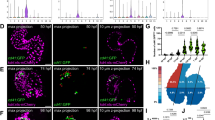
Abstract
Adaptor proteins play crucial roles in signal transduction across diverse signaling pathways. Src-homology 2 domain-containing E (SH2E) is the adaptor protein highly expressed in vascular endothelial cells and myocardium during zebrafish embryogenesis. In this study we investigated the function and mechanisms of SH2E in cardiogenesis. We first analyzed the spatiotemporal expression of SH2E and then constructed zebrafish lines with SH2E deficiency using the CRISPR-Cas9 system. We showed that homozygous mutants developed progressive pericardial edema (PCE), dilated atrium, abnormal atrioventricular looping and thickened atrioventricular wall from 3 days post fertilization (dpf) until death; inducible overexpression of SH2E was able to partially rescue the PCE phenotype. Using transcriptome sequencing analysis, we demonstrated that the MAPK/ERK and NF-κB signaling pathways might be involved in SH2E-deficiency-caused PCE. This study underscores the pivotal role of SH2E in cardiogenesis, and might help to identify innovative diagnostic techniques and therapeutic strategies for congenital heart disease.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Waksman G, Kumaran S, Lubman O. SH2 domains: role, structure and implications for molecular medicine. Expert Rev Mol Med. 2004;6:1–18.
Huang YJ, Zhu L, Tan JY, Guo WC, Yang Z, Shi WH, et al. Correlation between SHP-1 and carotid plaque vulnerability in humans. Cardiovasc Pathol. 2020;49:107258.
Pandit B, Sarkozy A, Pennacchio LA, Carta C, Oishi K, Martinelli S, et al. Gain-of-function RAF1 mutations cause Noonan and LEOPARD syndromes with hypertrophic cardiomyopathy. Nat Genet. 2007;39:1007–12.
Tartaglia M, Mehler EL, Goldberg R, Zampino G, Brunner HG, Kremer H, et al. Mutations in PTPN11, encoding the protein tyrosine phosphatase SHP-2, cause Noonan syndrome. Nat Genet. 2001;29:465–8.
Legius E, Schrander-Stumpel C, Schollen E, Pulles-Heintzberger C, Gewillig M, Fryns JP. PTPN11 mutations in LEOPARD syndrome. J Med Genet. 2002;39:571–4.
Lu YG, Tan H, Ma Q, Li XX, Cui J, Zhang X, et al. SH2 domain-containing protein tyrosine phosphatase-2 (SHP-2) prevents cardiac remodeling after myocardial infarction through ERK/SMAD signaling pathway. Hum Cell. 2021;34:325–34.
Kontaridis MI, Yang WT, Bence KK, Cullen Darragh, Wang B, Bodyak N, et al. Deletion of Ptpn11 (Shp2) in cardiomyocytes causes dilated cardiomyopathy via effects on the extracellular signal–regulated kinase/mitogen-activated protein kinase and RhoA signaling pathways. Circulation. 2008;117:1423–35.
Bonetti M, Paardekooper Overman J, Tessadori F, Noël E, Bakkers J, den Hertog J. Noonan and LEOPARD syndrome Shp2 variants induce heart displacement defects in zebrafish. Development. 2014;141:1961–70.
Flynn DC. Adaptor proteins. Oncogene. 2001;20:6270–2.
Diogo CV, Suski JM, Lebiedzinska M, Karkucinska-Wieckowska A, Wojtala A, Pronicki M, et al. Cardiac mitochondrial dysfunction during hyperglycemia—the role of oxidative stress and p66Shc signaling. Int J Biochem Cell Biol. 2013;45:114–22.
Akhmedov A, Montecucco F, Braunersreuther V, Camici GG, Jakob P, Reiner MF, et al. Genetic deletion of the adaptor protein p66Shc increases susceptibility to short-term ischaemic myocardial injury via intracellular salvage pathways. Eur Heart J. 2015;36:516–26.
Oda T, Kujovich J, Reis M, Newman B, Druker BJ, et al. Identification and characterization of two novel SH2 domain-containing proteins from a yeast two hybrid screen with the ABL tyrosine kinase. Oncogene. 1997;15:1255–62.
Wong KS, Proulx K, Rost MS, Sumanas S. Identification of vasculature‐specific genes by microarray analysis of Etsrp/Etv2 overexpressing zebrafish embryos. Dev Dyn. 2009;238:1836–50.
Chen J, Zhu RF, Li FF, Liang YL, Wang C, Qin YW, et al. MicroRNA-126a directs lymphangiogenesis through interacting with chemokine and Flt4 signaling in zebrafish. Arterioscler Thromb Vasc Biol. 2016;36:2381–93.
