Abstract
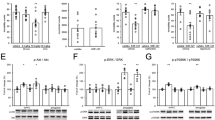
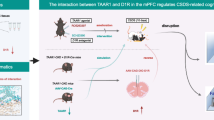
Major depressive disorder is a global mental illness associated with severe mortality and disability. The dopaminergic system is involved in both the etiology and therapeutics of depression. Distinct functions of dopamine D1 and D2 receptor subtypes have attracted considerable research interest, and their roles in the pathogenesis of depression and interaction with antidepressants need to be comprehensively elucidated. Herein, we investigated the antidepressant effects of a candidate antidepressant from a cinnamamide derivative, M2, and examined underlying neural mechanisms. We observed that a single dose of M2 (30 mg/kg, ip) produced rapid antidepressant-like effects in mice subjected to the forced swim and tail suspension tests. Using whole-cell recordings in mouse coronal brain slices, we found that application of M2 (10–150 μM) concentration-dependently increased the frequency of spontaneous excitatory postsynaptic currents (sEPSCs) of the pyramidal neurons in the medial prefrontal cortex (mPFC). Furthermore, M2-induced enhancement of sEPSC frequency was abolished by sulpiride (10 µM), a dopamine D2 receptor antagonist, but not by the dopamine receptor D1 antagonist, SCH23390 (10 μM). In addition, M2 administration significantly increased expression levels of synaptogenesis-related proteins, including p-mTOR and p-TrkB, in the mPFC at 30 min, and increased postsynaptic protein PSD-95 at 24 h. Our results demonstrated that M2 produces rapid antidepressant actions through a novel mechanism via dopamine D2 receptor-mediated enhancement of mPFC neurotransmission.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Walker ER, McGee RE, Druss BG. Mortality in mental disorders and global disease burden implications: a systematic review and meta-analysis. JAMA Psychiat. 2015;72:334–41.
Berman RM, Cappiello A, Anand A, Oren DA, Heninger GR, Charney DS, et al. Antidepressant effects of ketamine in depressed patients. Biol Psychiatry. 2000;47:351–4.
Sinyor M, Schaffer A, Levitt A. The sequenced treatment alternatives to relieve depression (STAR*D) trial: a review. Can J Psychiatry. 2010;55:126–35.
Rush AJ, Trivedi MH, Wisniewski SR, Nierenberg AA, Stewart JW, Warden D, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163:1905–17.
Dailly E, Chenu F, Renard CE, Bourin M. Dopamine, depression and antidepressants. Fundam Clin Pharmacol. 2004;18:601–7.
Kapur S, Mann JJ. Role of the dopaminergic system in depression. Biol Psychiatry. 1992;32:1–17.
Hitchcott PK, Quinn JJ, Taylor JR. Bidirectional modulation of goal-directed actions by prefrontal cortical dopamine. Cereb Cortex. 2007;17:2820–7.
Freund N, Thompson BS, Sonntag K, Meda S, Andersen SL. When the party is over: depressive-like states in rats following termination of cortical D1 receptor overexpression. Psychopharmacology. 2016;233:1191–201.
Zhang B, Guo F, Ma Y, Song Y, Lin R, Shen FY, et al. Activation of D1R/PKA/mTOR signaling cascade in medial prefrontal cortex underlying the antidepressant effects of l-SPD. Sci Rep. 2017;7:3809.
Desormeaux C, Demars F, Davenas E, Jay TM, Lavergne F. Selective activation of D1 dopamine receptors exerts antidepressant-like activity in rats. J Psychopharmacol. 2020;34:1443–8.
Castillo-Gomez E, Varea E, Blasco-Ibanez JM, Crespo C, Nacher J. Effects of chronic dopamine D2R agonist treatment and polysialic acid depletion on dendritic spine density and excitatory neurotransmission in the mPFC of adult rats. Neural Plast. 2016;2016:1615363.
Ruther E, Degner D, Munzel U, Brunner E, Lenhard G, Biehl J, et al. Antidepressant action of sulpiride. Results of a placebo-controlled double-blind trial. Pharmacopsychiatry. 1999;32:127–35.
