Abstract
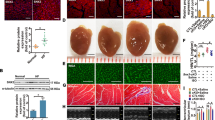
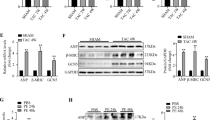
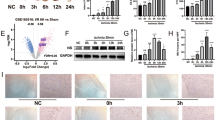
Sorting nexins (SNXs) as the key regulators of sorting cargo proteins are involved in diverse diseases. SNXs can form the specific reverse vesicle transport complex (SNXs-retromer) with vacuolar protein sortings (VPSs) to sort and modulate recovery and degradation of cargo proteins. Our previous study has shown that SNX3-retromer promotes both STAT3 activation and nuclear translocation in cardiomyocytes, suggesting that SNX3 might be a critical regulator in the heart. In this study we investigated the role of SNX3 in the development of pathological cardiac hypertrophy and heart failure. We generated abdominal aortic constriction (AAC) rat model and transverse aortic constriction (TAC) mouse model; hypertrophic neonatal rat cardiomyocytes (NRCMs) were induced by exposure to isoproterenol (10 μM). We showed that the expression of SNX3 was significantly upregulated in ISO-treated NRCMs and in the failing heart of AAC rats. Overexpression of SNX3 by intramyocardial injection of Ad-SNX3 induced heart failure in rats, and increased the susceptibility of NRCMs to ISO-induced myocardial injury in vitro. In contrast, conditional knockout of SNX3 in cardiac tissue in mice rescued the detrimental heart function in TAC mice, and knockdown of SNX3 protected against ISO-induced injury in NRCMs and AAC rats. We then conducted immunoprecipitation-based mass spectrometry and localized surface plasmon resonance, and demonstrated a direct interaction between SNX3-retromer and high mobility group box 1 (HMGB1), which mediated the efflux of nuclear HMGB1. Moreover, overexpression of HMGB1 in NRCMs inhibited the pro-hypertrophic effects of SNX3, whereas knockdown of HMGB1 abolished the protective effect of SNX3-deficiency. These results suggest that HMGB1 might be a direct cargo protein of SNX3-retromer, and its interaction with SNX3 promotes its efflux from the nucleus, leading to the pathological development of heart failure.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Dorn GW 2nd. The fuzzy logic of physiological cardiac hypertrophy. Hypertension. 2007;49:962–70.
Saucerman JJ, Tan PM, Buchholz KS, McCulloch AD, Omens JH. Mechanical regulation of gene expression in cardiac myocytes and fibroblasts. Nat Rev Cardiol. 2019;16:361–78.
Tagawa H, Wang N, Narishige T, Ingber DE, Zile MR, Cooper GT. Cytoskeletal mechanics in pressure-overload cardiac hypertrophy. Circ Res. 1997;80:281–9.
Tsuda T. Clinical assessment of ventricular wall stress in understanding compensatory hypertrophic response and maladaptive ventricular remodeling. J Cardiovasc Dev Dis. 2021;8:122.
Hanley SE, Cooper KF. Sorting nexins in protein homeostasis. Cells. 2020;10:17.
Abubakar YS, Zheng W, Olsson S, Zhou J. Updated insight into the physiological and pathological roles of the retromer complex. Int J Mol Sci. 2017;18:1601.
Lucas M, Gershlick DC, Vidaurrazaga A, Rojas AL, Bonifacino JS, Hierro A. Structural mechanism for cargo recognition by the retromer complex. Cell. 2016;167:1623–35.
Lu L, Hong W. From endosomes to the trans-Golgi network. Semin Cell Dev Biol. 2014;31:30–9.
Harterink M, Port F, Lorenowicz MJ, McGough IJ, Silhankova M, Betist MC, et al. A SNX3-dependent retromer pathway mediates retrograde transport of the Wnt sorting receptor Wntless and is required for Wnt secretion. Nat Cell Biol. 2011;13:914–23.
Zhang P, Wu Y, Belenkaya TY, Lin X. SNX3 controls Wingless/Wnt secretion through regulating retromer-dependent recycling of Wntless. Cell Res. 2011;21:1677–90.
McGough IJ, de Groot REA, Jellett AP, Betist MC, Varandas KC, Danson CM, et al. SNX3-retromer requires an evolutionary conserved MON2:DOPEY2:ATP9A complex to mediate Wntless sorting and Wnt secretion. Nat Commun. 2018;9:3737.
Li J, Li C, Zhang D, Shi D, Qi M, Feng J, et al. SNX13 reduction mediates heart failure through degradative sorting of apoptosis repressor with caspase recruitment domain. Nat Commun. 2014;5:5177.
Lu J, Xu S, Huo Y, Sun D, Hu Y, Wang J, et al. Sorting nexin 3 induces heart failure via promoting retromer-dependent nuclear trafficking of STAT3. Cell Death Differ. 2021;28:2871–87.
Raucci A, Di Maggio S, Scavello F, D’Ambrosio A, Bianchi ME, Capogrossi MC. The Janus face of HMGB1 in heart disease: a necessary update. Cell Mol Life Sci. 2019;76:211–29.
Funayama A, Shishido T, Netsu S, Narumi T, Kadowaki S, Takahashi H, et al. Cardiac nuclear high mobility group box 1 prevents the development of cardiac hypertrophy and heart failure. Cardiovasc Res. 2013;99:657–64.
