Abstract
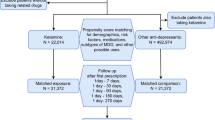
This study aimed to evaluate the long-term effectiveness and safety of esketamine in adults with Major depressive disorder (MDD), focusing on suicide-related events, all-cause mortality, and major adverse cardiovascular events (MACEs). This target-trial simulation used the TriNetX Global Collaborative Network. Adults (≥ 18 years) with MDD who received either esketamine or a conventional antidepressant during an inpatient episode were included. After propensity-score matching for demographic and clinical variables, matched pairs were analyzed followed up to 2 years. The primary outcome was a composite of suicide-related events. Hazard ratios (HRs) with 95% CIs were estimated across acute (days 1–14), intermediate (days 15–365), and long-term (days 15–730) periods. After 1:1 matching, 3383 pairs were analyzed. Esketamine use was associated with lower suicide-related events at days 1–14 (0.77% vs 3.78%; HR, 0.19; 95% CI, 0.12–0.29; P < 0.001), days 15–365 (2.13% vs 2.63%; HR, 0.63; 95% CI, 0.46–0.86; P = 0.003), and days 15–730 (2.66% vs 2.78%; HR, 0.74; 95% CI, 0.55–0.99; P = 0.04). Mortality was lower (6.03% vs 7.63%; HR, 0.61; 95% CI, 0.51–0.73; P < 0.001), as was recurrent MDD at 2 years (3.46% vs 4.67%; HR, 0.76; 95% CI, 0.59–0.98; P = 0.04). MACEs were lower during days 1–14 (2.10% vs 4.97%; HR, 0.39; 95% CI, 0.30–0.51; P < 0.001) and not significantly different thereafter. In conclusion, esketamine treatment was associated with sustained reductions in suicidal ideation and all-cause mortality without increased MACEs risk. The composite suicide-related outcome was primarily driven by reductions in suicidal ideation; differences in suicide attempt and intentional self-harm could not be determined due to low event counts.
Similar content being viewed by others
Data availability
My manuscript has no associated data.
References
Su YA, Ye C, Xin Q, Si T. Major depressive disorder with suicidal ideation or behavior in Chinese population: a scoping review of current evidence on disease assessment, burden, treatment and risk factors. J Affect Disord. 2023;340:732–42. https://doi.org/10.1016/j.jad.2023.08.106.
Cai H, Xie XM, Zhang Q, Cui X, Lin JX, Sim K, et al. Prevalence of suicidality in major depressive disorder: a systematic review and meta-analysis of comparative studies. Front Psychiatry. 2021;12:690130. https://doi.org/10.3389/fpsyt.2021.690130.
Aaltonen K, Sund R, Hakulinen C, Pirkola S, Isometsä E. Variations in suicide risk and risk factors after hospitalization for depression in finland, 1996-2017. JAMA Psychiatry. 2024;81:506–15. https://doi.org/10.1001/jamapsychiatry.2023.5512.
Arnone D, Karmegam SR, Östlundh L, Alkhyeli F, Alhammadi L, Alhammadi S, et al. Risk of suicidal behavior in patients with major depression and bipolar disorder - a systematic review and meta-analysis of registry-based studies. Neurosci Biobehav Rev. 2024;159:105594. https://doi.org/10.1016/j.neubiorev.2024.105594.
McIntyre RS, Alsuwaidan M, Baune BT, Berk M, Demyttenaere K, Goldberg JF, et al. Treatment-resistant depression: definition, prevalence, detection, management, and investigational interventions. World Psychiatry. 2023;22:394–412. https://doi.org/10.1002/wps.21120.
Al-Harbi KS. Treatment-resistant depression: therapeutic trends, challenges, and future directions. Patient Prefer Adherence. 2012;6:369–88. https://doi.org/10.2147/ppa.S29716.
Kennedy SH A review of antidepressant therapy in primary care: current practices and future directions. Prim Care Companion CNS Disord. 2013;15. https://doi.org/10.4088/PCC.12r01420
Lv S, Yao K, Zhang Y, Zhu S. NMDA receptors as therapeutic targets for depression treatment: Evidence from clinical to basic research. Neuropharmacology. 2023;225:109378. https://doi.org/10.1016/j.neuropharm.2022.109378.
Canuso CM, Ionescu DF, Li X, Qiu X, Lane R, Turkoz I, et al. Esketamine nasal spray for the rapid reduction of depressive symptoms in major depressive disorder with acute suicidal ideation or behavior. J Clin Psychopharmacol. 2021;41:516–24. https://doi.org/10.1097/jcp.0000000000001465.
