Abstract
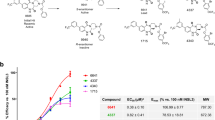
Relaxin/insulin-like family peptide receptor 4 (RXFP4) is a class A G protein-coupled receptor (GPCR), and insulin-like peptide 5 (INSL5) is its endogenous ligand. Although the precise physiological role of INSL5/RXFP4 remains elusive, a number of studies have suggested it to be a potential therapeutic target for obesity and other metabolic disorders. Since selective agonists of RXFP4 are scarcely available and peptidic analogs of INSL5 are hard to make, we conducted a high-throughput screening campaign against 52,000 synthetic and natural compounds targeting RXFP4. Of the 109 initial hits discovered, only 3 compounds were confirmed in secondary screening, with JK0621-D008 displaying the best agonism at human RXFP4. Its S-configuration stereoisomer (JK1) was subsequently isolated and validated by a series of bioassays, demonstrating a consistent agonistic effect in cells overexpressing RXFP4. This scaffold may provide a valuable tool to further explore the biological functions of RXFP4.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Liu C, Kuei C, Sutton S, Chen J, Bonaventure P, Wu J, et al. INSL5 is a high affinity specific agonist for GPCR142 (GPR100). J Biol Chem. 2005;280:292–300.
Fredriksson R, Lagerstrom MC, Lundin LG, Schioth HB. The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol Pharmacol. 2003;63:1256–72.
Boels K, Schaller HC. Identification and characterisation of GPR100 as a novel human G-protein-coupled bradykinin receptor. Br J Pharmacol. 2003;140:932–8.
Kong RC, Shilling PJ, Lobb DK, Gooley PR, Bathgate RA. Membrane receptors: structure and function of the relaxin family peptide receptors. Mol Cell Endocrinol. 2010;320:1–15.
Akhter Hossain M, Bathgate RA, Kong CK, Shabanpoor F, Zhang S, Haugaard-Jonsson LM, et al. Synthesis, conformation, and activity of human insulin-like peptide 5 (INSL5). ChemBioChem. 2008;9:1816–22.
Conklin D, Lofton-Day CE, Haldeman BA, Ching A, Whitmore TE, Lok S, et al. Identification of INSL5, a new member of the insulin superfamily. Genomics. 1999;60:50–6.
Ang SY, Hutchinson DS, Patil N, Evans BA, Bathgate RAD, Halls ML, et al. Signal transduction pathways activated by insulin-like peptide 5 at the relaxin family peptide RXFP4 receptor. Br J Pharmacol. 2017;174:1077–89.
Luo X, Li T, Zhu Y, Dai Y, Zhao J, Guo ZY, et al. The insulinotrophic effect of insulin-like peptide 5 in vitro and in vivo. Biochem J. 2015;466:467–73.
Aparicio S, Dixon J, Hendrick A. Use of GPR100 receptor in diabetes and obesity regulation. US Patent Appl 20080269118.
Burnicka-Turek O, Mohamed BA, Shirneshan K, Thanasupawat T, Hombach-Klonisch S, Klonisch T, et al. INSL5-deficient mice display an alteration in glucose homeostasis and an impaired fertility. Endocrinology. 2012;153:4655–65.
Grosse J, Heffron H, Burling K, Akhter Hossain M, Habib AM, Rogers GJ, et al. Insulin-like peptide 5 is an orexigenic gastrointestinal hormone. Proc Natl Acad Sci U S A. 2014;111:11133–8.
DeChristopher B, Park SH, Vong L, Bamford D, Cho HH, Duvadie R, et al. Discovery of a small molecule RXFP3/4 agonist that increases food intake in rats upon acute central administration. Bioorg Med Chem Lett. 2019;29:991–4.
Luo X, Bathgate RA, Zhang WJ, Liu YL, Shao XX, Wade JD, et al. Design and recombinant expression of insulin-like peptide 5 precursors and the preparation of mature human INSL5. Amino Acids. 2010;39:1343–52.
Patil NA, Hughes RA, Rosengren KJ, Kocan M, Ang SY, Tailhades J, et al. Engineering of a novel simplified human insulin-like peptide 5 agonist. J Med Chem. 2016;59:2118–25.
Belgi A, Hossain MA, Shabanpoor F, Chan L, Zhang S, Bathgate RA, et al. Structure and function relationship of murine insulin-like peptide 5 (INSL5): free C-terminus is essential for RXFP4 receptor binding and activation. Biochemistry. 2011;50:8352–61.
