Abstract
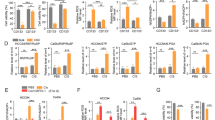
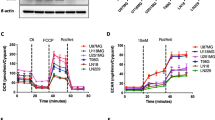
Inhibition of glycolysis process has been an attractive approach for cancer treatment due to the evidence that tumor cells are more dependent on glycolysis rather than oxidative phosphorylation pathway. Preliminary evidence shows that inhibition of phosphoglycerate kinase 1 (PGK1) kinase activity would reverse the Warburg effect and make tumor cells lose the metabolic advantage for fueling the proliferation through restoration of the pyruvate dehydrogenase (PDH) activity and subsequently promotion of pyruvic acid to enter the Krebs cycle in glioma. However, due to the lack of small molecule inhibitors of PGK1 kinase activity to treat glioma, whether PGK1 could be a therapeutic target of glioma has not been pharmacologically verified yet. In this study we developed a high-throughput screening and discovered that NG52, previously known as a yeast cell cycle-regulating kinase inhibitor, could inhibit the kinase activity of PGK1 (the IC50 = 2.5 ± 0.2 μM). We showed that NG52 dose-dependently inhibited the proliferation of glioma U87 and U251 cell lines with IC50 values of 7.8 ± 1.1 and 5.2 ± 0.2 μM, respectively, meanwhile it potently inhibited the proliferation of primary glioma cells. We further revealed that NG52 (12.5–50 μM) effectively inhibited the phosphorylation of PDHK1 at Thr338 site and the phosphorylation of PDH at Ser293 site in U87 and U251 cells, resulting in more pyruvic acid entering the Krebs cycle with increased production of ATP and ROS. Therefore, NG52 could reverse the Warburg effect by inhibiting PGK1 kinase activity, and switched cellular glucose metabolism from anaerobic mode to aerobic mode. In nude mice bearing patient-derived glioma xenograft, oral administration of NG52 (50, 100, 150 mg· kg−1·d−1, for 13 days) dose-dependently suppressed the growth of glioma xenograft. Together, our results demonstrate that targeting PGK1 kinase activity might be a potential strategy for glioma treatment.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Aldape K, Zadeh G, Mansouri S, Reifenberger G, von Deimling A. Glioblastoma: pathology, molecular mechanisms and markers. Acta Neuropathol. 2015;129:829–48.
Hess KR, Broglio KR, Bondy ML. Adult glioma incidence trends in the United States, 1977–2000. Cancer. 2004;101:2293–9.
Roth P, Gramatzki D, Weller M. Management of elderly patients with glioblastoma. Curr Neurol Neurosci Rep. 2017;17:35.
Molina JR, Hayashi Y, Stephens C, Georgescu M-M. Invasive glioblastoma cells acquire stemness and increased Akt activation. Neoplasia. 2010;12:453–63.
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJB, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352:987–96.
Wen PY, Kesari S. Malignant gliomas in adults. N Engl J Med. 2008;359:492–507.
Dolecek TA, Propp JM, Stroup NE, Kruchko C. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2005–2009. Neuro Oncol. 2012;14:1–49.
Cairns RA, Harris IS, Mak TW. Regulation of cancer cell metabolism. Nat Rev Cancer. 2011;11:85–95.
Yang W, Lu Z. Regulation and function of pyruvate kinase M2 in cancer. Cancer Lett. 2013;339:153–8.
Bernstein BE, Hol WGJ. Crystal structures of substrates and products bound to the phosphoglycerate kinase active site reveal the catalytic mechanism. Biochemistry. 1998;37:4429–36.
Li X, Zheng Y, Lu Z. PGK1 is a new member of the protein kinome. Cell Cycle. 2016;15:1803–4.
Qian X, Li X, Cai Q, Zhang C, Yu Q, Jiang Y, et al. Phosphoglycerate kinase 1 phosphorylates Beclin1 to induce autophagy. Mol Cell. 2017;65:917–31.e6.
Qian X, Li X, Lu Z. Protein kinase activity of the glycolytic enzyme PGK1 regulates autophagy to promote tumorigenesis. Autophagy. 2017;13:1246–7.
Li X, Jiang Y, Meisenhelder J, Yang W, Hawke DH, Zheng Y, et al. Mitochondria-translocated PGK1 functions as a protein kinase to coordinate glycolysis and the TCA cycle in tumorigenesis. Mol Cell. 2016;61:705–19.
Zhang D, Tai LK, Wong LL, Chiu LL, Sethi SK, Koay ES. Proteomic study reveals that proteins involved in metabolic and detoxification pathways are highly expressed in HER-2/neu-positive breast cancer. Mol Cell Proteomics. 2005;4:1686–96.
Grundker C, Wokoun U, Hellriegel M, Emons G. Inhibition of aerobic glycolysis enhances the anti-tumor efficacy of Zoptarelin doxorubicin in triple-negative breast cancer cells. J Obstet Gynaecol Res. 2019;45:1334–42.
Ai J, Huang H, Lv X, Tang Z, Chen M, Chen T, et al. FLNA and PGK1 are two potential markers for progression in hepatocellular carcinoma. Cell Physiol Biochem. 2011;27:207–16.
Xie H, Tong G, Zhang Y, Liang S, Tang K, Yang Q. PGK1 drives hepatocellular carcinoma metastasis by enhancing metabolic process. Int J Mol Sci. 2017;18:1630.