Zhang L, Liu M, Jiang H, Yu Y, Yu P, Tong R, et al. Extracellular high-mobility group box 1 mediates pressure overload-induced cardiac hypertrophy and heart failure. J Cell Mol Med. 2016;20:459–70.
Li Q, Li ZM, Sun SY, Wang LP, Wang PX, Guo Z, et al. PARP1 interacts with HMGB1 and promotes its nuclear export in pathological myocardial hypertrophy. Acta Pharmacol Sin. 2019;40:589–98.
Wang L, Wang P, Xu S, Li Z, Duan DD, Ye J, et al. The cross-talk between PARylation and SUMOylation in C/EBPβ at K134 site participates in pathological cardiac hypertrophy. Int J Biol Sci. 2022;18:783–99.
Guo Z, Hu YH, Feng GS, Valenzuela Ripoll C, Li ZZ, Cai SD, et al. JMJD6 protects against isoproterenol-induced cardiac hypertrophy via inhibition of NF-κB activation by demethylating R149 of the p65 subunit. Acta Pharmacol Sin. 2023;44:1777–89.
Li H, Li Q, Zhang Y, Liu W, Gu B, Narumi T, et al. Novel treatment of hypertension by specifically targeting E2F for restoration of endothelial dihydrofolate reductase and eNOS function under oxidative stress. Hypertension. 2019;73:179–89.
Lu J, Sun D, Liu Z, Li M, Hong H, Liu C, et al. SIRT6 suppresses isoproterenol-induced cardiac hypertrophy through activation of autophagy. Transl Res. 2016;172:96–112.
Glembotski CC. Classic studies of cultured cardiac myocyte hypertrophy: interview with a transformer. Circ Res. 2013;113:1112–6.
Pellegrini L, Foglio E, Pontemezzo E, Germani A, Russo MA, Limana F. HMGB1 and repair: focus on the heart. Pharmacol Ther. 2019;196:160–82.
Kastritis PL, Moal IH, Hwang H, Weng Z, Bates PA, Bonvin AM, et al. A structure-based benchmark for protein-protein binding affinity. Protein Sci. 2011;20:482–91.
Kümmel A, Gross S, Feldtmann R, Chamling B, Strohbach A, Lehnert K, et al. High-mobility group box protein 1 is an independent prognostic marker for all-cause mortality in patients with dilated cardiomyopathy. Am J Cardiol. 2022;178:119–23.
Raucci A, Capogrossi MC. Nuclear Hmgb1: the fix for the failing heart. JACC Basic Transl Sci 2019;4:248–50.
Takahashi T, Shishido T, Kinoshita D, Watanabe K, Toshima T, Sugai T, et al. Cardiac nuclear high-mobility group box 1 ameliorates pathological cardiac hypertrophy by inhibiting DNA damage response. JACC Basic Transl Sci. 2019;4:234–47.
Huang J, Tiu AC, Jose PA, Yang J. Sorting nexins: role in the regulation of blood pressure. FEBS J. 2023;290:600–19.
Vieira N, Rito T, Correia-Neves M, Sousa N. Sorting out sorting nexins functions in the nervous system in health and disease. Mol Neurobiol. 2021;58:4070–106.
Temkin P, Lauffer B, Jäger S, Cimermancic P, Krogan NJ, von Zastrow M. SNX27 mediates retromer tubule entry and endosome-to-plasma membrane trafficking of signalling receptors. Nat Cell Biol. 2011;13:715–21.
Chen C, Garcia-Santos D, Ishikawa Y, Seguin A, Li L, Fegan KH, et al. Snx3 regulates recycling of the transferrin receptor and iron assimilation. Cell Metab. 2013;17:343–52.
Chiow KH, Tan Y, Chua RY, Huang D, Ng ML, Torta F, et al. SNX3-dependent regulation of epidermal growth factor receptor (EGFR) trafficking and degradation by aspirin in epidermoid carcinoma (A-431) cells. Cell Mol Life Sci. 2012;69:1505–21.
Joyal JS, Nim S, Zhu T, Sitaras N, Rivera JC, Shao Z, et al. Subcellular localization of coagulation factor II receptor-like 1 in neurons governs angiogenesis. Nat Med. 2014;20:1165–73.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82473914, 82173808, U21A20419, 81903606), Guangzhou Basic and Applied Basic Research Project (2024A04J10013, 202102021272), Guangdong Provincial Key Laboratory of Construction Foundation (2017B030314030), and Traditional Chinese Medicine Bureau of Guangdong Province (20231134, 20231158).
Author information
Authors and Affiliations
Contributions
JL and PQL conceived the study and revised the manuscript. HL and MXP designed and performed majority of the experiments and data analyses. RXY, JXC, YMW, PXW, YHH and DYP performed several in-vitro experiments. HL, MXP and JL wrote the manuscript. PQL provided scientific advice. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, H., Peng, Mx., Yang, Rx. et al. SNX3 mediates heart failure by interacting with HMGB1 and subsequently facilitating its nuclear-cytoplasmic translocation. Acta Pharmacol Sin 46, 964–975 (2025). https://doi.org/10.1038/s41401-024-01436-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41401-024-01436-z