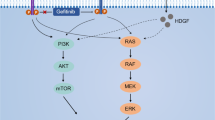
Abstract
Recent studies show that intracellular accumulation of cholesterol leads to acquired resistance to gefitinib in non-small cell lung cancer (NSCLC) cells. In this study we investigated how to regulate the cholesterol levels in gefitinib-resistant NSCLC cells. We showed that intracellular cholesterol levels in gefitinib-resistant cell lines (PC-9/GR, H1975, H1650, and A549) were significantly higher than that in gefitinib-sensitive cell line (PC-9). Treatment with gefitinib (5 μM) significantly increased intracellular cholesterol levels in PC-9/GR, H1975, and H1650 cells. Gefitinib treatment downregulated the expression of PPARα, LXRα, and ABCA1, leading to dysregulation of cholesterol efflux pathway. We found that a lipid-lowering drug fenofibrate (20, 40 μM) dose-dependently increased the expression of PPARα, LXRα, and ABCA1, decreased the intracellular cholesterol levels, and enhanced the antiproliferative effects of gefitinib in PC-9/GR, H1975, and H1650 cells. We revealed that fenofibrate increased the gefitinib-induced apoptosis via regulating the key proteins involved in the intrinsic apoptosis pathway. In PC-9/GR, H1975 and H1650 cells, fenofibrate dose-dependently increased the expression of AMPK, FoxO1, and decreased the expression of AKT, which were remarkably weakened by knockdown of PPARα. In PC-9/GR cell xenograft mice, combined administration of gefitinib (25 mg · kg−1 · d−1) and fenofibrate (100 mg · kg−1 · d−1) caused remarkable inhibition on tumor growth as compared to treatment with either drug alone. All the results suggest that fenofibrate relieves acquired resistance to gefitinib in NSCLC by promoting apoptosis via regulating PPARα/AMPK/AKT/FoxO1 pathway. We propose that combination of gefitinib and fenofibrate is a potential strategy for overcoming the gefitinib resistance in NSCLC.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018;553:446–54.
Ettinger DS, Aisner DL, Wood DE, Akerley W, Bauman J, Chang JY, et al. NCCN guidelines insights: non-small cell lung cancer, version 5.2018. J Natl Compr Canc Netw. 2018;16:807–21.
Kosaka T, Yatabe Y, Endoh H, Yoshida K, Hida T, Tsuboi M, et al. Analysis of epidermal growth factor receptor gene mutation in patients with non-small cell lung cancer and acquired resistance to gefitinib. Clin Cancer Res. 2006;12:5764–9.
Rocco D, Battiloro C, Gravara LD, Gridelli C. Advanced non-small cell lung cancer with activating epidermal growth factor receptor mutation: first line treatment and beyond. Rev Recent Clin Trials. 2019;14:120–8.
Wu SG, Shih JY. Management of acquired resistance to EGFR TKI-targeted therapy in advanced non-small cell lung cancer. Mol Cancer. 2018;17:38.
Hirsch HA, Iliopoulos D, Joshi A, Zhang Y, Jaeger SA, Bulyk M, et al. A transcriptional signature and common gene networks link cancer with lipid metabolism and diverse human diseases. Cancer Cell. 2010;17:348–61.
Ali A, Levantini E, Fhu CW, Teo JT, Clohessy JG, Goggi JL, et al. CAV1– GLUT3 signaling is important for cellular energy and can be targeted by atorvastatin in non-small cell lung cancer. Theranostics. 2019;9:6157–74.
Ercan D, Choi HG, Yun CH, Capelletti M, Xie T, Eck MJ, et al. EGFR mutations and resistance to irreversible pyrimidine-based EGFR inhibitors. Clin Cancer Res. 2015;21:3913–23.
Chen Q, Pan Z, Zhao M, Wang Q, Qiao C, Miao L, et al. High cholesterol in lipid rafts reduces the sensitivity to EGFR-TKI therapy in non-small cell lung cancer. J Cell Physiol. 2018;233:6722–32.
