrogliopathy (ARTAG): not only tau phosphorylation in astrocytes. Brain Pathol. 2018;28:965–85.
Reid MJ, Beltran-Lobo P, Johnson L, Perez-Nievas BG, Noble W. Astrocytes in Tauopathies. Front Neurol. 2020;11:572850.
Chandra A, Farrell C, Wilson H, Dervenoulas G, De Natale ER, Politis M. Aquaporin-4 polymorphisms predict amyloid burden and clinical outcome in the Alzheimer’s disease spectrum. Neurobiol Aging. 2021;97:1–9.
McGrath ER, Himali JJ, Levy D, Conner SC, DeCarli CS, Pase MP, et al. Circulating IGFBP-2: a novel biomarker for incident dementia. Ann Clin Transl Neurol. 2019;6:1659–70.
Alanko V, Mravinacová S, Hall A, Hagman G, Mohanty R, Westman E, et al. Biological signatures in the Alzheimer’s continuum discriminate between diagnosis-related and -unrelated associations to ATN categories. Brain Commun. 2025;7:fcaf078.
Acknowledgements
Data collection and sharing for this project was funded by the Alzheimer’s Disease Neuroimaging Initiative (ADNI).
Funding
This work was partially supported by the startup package and developmental funds of the First Affiliated Hospital of Nanchang University (#500021001, #500021002), National Natural Science Foundation (NSFC) (82471499), and Jiangxi Key Laboratory of Neurological Diseases (2024SSY06072) to WQZ; Cultivation Project of Jiangxi Medicine Academy of Nutrition and Health Management (2022-PYXM-02), the science and technology research project of Education Department of Jiangxi Province (GJJ2200101), and the Jiangxi Province 03 Special Project and 5 G Project (20232ABC03A23) to XH.
Author information
Authors and Affiliations
Contributions
ZH-Z designed the study, performed statistical analyses, interpreted the data, and drafted the manuscript. PC-H and YR-Y contributed to data analysis, data interpretation, and manuscript revision. HL-W and XR-Y participated in data collection and data curation. YF-W and K.N. contributed to statistical analysis, methodology development, and visualization. YJ-C and YH-W provided resources and assisted with data curation. ZY-C and PP-S contributed to data acquisition and validation. DJ-H supervised the study and contributed to project administration. X.H. and WQ-Z. conceived and supervised the study, secured funding, and critically revised the manuscript. All authors reviewed the manuscript and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent Statement
The study was approved by the Institutional Review Board of the University of Southern California and by the institutional review boards of all participating ADNI sites. Written informed consent was obtained from all participants or their authorized representatives in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, Z., Huang, P., Yang, Y. et al. Astrocyte-related proteins mediate the association of YWHAG with Alzheimer’s pathology and enhance its diagnostic value. Transl Psychiatry (2026). https://doi.org/10.1038/s41398-026-04020-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-026-04020-7