Olson EN. Gene regulatory networks in the evolution and development of the heart. Science. 2006;313:1922–7.
Fahed AC, Gelb BD, Seidman JG, Seidman CE. Genetics of congenital heart disease: the glass half empty. Circ Res. 2013;112:707–20.
Asnani A, Peterson RT. The zebrafish as a tool to identify novel therapies for human cardiovascular disease. Dis Model Mech. 2014;7:763–7.
Huang CJ, Tu CT, Hsiao CD, Hsieh FJ, Tsai HJ. Germ‐line transmission of a myocardium‐specific GFP transgene reveals critical regulatory elements in the cardiac myosin light chain 2 promoter of zebrafish. Dev Dyn. 2003;228:30–40.
Kawakami K, Takeda H, Kawakami N, Kobasashi M, Matsuda N, Mishina M. A transposon-mediated gene trap approach identifies developmentally regulated genes in zebrafish. Dev Cell. 2004;7:133–44.
Brown DR, Samsa LA, Qian L, Liu JD. Advances in the study of heart development and disease using zebrafish. J Cardiovasc Dev Dis. 2016;3:13.
Stainier DY. Zebrafish genetics and vertebrate heart formation. Nat Rev Genet. 2001;2:39–48.
Bakkers J. Zebrafish as a model to study cardiac development and human cardiac disease. Cardiovasc Res. 2011;91:279–88.
González-Rosa JM. Zebrafish models of cardiac disease: From fortuitous mutants to precision medicine. Cir Res. 2022;130:1803–26.
Doyon Y, McCammon JM, Miller JC, Faraji F, Ngo C, Katibah GE, et al. Heritable targeted gene disruption in zebrafish using designed zinc-finger nucleases. Nat Biotechnol. 2008;26:702–8.
Bedell VM, Wang Y, Campbell JM, Poshusta TL, Starker CG, Krug II RG, et al. In vivo genome editing using a high-efficiency TALEN system. Nature. 2012;491:114–8.
Hwang WY, Fu YF, Reyon D, Maeder ML, Tsai SQ, Sander JD, et al. Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat Biotechnol. 2013;31:227–9.
Li FF, Liang YL, Han XS, Guan YN, Chen J, Wu P, et al. ADP receptor P2y12 prevents excessive primitive hematopoiesis in zebrafish by inhibiting Gata1. Acta Pharmacol Sin. 2021;42:414–21.
Thisse C, Thisse B. High-resolution in situ hybridization to whole-mount zebrafish embryos. Nat Protoc. 2008;3:59–69.
He JB, Mo DS, Chen JY, Luo LF. Combined whole-mount fluorescence in situ hybridization and antibody staining in zebrafish embryos and larvae. Nat Protoc. 2020;15:3361–79.
Copper JE, Budgeon LR, Foutz CA, van Rossum DB, Vanselow DJ, hubley MJ, et al. Comparative analysis of fixation and embedding techniques for optimized histological preparation of zebrafish. Comp Biochem Physiol C Toxicol Pharmacol. 2018;208:38–46.
Zhang RL, Yang JC, Zhu J, Xu XL. Depletion of zebrafish Tcap leads to muscular dystrophy via disrupting sarcomere–membrane interaction, not sarcomere assembly. Hum Mol Genet. 2009;18:4130–40.
French VM, van de Laar IM, Wessels MW, Rohe C, Roos-Hesselink JW, Wang GL, et al. NPHP4 variants are associated with pleiotropic heart malformations. Circ Res. 2012;110:1564–74.
Slanchev K, Pütz M, Schmitt A, Kramer-Zucker A, Walz G. Nephrocystin-4 is required for pronephric duct-dependent cloaca formation in zebrafish. Hum Mol Genet. 2011;20:3119–28.
Schleich JM, Abdulla T, Summers R, Houyel L. An overview of cardiac morphogenesis. Arch Cardiovasc Dis. 2013;106:612–23.
Desgrange A, Le Garrec JF, Meilhac SM. Left-right asymmetry in heart development and disease: forming the right loop. Development. 2018;145:dev162776.
Hu B, Lelek S, Spanjaard B, EI-Sammak H, Simões MG, Mintcheva J, et al. Origin and function of activated fibroblast states during zebrafish heart regeneration. Nat Genet. 2022;54:1227–37.
de Sena-Tomás C, Aleman AG, Ford C, Varshney A, Yao D, Harrington JK, et al. Activation of Nkx2. 5 transcriptional program is required for adult myocardial repair. Nat Commun. 2022;13:2970.