Ali A, Levantini E, Teo JT, Goggi J, Clohessy JG, Wu CS, et al. Fatty acid synthase mediates EGFR palmitoylation in EGFR mutated non-small cell lung cancer. EMBO Mol Med. 2018;10:e8313.
Smith B, Land H. Anticancer activity of the cholesterol exporter ABCA1 gene. Cell Rep. 2012;2:580–90.
Sharma B, Agnihotri N. Role of cholesterol homeostasis and its efflux pathways in cancer progression. J Steroid Biochem Mol Biol. 2019;191:105377.
Pawlak M, Lefebvre P, Staels B. Molecular mechanism of PPARalpha action and its impact on lipid metabolism, inflammation and fibrosis in non-alcoholic fatty liver disease. J Hepatol. 2015;62:720–33.
Ogata M, Tsujita M, Hossain MA, Akita N, Gonzalez FJ, Staels B, et al. On the mechanism for PPAR agonists to enhance ABCA1 gene expression. Atherosclerosis. 2009;205:413–9.
Xu N, Wang Q, Jiang S, Wang Q, Hu W, Zhou S, et al. Fenofibrate improves vascular endothelial function and contractility in diabetic mice. Redox Biol. 2019;20:87–97.
Desvergne B, Wahli W. Peroxisome proliferator-activated receptors: nuclear control of metabolism. Endocr Rev. 1999;20:649–88.
Lian X, Wang G, Zhou H, Zheng Z, Fu Y, Cai L. Anticancer properties of fenofibrate: a repurposing use. J Cancer. 2018;9:1527–37.
Sun J, Zheng Z, Chen Q, Pan Y, Quan M, Dai Y. Fenofibrate potentiates chemosensitivity to human breast cancer cells by modulating apoptosis via AKT/NF-kappaB pathway. Onco Targets Ther. 2019;12:773–83.
Luty M, Piwowarczyk K, Labedz-Maslowska A, Wrobel T, Szczygiel M, Catapano J, et al. Fenofibrate augments the sensitivity of drug-resistant prostate cancer cells to docetaxel. Cancers (Basel). 2019;11:77.
Sirotnak FM, Zakowski MF, Miller VA, Scher HI, Kris MG. Efficacy of cytotoxic agents against human tumor xenografts is markedly enhanced by coadministration of ZD1839 (Iressa), an inhibitor of EGFR tyrosine kinase. Clin Cancer Res. 2000;6:4885–92.
Ding X, Zhang W, Li S, Yang H. The role of cholesterol metabolism in cancer. Am J Cancer Res. 2019;9:219–27.
Mabuchi H, Kita T, Matsuzaki M, Matsuzawa Y, Nakaya N, Oikawa S, et al. Largescale cohort study of the relationship between serum cholesterol concentration and coronary events with low-dose simvastatin therapy in Japanese patients with hypercholesterolemia and coronary heart disease: secondary prevention cohort study of the Japan Lipid Intervention Trial (J-LIT). Circ J. 2002;66:1096–100.
Ikonen E. Cellular cholesterol trafficking and compartmentalization. Nat Rev Mol Cell Biol. 2008;9:125–38.
Kuzu OF, Noory MA, Robertson GP. The role of cholesterol in cancer. Cancer Res. 2016;76:2063–70.
Gobel A, Breining D, Rauner M, Hofbauer LC, Rachner TD. Induction of 3-hydroxy-3-methylglutaryl-CoA reductase mediates statin resistance in breast cancer cells. Cell Death Dis. 2019;10:91.
Villa GR, Hulce JJ, Zanca C, Bi J, Ikegami S, Cahill GL, et al. An LXR-cholesterol axis creates a metabolic co-dependency for brain cancers. Cancer Cell. 2016;30:683–93.
Staels B, Dallongeville J, Auwerx J, Schoonjans K, Leitersdorf E, Fruchart JC. Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation. 1998;98:2088–93.