Van der Westhuizen ET, Sexton PM, Bathgate RA, Summers RJ. Responses of GPCR135 to human gene 3 (H3) relaxin in CHO-K1 cells determined by microphysiometry. Ann N Y Acad Sci. 2005;1041:332–7.
Zhang JH, Chung TD, Oldenburg KR. A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J Biomol Screen. 1999;4:67–73.
Bathgate RA, Ivell R, Sanborn BM, Sherwood OD, Summers RJ. International union of pharmacology LVII: recommendations for the nomenclature of receptors for relaxin family peptides. Pharmacol Rev. 2006;58:7–31.
Faure M, Voyno-Yasenetskaya TA, Bourne HR. cAMP and beta gamma subunits of heterotrimeric G proteins stimulate the mitogen-activated protein kinase pathway in COS-7 cells. J Biol Chem. 1994;269:7851–4.
Crespo P, Xu N, Simonds WF, Gutkind JS. Ras-dependent activation of MAP kinase pathway mediated by G-protein beta gamma subunits. Nature. 1994;369:418–20.
Lopez-Ilasaca M, Crespo P, Pellici PG, Gutkind JS, Wetzker R. Linkage of G protein-coupled receptors to the MAPK signaling pathway through PI3-kinase gamma. Science. 1997;275:394–7.
Bathgate RAD, Halls ML, van der Westhuizen ET, Callander GE, Kocan M, Summers RJ, et al. Relaxin family peptides and their receptors. Physiol Rev. 2013;93:405–80.
Hu MJ, Shao XX, Wang JH, Wei D, Guo YQ, Liu YL, et al. Mechanism for insulin-like peptide 5 distinguishing the homologous relaxin family peptide receptor 3 and 4. Sci Rep. 2016;6:29648.
Alvarez-Jaimes L, Sutton SW, Nepomuceno D, Motley ST, Cik M, et al. In vitro pharmacological characterization of RXFP3 allosterism: an example of probe dependency. PLoS ONE. 2012;7:e30792.
Chen CZ, Southall N, Xiao J, Marugan JJ, Ferrer M, Hu X, et al. Identification of small-molecule agonists of human relaxin family receptor 1 (RXFP1) by using a homogenous cell-based cAMP assay. J Biomol Screen. 2013;18:670–7.
Xiao J, Huang Z, Chen CZ, Agoulnik IU, Southall N, Hu X, et al. Identification and optimization of small-molecule agonists of the human relaxin hormone receptor RXFP1. Nat Commun. 2013;4:1953.
Acknowledgements
We are indebted to Ji Wu and Qiang Shen for technical assistance. This work was partially supported by grants from the National Natural Science Foundation of China 81872915 (MWW), 81573479 (DHY), and 81773792 (DHY); the National Science & Technology Major Project “Key New Drug Creation and Manufacturing Program” of China (2018ZX09735-001 to MWW, 2018ZX09711002-002-005 to DHY and 2018ZX09711002-002-011 to QL); the National Key R&D Program of China 2018YFA0507000 (MWW); and the Novo Nordisk-CAS Research Fund (NNCAS-2017-1-CC to DHY). Research at the Florey was supported by the Victorian Government’s Operational Infrastructure Support Program. We are grateful to Tania Ferraro and Sharon Layfield for assistance with assays at the Florey. RADB is an NHMRC Senior Research Fellow. The funders had no role in the study design, data collection and analysis, decision to publish, or paper preparation.
Author information
Authors and Affiliations
Contributions
MWW and RADB designed the research. GYL, XQC, ATD, and JL performed the research. YZ established the hRXFP4-Gα16-CHO cell line. LL and QL synthesized JK1. GYL, LL, QL, DHY, RADB, and MWW analyzed the data. GYL, QL, MWW, and RADB drafted the paper, and GYL, QL, DHY, RADB, and MWW edited and revised the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Lin, Gy., Lin, L., Cai, Xq. et al. High-throughput screening campaign identifies a small molecule agonist of the relaxin family peptide receptor 4. Acta Pharmacol Sin 41, 1328–1336 (2020). https://doi.org/10.1038/s41401-020-0390-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-020-0390-x
Keywords
This article is cited by
-
Ligand recognition mechanism of the human relaxin family peptide receptor 4 (RXFP4)
Nature Communications (2023)