Hu H, Zhu W, Qin J, Chen M, Gong L, Li L, et al. Acetylation of PGK1 promotes liver cancer cell proliferation and tumorigenesis. Hepatology. 2017;65:515–28.
Gray NS, Wodicka L, Thunnissen A-MWH, Norman TC, Kwon S, Espinoza FH, et al. Exploiting chemical libraries, structure, and genomics in the search for kinase inhibitors. Science. 1998;281:533–8.
Chen X, Zhao C, Li X, Wang T, Li Y, Cao C, et al. Terazosin activates Pgk1 and Hsp90 to promote stress resistance. Nat Chem Biol. 2015;11:19–25.
Lu J, Tan M, Cai Q. The Warburg effect in tumor progression: mitochondrial oxidative metabolism as an anti-metastasis mechanism. Cancer Lett. 2015;356:156–64.
Li L, Dong B. The correlation of hypoxic tumor microenvironment, Warburg effect and epithelial-mesenchymal transition. Chin J Biochem Mol Biol. 2018;34:949–53.
Zhang Q, Dong P, Liu X, Sakuragi N, Guo SW. Enhancer of Zeste homolog 2 (EZH2) induces epithelial-mesenchymal transition in endometriosis. Sci Rep. 2017;7:6804.
Warburg O. On the origin of cancer cells. Science. 1956;123:309–14.
Luengo A, Gui DY, Vander Heiden MG. Targeting metabolism for cancer therapy. Cell Chem Biol. 2017;24:1161–80.
Nakazawa MS, Keith B, Simon MC. Oxygen availability and metabolic adaptations. Nat Rev Cancer. 2016;16:663–73.
Papandreou I, Cairns RA, Fontana L, Lim AL, Denko NC. HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab. 2006;3:187–97.
Kim JW, Tchernyshyov I, Semenza GL, Dang CV. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 2006;3:177–85.
Le A, Cooper CR, Gouw AM, Dinavahi R, Maitra A, Deck LM, et al. Inhibition of lactate dehydrogenase A induces oxidative stress and inhibits tumor progression. Proc Natl Acad Sci U S A. 2010;107:2037–42.
Fantin VR, St-Pierre J, Leder P. Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell. 2006;9:425–34.
Zachar Z, Marecek J, Maturo C, Gupta S, Stuart SD, Howell K, et al. Non-redox-active lipoate derivates disrupt cancer cell mitochondrial metabolism and are potent anticancer agents in vivo. J Mol Med. 2011;89:1137–48.
Yang L, Xie M, Yang M, Yu Y, Zhu S, Hou W, et al. PKM2 regulates the Warburg effect and promotes HMGB1 release in sepsis. Nat Commun. 2014;5:4436.
Anastasiou D, Yu Y, Israelsen WJ, Jiang JK, Boxer MB, Hong BS, et al. Pyruvate kinase M2 activators promote tetramer formation and suppress tumorigenesis. Nat Chem Biol. 2012;8:839–47.
Chen J, Xie J, Jiang Z, Wang B, Wang Y, Hu X. Shikonin and its analogs inhibit cancer cell glycolysis by targeting tumor pyruvate kinase-M2. Oncogene. 2011;30:4297–306.
Giannoni E, Taddei ML, Morandi A, Comito G, Calvani M, Bianchini F, et al. Targeting stromal-induced pyruvate kinase M2 nuclear translocation impairs OXPHOS and prostate cancer metastatic spread. Oncotarget. 2015;6:24061–74.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grants 81773777, 81673469, 81872748, 81803366), the National Key Research and Development Program of China (Grant 2016YFA0400900), the Natural Science Foundation of Anhui Province (Grant 1908085QH348), the Science and Technology Major Projects of Anhui Province (Grant 17030801025), the China Postdoctoral Science Foundation (Grants 2018T110634, 2018M630720, 2019M652057), the Frontier Science Key Research Program of CAS (Grant QYZDB-SSW-SLH037), the CASHIPS Director’s Fund (Grant BJPY2019A03), and the Key Program of 13th Five-Year Plan of CASHIPS (Grant KP-2017-26). We are also grateful to the National Program for Support of Top-Notch Young Professionals for Jing Liu.
Author information
Authors and Affiliations
Contributions
QSL and JL designed the research. WLW, ZRJ, ALW, and CH developed the methodology. WLW, ZRJ, ZQH, and LW performed the experiments and collected the data. WLW, WCW, and QSL wrote the manuscript. LW, CC, and CH provided the experiment materials. QSL, WCW, and JL revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, Wl., Jiang, Zr., Hu, C. et al. Pharmacologically inhibiting phosphoglycerate kinase 1 for glioma with NG52. Acta Pharmacol Sin 42, 633–640 (2021). https://doi.org/10.1038/s41401-020-0465-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-020-0465-8
Keywords
This article is cited by
-
Exploring a specialized lactylation pattern to predict the prognosis and sensitivity of immunotherapy in breast cancer via machine learning
Breast Cancer (2026)
-
PGK1 enhances productive bovine herpesvirus 1 infection by stimulating β-catenin-dependent transcription
Veterinary Research (2025)
-
Pan-cancer investigation regarding the prognostic predictive and immunological regulation functions of PGK1 and experimental validation in esophageal squamous cell carcinoma
Functional & Integrative Genomics (2025)
-
Post-translational modifications and the reprogramming of tumor metabolism
Discover Oncology (2025)
-
Glycolytic enzyme PGK1 promotes M1 macrophage polarization and induces pyroptosis of acute lung injury via regulation of NLRP3
Respiratory Research (2024)