Canuso CM, Singh JB, Fedgchin M, Alphs L, Lane R, Lim P, et al. Efficacy and safety of intranasal esketamine for the rapid reduction of symptoms of depression and suicidality in patients at imminent risk for suicide: results of a double-blind, randomized, placebo-controlled study. Am J Psychiatry. 2018;175:620–30. https://doi.org/10.1176/appi.ajp.2018.17060720.
Daly EJ, Singh JB, Fedgchin M, Cooper K, Lim P, Shelton RC, et al. Efficacy and safety of intranasal esketamine adjunctive to oral antidepressant therapy in treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2018;75:139–48. https://doi.org/10.1001/jamapsychiatry.2017.3739.
Fu DJ, Zhang Q, Shi L, Borentain S, Guo S, Mathews M, et al. Esketamine versus placebo on time to remission in major depressive disorder with acute suicidality. BMC Psychiatry. 2023;23:587. https://doi.org/10.1186/s12888-023-05017-y.
Ochs-Ross R, Wajs E, Daly EJ, Zhang Y, Lane R, Lim P, et al. Comparison of long-term efficacy and safety of esketamine nasal spray plus oral antidepressant in younger versus older patients with treatment-resistant depression: post-hoc analysis of SUSTAIN-2, a long-term open-label phase 3 safety and efficacy study. Am J Geriatr Psychiatry. 2022;30:541–56. https://doi.org/10.1016/j.jagp.2021.09.014.
Rosso G, d’Andrea G, Barlati S, Di Nicola M, Andriola I, Marcatili M, et al. Esketamine treatment trajectory of patients with treatment-resistant depression in the mid and long-term run: data from REAL-ESK study group. Curr Neuropharmacol. 2025;23:612–9. https://doi.org/10.2174/011570159x337670241029062524.
Samalin L, Mekaoui L, Rothärmel M, Sauvaget A, Wicart C, Dupin J, et al. Use of esketamine nasal spray in patients with treatment-resistant depression in routine practice: a real-world French study. Depression Anxiety. 2024;2024:7262794.
Haukoos JS, Lewis RJ. The propensity score. Jama. 2015;314:1637–8. https://doi.org/10.1001/jama.2015.13480.
Taquet M, Geddes JR, Husain M, Luciano S, Harrison PJ. 6-month neurological and psychiatric outcomes in 236 379 survivors of COVID-19: a retrospective cohort study using electronic health records. Lancet Psychiatry. 2021;8:416–27. https://doi.org/10.1016/s2215-0366(21)00084-5.
Wang W, Wang CY, Wang SI, Wei JC. Long-term cardiovascular outcomes in COVID-19 survivors among non-vaccinated population: a retrospective cohort study from the TriNetX US collaborative networks. EClinicalMedicine. 2022;53:101619. https://doi.org/10.1016/j.eclinm.2022.101619.
Liu T-H, Wu J-Y, Huang P-Y, Tsai Y-W, Lai C-C. The effect of nirmatrelvir plus ritonavir on the long-term risk of epilepsy and seizure following COVID-19: a retrospective cohort study including 91,528 patients. J Infect. 2023;86:256–308.
VanderWeele TJ, Ding P. Sensitivity analysis in observational research: introducing the E-value. Ann Intern Med. 2017;167:268–74.
Fu DJ, Ionescu DF, Li X, Lane R, Lim P, Sanacora G, et al. Esketamine nasal spray for rapid reduction of major depressive disorder symptoms in patients who have active suicidal ideation with intent: double-blind, randomized study (ASPIRE I). J Clin Psychiatry. 2020;81: https://doi.org/10.4088/JCP.19m13191
Ionescu DF, Fu DJ, Qiu X, Lane R, Lim P, Kasper S, et al. Esketamine nasal spray for rapid reduction of depressive symptoms in patients with major depressive disorder who have active suicide ideation with intent: results of a phase 3, double-blind, randomized study (ASPIRE II). Int J Neuropsychopharmacol. 2021;24:22–31. https://doi.org/10.1093/ijnp/pyaa068.
Fedgchin M, Trivedi M, Daly EJ, Melkote R, Lane R, Lim P, et al. Efficacy and safety of fixed-dose esketamine nasal spray combined with a new oral antidepressant in treatment-resistant depression: results of a randomized, double-blind, active-controlled study (TRANSFORM-1). Int J Neuropsychopharmacology. 2019;22:616–30.