D’Haenen HA, Bossuyt A. Dopamine D2 receptors in depression measured with single photon emission computed tomography. Biol Psychiatry. 1994;35:128–32.
Willner P, Hale AS, Argyropoulos S. Dopaminergic mechanism of antidepressant action in depressed patients. J Affect Disord. 2005;86:37–45.
Duman RS, Aghajanian GK, Sanacora G, Krystal JH. Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat Med. 2016;22:238–49.
Duman RS, Aghajanian GK. Synaptic dysfunction in depression: potential therapeutic targets. Science. 2012;338:68–72.
Duman RS, Li N, Liu RJ, Duric V, Aghajanian G. Signaling pathways underlying the rapid antidepressant actions of ketamine. Neuropharmacology. 2012;62:35–41.
Li Q, Zhang B, Cao H, Liu W, Guo F, Shen F, et al. Oxytocin exerts antidepressant-like effect by potentiating dopaminergic synaptic transmission in the mPFC. Neuropharmacology. 2020;162:107836.
Socodato R. Dopamine D1 receptor signaling and endocannabinoid cooperate to fuel striatal plasticity: an editorial highlight for “Cyclic AMP-dependent protein kinase and D1 dopamine receptors regulate diacylglycerol lipase-alpha and synaptic 2-arachidonoyl glycerol signaling” on page 334. J Neurochem. 2020;153:297–9.
Hagena H, Manahan-Vaughan D. Dopamine D1/D5, but not D2/D3, receptor dependency of synaptic plasticity at hippocampal mossy fiber synapses that is enabled by patterned afferent stimulation, or spatial learning. Front Synaptic Neurosci. 2016;8:31.
Urakubo H, Yagishita S, Kasai H, Ishii S. Signaling models for dopamine-dependent temporal contiguity in striatal synaptic plasticity. PLoS Comput Biol. 2020;16:e1008078.
Anderson EM, Gomez D, Caccamise A, McPhail D, Hearing M. Chronic unpredictable stress promotes cell-specific plasticity in prefrontal cortex D1 and D2 pyramidal neurons. Neurobiol Stress. 2019;10:100152.
Wang S, Liu S, Wang Q, Sun Y, Yao L, Li D, et al. Dopamine modulates excitatory synaptic transmission by activating presynaptic D1-like dopamine receptors in the RA projection neurons of zebra finches. Front Cell Neurosci. 2020;14:126.
Kawamoto K, Otsuguro K, Ishizuka M, Ito S. Inhibitory effects of dopamine on spinal synaptic transmission via dopamine D1-like receptors in neonatal rats. Br J Pharmacol. 2012;166:788–800.
Koenigs M, Grafman J. The functional neuroanatomy of depression: distinct roles for ventromedial and dorsolateral prefrontal cortex. Behav Brain Res. 2009;201:239–43.
Hamani C, Diwan M, Isabella S, Lozano AM, Nobrega JN. Effects of different stimulation parameters on the antidepressant-like response of medial prefrontal cortex deep brain stimulation in rats. J Psychiatr Res. 2010;44:683–7.
Riga D, Matos MR, Glas A, Smit AB, Spijker S, Van den Oever MC. Optogenetic dissection of medial prefrontal cortex circuitry. Front Syst Neurosci. 2014;8:230.
Santana N, Artigas F. Laminar and cellular distribution of monoamine receptors in rat medial prefrontal cortex. Front Neuroanat. 2017;11:87.
Li N, Lee B, Liu RJ, Banasr M, Dwyer JM, Iwata M, et al. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science. 2010;329:959–64.
Guo F, Zhang B, Fu Z, Ma Y, Gao Y, Shen F, et al. The rapid antidepressant and anxiolytic-like effects of YY-21 involve enhancement of excitatory synaptic transmission via activation of mTOR signaling in the mPFC. Eur Neuropsychopharmacol. 2016;26:1087–98.
Neto FL, Borges G, Torres-Sanchez S, Mico JA, Berrocoso E. Neurotrophins role in depression neurobiology: a review of basic and clinical evidence. Curr Neuropharmacol. 2011;9:530–52.