Chen L, Peng J, Wang Y, Jiang H, Wang W, Dai J, et al. Fenofibrate-induced mitochondrial dysfunction and metabolic reprogramming reversal: the anti-tumor effects in gastric carcinoma cells mediated by the PPAR pathway. Am J Transl Res. 2020;12:428–46.
Gardette V, Bongard V, Dallongeville J, Arveiler D, Bingham A, Ruidavets JB, et al. Ten-year all-cause mortality in presumably healthy subjects on lipid-lowering drugs (from the Prospective Epidemiological Study of Myocardial Infarction [PRIME] prospective cohort). Am J Cardiol. 2009;103:381–6.
Goncalves MD, Hwang SK, Pauli C, Murphy CJ, Cheng Z, Hopkins BD, et al. Fenofibrate prevents skeletal muscle loss in mice with lung cancer. Proc Natl Acad Sci USA. 2018;115:E743–52.
Li T, Zhang Q, Zhang J, Yang G, Shao Z, Luo J, et al. Fenofibrate induces apoptosis of triple-negative breast cancer cells via activation of NF-kappaB pathway. BMC Cancer. 2014;14:96.
Zhao H, Zhu C, Qin C, Tao T, Li J, Cheng G, et al. Fenofibrate down-regulates the expressions of androgen receptor (AR) and AR target genes and induces oxidative stress in the prostate cancer cell line LNCaP. Biochem Biophys Res Commun. 2013;432:320–5.
Binello E, Mormone E, Emdad L, Kothari H, Germano IM. Characterization of fenofibrate-mediated anti-proliferative pro-apoptotic effects on high-grade gliomas and anti-invasive effects on glioma stem cells. J Neurooncol. 2014;117:225–34.
Sun J, Zheng Z, Chen Q, Pan Y, Quan M, Dai Y. Fenofibrate potentiates chemosensitivity to human breast cancer cells by modulating apoptosis via AKT/NF-kappaB pathway. Oncol Targets Ther. 2019;12:773–83.
Zou J, Hong L, Luo C, Li Z, Zhu Y, Huang T, et al. Metformin inhibits estrogen-dependent endometrial cancer cell growth by activating the AMPK-FOXO1 signal pathway. Cancer Sci. 2016;107:1806–17.
Chen WL, Chen YL, Chiang YM, Wang SG, Lee HM. Fenofibrate lowers lipid accumulation in myotubes by modulating the PPARalpha/AMPK/FoxO1/ATGL pathway. Biochem Pharmacol. 2012;84:522–31.
Acknowledgements
We thank molecular Experiment Platform of Stat Key Laboratory of Natural Medicines at China Pharmaceutical University for providing Flow Cytometry.
Author information
Authors and Affiliations
Contributions
MSW, QSH, and ZRJ designed the research methods, performed the experiments, and prepared the manuscript. MSW analyzed the data. CSC, QQL, YMZ, KWW, JW, KX, and CQ participated in experiments. XSD conceived the projects, designed, and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Wang, Ms., Han, Qs., Jia, Zr. et al. PPARα agonist fenofibrate relieves acquired resistance to gefitinib in non-small cell lung cancer by promoting apoptosis via PPARα/AMPK/AKT/FoxO1 pathway. Acta Pharmacol Sin 43, 167–176 (2022). https://doi.org/10.1038/s41401-021-00638-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-021-00638-z
Keywords
This article is cited by
-
Intracellular cholesterol: new functions and therapeutic approaches in NSCLC EGFR-TKI resistance
Lipids in Health and Disease (2025)
-
FOXO1 phosphorylation as a context-dependent molecular switch in cancer: from mechanisms to precision therapy
World Journal of Surgical Oncology (2025)
-
Lipid-lowering drugs and cancer: an updated perspective
Pharmacological Reports (2024)
-
Dual-responsive nanoparticles loading bevacizumab and gefitinib for molecular targeted therapy against non-small cell lung cancer
Acta Pharmacologica Sinica (2023)
-
Construction of an anoikis-associated lncRNA-miRNA-mRNA network reveals the prognostic role of β-elemene in non-small cell lung cancer
Scientific Reports (2023)