Pan Y, Gorenflo MP, Davis PB, Kaelber DC, De Luca S, Xu R. Suicidal ideation following ketamine prescription in patients with recurrent major depressive disorder: a nation-wide cohort study. Transl Psychiatry. 2024;14:327. https://doi.org/10.1038/s41398-024-03033-4.
Sanacora G, Ahmed M, Brown B, Cabrera P, Doherty T, Himedan M, et al. Real-world safety of esketamine nasal spray: a comprehensive analysis almost 5 years after first approval. Am J Psychiatry. 2025;182:913–21. https://doi.org/10.1176/appi.ajp.20240655.
Fountoulakis KN, Saitis A, Schatzberg AF. Esketamine treatment for depression in adults: a PRISMA systematic review and meta-analysis. Am J Psychiatry. 2025;182:259–75. https://doi.org/10.1176/appi.ajp.20240515.
Zhang Y, Cai Q, Wang L, Zhang B. The impact of esketamine on depression: targeting oxidative stress and neuronal apoptosis through BDNF/TrkB/PI3K/AKT pathway activation. Neuropsychiatr Dis Treat. 2025;21:1783–93. https://doi.org/10.2147/ndt.S502090.
Halaris A, Cook J. The glutamatergic system in treatment-resistant depression and comparative effectiveness of ketamine and esketamine: role of inflammation?. Adv Exp Med Biol. 2023;1411:487–512. https://doi.org/10.1007/978-981-19-7376-5_21.
Li N, Lee B, Liu RJ, Banasr M, Dwyer JM, Iwata M, et al. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science. 2010;329:959–64. https://doi.org/10.1126/science.1190287.
Pardossi S, Fagiolini A, Cuomo A. Variations in BDNF and their role in the neurotrophic antidepressant mechanisms of ketamine and esketamine: a review. Int J Mol Sci. 2024;25 https://doi.org/10.3390/ijms252313098.
Zaki N, Chen L, Lane R, Doherty T, Drevets WC, Morrison RL, et al. Long-term safety and maintenance of response with esketamine nasal spray in participants with treatment-resistant depression: interim results of the SUSTAIN-3 study. Neuropsychopharmacology. 2023;48:1225–33.
Reutfors J, Andersson TM, Brenner P, Brandt L, DiBernardo A, Li G, et al. Mortality in treatment-resistant unipolar depression: a register-based cohort study in Sweden. J Affect Disord. 2018;238:674–9.
Feldman RL, Dunner DL, Muller JS, Stone DA. Medicare patient experience with vagus nerve stimulation for treatment-resistant depression. J Med Econ. 2013;16:62–74.
Wajs E, Aluisio L, Holder R, Daly EJ, Lane R, Lim P, et al. Esketamine nasal spray plus oral antidepressant in patients with treatment-resistant depression: assessment of long-term safety in a phase 3, open-label study (SUSTAIN-2). J Clin Psychiatry. 2020;81. https://doi.org/10.4088/JCP.19m12891.
Doherty T, Wajs E, Melkote R, Miller J, Singh JB, Weber MA. Cardiac safety of esketamine nasal spray in treatment-resistant depression: results from the clinical development program. CNS Drugs. 2020;34:299–310. https://doi.org/10.1007/s40263-020-00699-4.
Janik A, Qiu X, Lane R, Popova V, Drevets WC, Canuso CM, et al. Esketamine monotherapy in adults with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2025;82:877–87. https://doi.org/10.1001/jamapsychiatry.2025.1317.
Harding L, Joshi K, Zhdanava M, Shah A, Voegel A, Chen C, et al. Treatment patterns, acute healthcare resource use, and costs of patients with treatment-resistant depression completing induction phase of esketamine in the united states. Drugs Real World Outcomes. 2024;11:209–19. https://doi.org/10.1007/s40801-024-00425-2.
Janssen Pharmaceuticals, Inc. SPRAVATO (esketamine) REMS Program Overview: Risk Evaluation and Mitigation Strategy. Titusville, NJ: Janssen Pharmaceuticals, Inc.; 2019. Accessed October 10, 2025. https://www.spravatorems.com.
Yoshida K, Solomon DH, Kim SC. Active-comparator design and new-user design in observational studies. Nat Rev Rheumatol. 2015;11:437–41.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, TH., Shen, HS., Wu, JY. et al. Sustained effectiveness and safety of esketamine for major depressive disorder: a target trial simulation of real-world data. Transl Psychiatry (2026). https://doi.org/10.1038/s41398-026-04032-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-026-04032-3