Cipriani A, Furukawa TA, Salanti G, Chaimani A, Atkinson LZ, Ogawa Y, et al. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: a systematic review and network meta-analysis. Lancet. 2018;391:1357–66.
McEwen BS. Brain on stress: how the social environment gets under the skin. Proc Natl Acad Sci USA. 2012;109(Suppl 2):17180–5.
Ongur D, Price JL. The organization of networks within the orbital and medial prefrontal cortex of rats, monkeys and humans. Cereb Cortex. 2000;10:206–19.
Mayberg HS, Liotti M, Brannan SK, McGinnis S, Mahurin RK, Jerabek PA, et al. Reciprocal limbic-cortical function and negative mood: converging PET findings in depression and normal sadness. Am J Psychiatry. 1999;156:675–82.
Gershon AA, Vishne T, Grunhaus L. Dopamine D2-like receptors and the antidepressant response. Biol Psychiatry. 2007;61:145–53.
Narayanan NS, Laubach M. Top-down control of motor cortex ensembles by dorsomedial prefrontal cortex. Neuron. 2006;52:921–31.
Santana N, Mengod G, Artigas F. Quantitative analysis of the expression of dopamine D1 and D2 receptors in pyramidal and GABAergic neurons of the rat prefrontal cortex. Cereb Cortex. 2009;19:849–60.
Andersen PH, Gingrich JA, Bates MD, Dearry A, Falardeau P, Senogles SE, et al. Dopamine receptor subtypes: beyond the D1/D2 classification. Trends Pharmacol Sci. 1990;11:231–6.
Lavin A, Grace AA. Stimulation of D1-type dopamine receptors enhances excitability in prefrontal cortical pyramidal neurons in a state-dependent manner. Neuroscience. 2001;104:335–46.
Wallace J, Jackson RK, Shotton TL, Munjal I, McQuade R, Gartside SE. Characterization of electrically evoked field potentials in the medial prefrontal cortex and orbitofrontal cortex of the rat: modulation by monoamines. Eur Neuropsychopharmacol. 2014;24:321–32.
Real JI, Simoes AP, Cunha RA, Ferreira SG, Rial D. Adenosine A2A receptors modulate the dopamine D2 receptor-mediated inhibition of synaptic transmission in the mouse prefrontal cortex. Eur J Neurosci. 2018;47:1127–34.
Xia B, Huang X, Sun G, Tao W. Iridoids from Gardeniae fructus ameliorates depression by enhancing synaptic plasticity via AMPA receptor-mTOR signaling. J Ethnopharmacol. 2021;268:113665.
Dadalko OI, Siuta M, Poe A, Erreger K, Matthies HJ, Niswender K, et al. mTORC2/rictor signaling disrupts dopamine-dependent behaviors via defects in striatal dopamine neurotransmission. J Neurosci. 2015;35:8843–54.
Casarotto PC, Girych M, Fred SM, Kovaleva V, Moliner R, Enkavi G, et al. Antidepressant drugs act by directly binding to TRKB neurotrophin receptors. Cell. 2021;184:1299–313 e19.
Acknowledgements
This work was supported by funds from the Strategic Priority Research Program of the Chinese Academy of Sciences (XDA12040220), the National Natural Science Foundation of China (31671049, 31371066 to YL), the National Key New Drug Creation Program of China (No. 2018ZX09711002-002-012), and the Shanghai Municipal Science and Technology Major Project and the Science and Technology Commission of Shanghai Municipality (Nos. 184319071000 and 19140903102 to YL and FG). The work in this manuscript was funded in part by a research grant from Tasly Pharma, Inc.
Author information
Authors and Affiliations
Contributions
YXC and XYJ performed the experiments and analyzed the data. RHX and MZ assisted with behavioral experiments. XHM, FG, and YL designed the study and supervised the project; YXC, FG, and YL wrote and edited the manuscript. All authors approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Che, Yx., Jin, Xy., Xiao, Rh. et al. Antidepressant-like effects of cinnamamide derivative M2 via D2 receptors in the mouse medial prefrontal cortex. Acta Pharmacol Sin 43, 2267–2275 (2022). https://doi.org/10.1038/s41401-021-00854-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-021-00854-7