Lints TJ, Parsons LM, Hartley L, Lyons I, Harvey RP. Nkx-2. 5: a novel murine homeobox gene expressed in early heart progenitor cells and their myogenic descendants. Development. 1993;119:419–31.
Lepilina A, Coon AN, Kikuchi K, Holdway JE, Roberts RW, Burns CG, et al. A dynamic epicardial injury response supports progenitor cell activity during zebrafish heart regeneration. Cell. 2006;127:607–19.
Singleman C, Holtzman NG. Analysis of postembryonic heart development and maturation in the zebrafish, Danio rerio. Dev Dyn. 2012;241:1993–2004.
Zenker M, Edouard T, Blair JC, Cappa M. Noonan syndrome: improving recognition and diagnosis. Arch Dis Child. 2022;107:1073–8.
Jiménez-Martínez M, Stamatakis K, Fresno M. The dual-specificity phosphatase 10 (DUSP10): its role in cancer, inflammation, and immunity. Int J Mol Sci. 2019;20:1626.
Gordon JW, Shaw JA, Kirshenbaum LA. Multiple facets of NF-κB in the heart: to be or not to NF-κB. Circ Res. 2011;108:1122–32.
Kim BY, Kang DO, Oh WK, Kim JH, Choi YK, Jang JS, et al. Involvement of SH2-SH2-SH3 domain of phospholipase Cγ1 in NF-κB signaling. FEBS Lett. 2000;472:45–9.
Zelarayan L, Renger A, Noack C, Zafiriou MP, Gehrke C, van der Nagel R, et al. NF-κB activation is required for adaptive cardiac hypertrophy. Cardiovasc Res. 2009;84:416–24.
Ishida H, Kogaki S, Narita J, Ichimori H, Nawa N, Okada Y, et al. LEOPARD-type SHP2 mutant Gln510Glu attenuates cardiomyocyte differentiation and promotes cardiac hypertrophy via dysregulation of Akt/GSK-3β/β-catenin signaling. Am J Physiol Heart Circ Physiol. 2011;301:H1531–9.
Marin TM, Keith K, Davies B, Conner DA, Guha P, Kalaitzidis D, et al. Rapamycin reverses hypertrophic cardiomyopathy in a mouse model of LEOPARD syndrome–associated PTPN11 mutation. J Clin Invest. 2011;121:1026–43.
Schramm C, Fine DM, Edwards MA, Reeb AN, Krenz M. The PTPN11 loss-of-function mutation Q510E-Shp2 causes hypertrophic cardiomyopathy by dysregulating mTOR signaling. Am J Physiol Heart Circ Physiol. 2012;302:H231–43.
Acknowledgements
We thank Dr. Nathan Lawson from UMass Chan Medical School and Dr. Jing-wei Xiong from Peking University for providing the Tg(fli1:EGFP) fish line and CRISPR/Cas9 plasmids, respectively. This work was funded by the National Key Research and Development Program of China (2019YFA0802700), the National Natural Science Foundation of China (92168206, 82400541) and the Postdoctoral Fellowship Program of CPSF under Grant Number GZC20242284.
Author information
Authors and Affiliations
Contributions
QJ, YLL and JC conceived and designed the project. YLL carried out the experiments and analyzed the data. YXH and HMY contributed to confirming and maintaining the knockout line. YXH helped to perform the pharmacological treatment and analyze the data. FFL and JC directed the FISH analysis. ZFG supported the zebrafish platform. CL supervised the study. YLL and YXH drafted the manuscript. YLL, YXH and FFL reviewed and edited the manuscript. QJ supervised the study and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest. Shanghai Institute of Nutrition and Health, Chinese Academy of Sciences has applied for a patent, China Patent Application No. 202311014379.7 by inventors Qing Jing, Yang-xi Hu, Yu-lai Liang and Zhi-hua Liu for the usage of L-selenomethionine in antioxidant stress and alleviation of cardiac developmental defects caused by SH2E gene mutations described in this paper under the support of the Program of EnShi TuJia and Miao Autonomous Prefecture Bureau of Scientific and Technological Affairs.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liang, Yl., Hu, Yx., Li, Ff. et al. Adaptor protein Src-homology 2 domain containing E (SH2E) deficiency induces heart defect in zebrafish. Acta Pharmacol Sin 46, 404–415 (2025). https://doi.org/10.1038/s41401-024-01392-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-024-01392-8
Keywords
This article is cited by
-
1,24,25(OH)3D3 is a fully active catabolite of vitamin D in keratinocytes
Scientific Reports (2025)